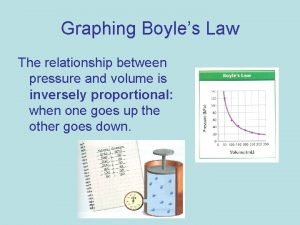
Gas Laws Boyles Law Charles law GayLussacs Law

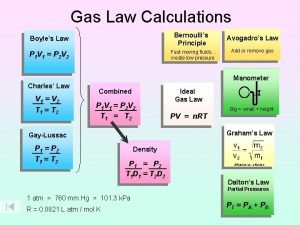
Gas Laws Boyle’s Law Charle’s law Gay-Lussac’s Law Avogadro’s Law Dalton’s Law Henry’s Law 1

What is Pressure? • Gas molecules cause pressure 2
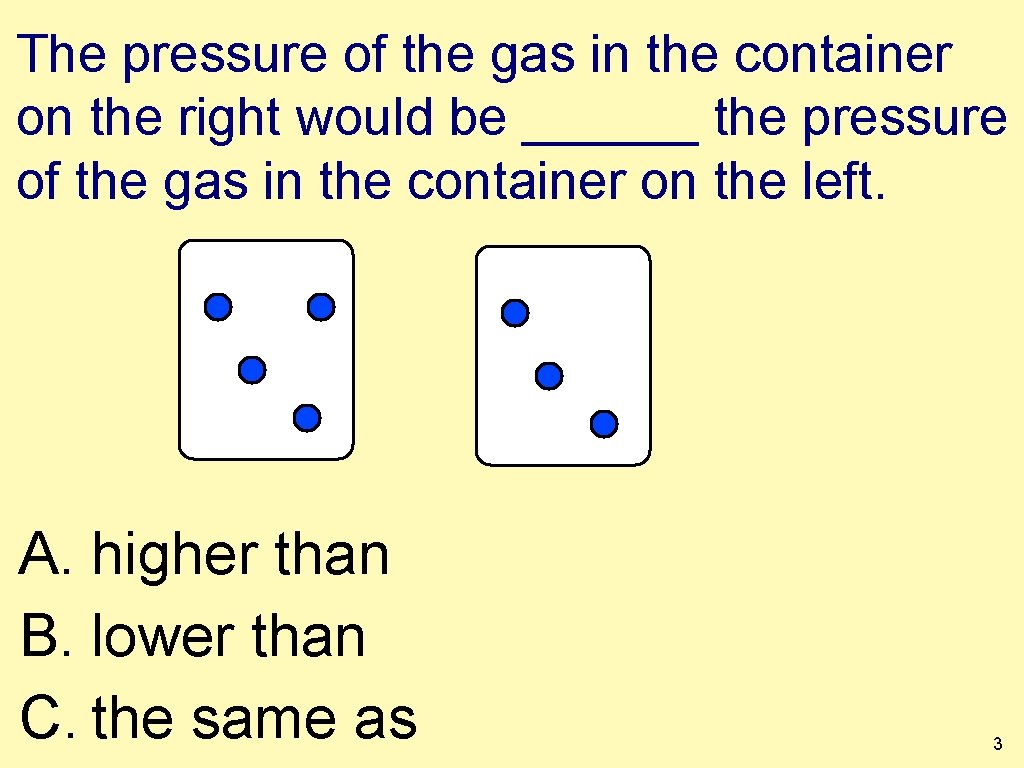
The pressure of the gas in the container on the right would be ______ the pressure of the gas in the container on the left. A. higher than B. lower than C. the same as 3
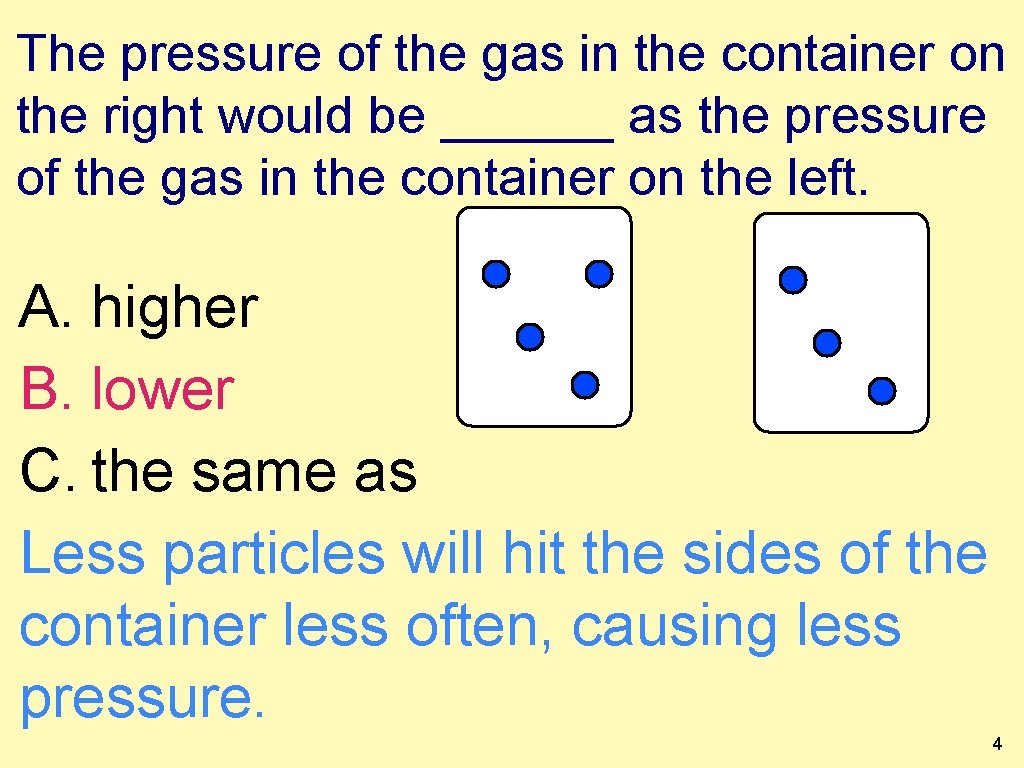
The pressure of the gas in the container on the right would be ______ as the pressure of the gas in the container on the left. A. higher B. lower C. the same as Less particles will hit the sides of the container less often, causing less pressure. 4
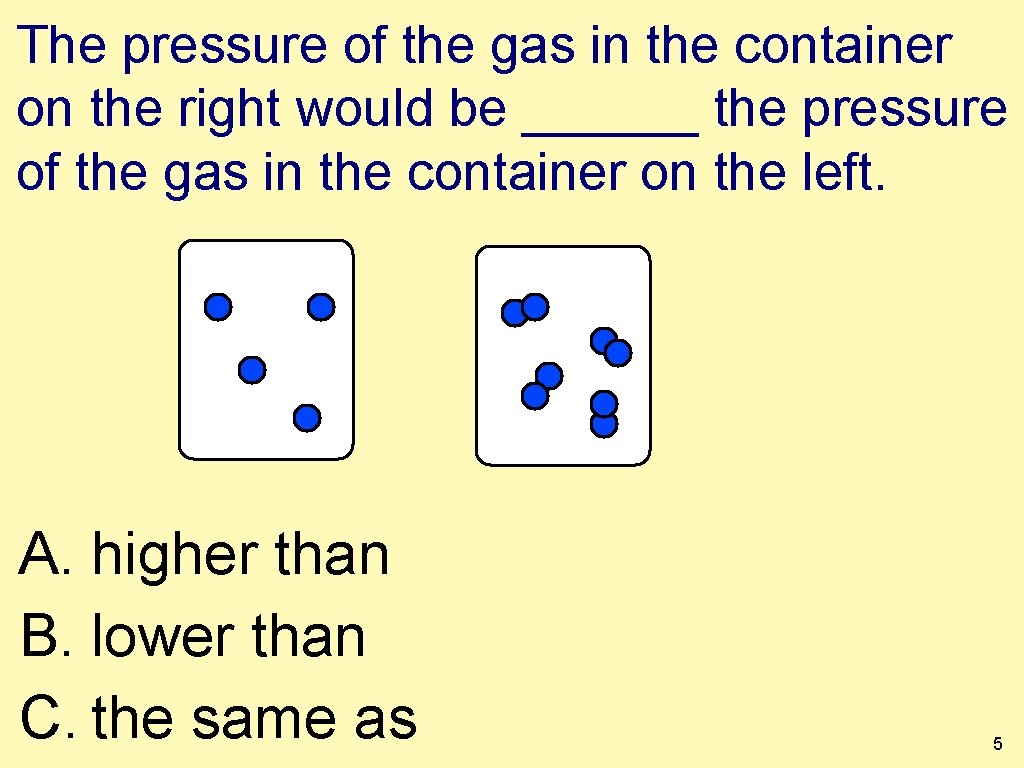
The pressure of the gas in the container on the right would be ______ the pressure of the gas in the container on the left. A. higher than B. lower than C. the same as 5
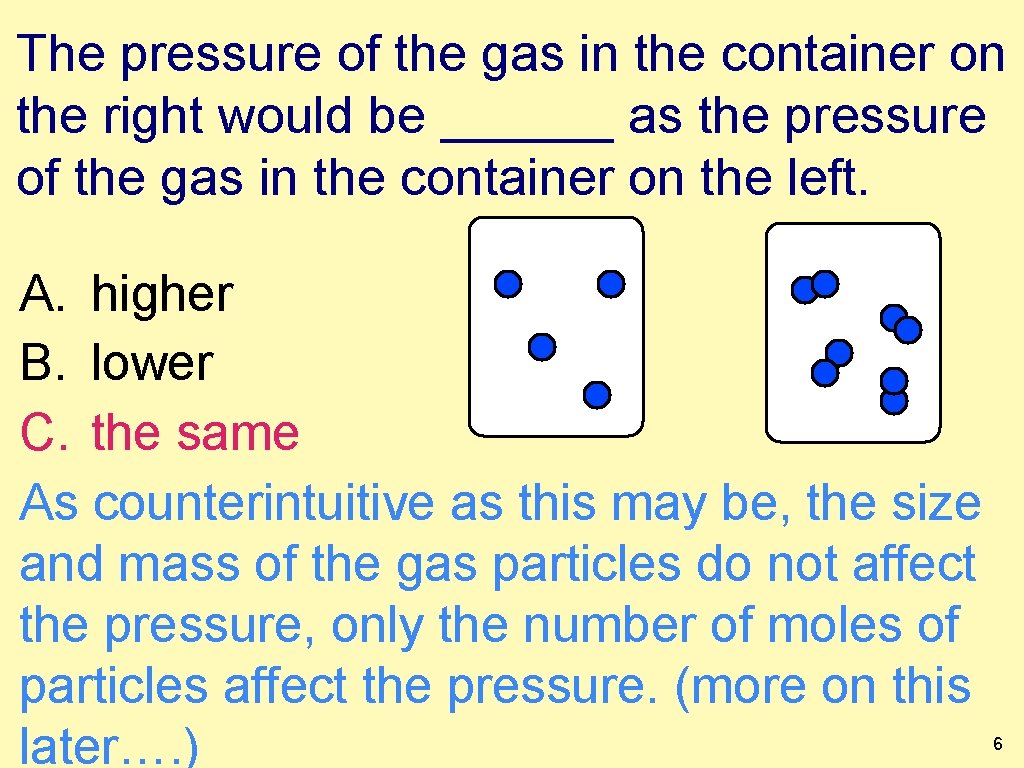
The pressure of the gas in the container on the right would be ______ as the pressure of the gas in the container on the left. A. higher B. lower C. the same As counterintuitive as this may be, the size and mass of the gas particles do not affect the pressure, only the number of moles of particles affect the pressure. (more on this later…. ) 6
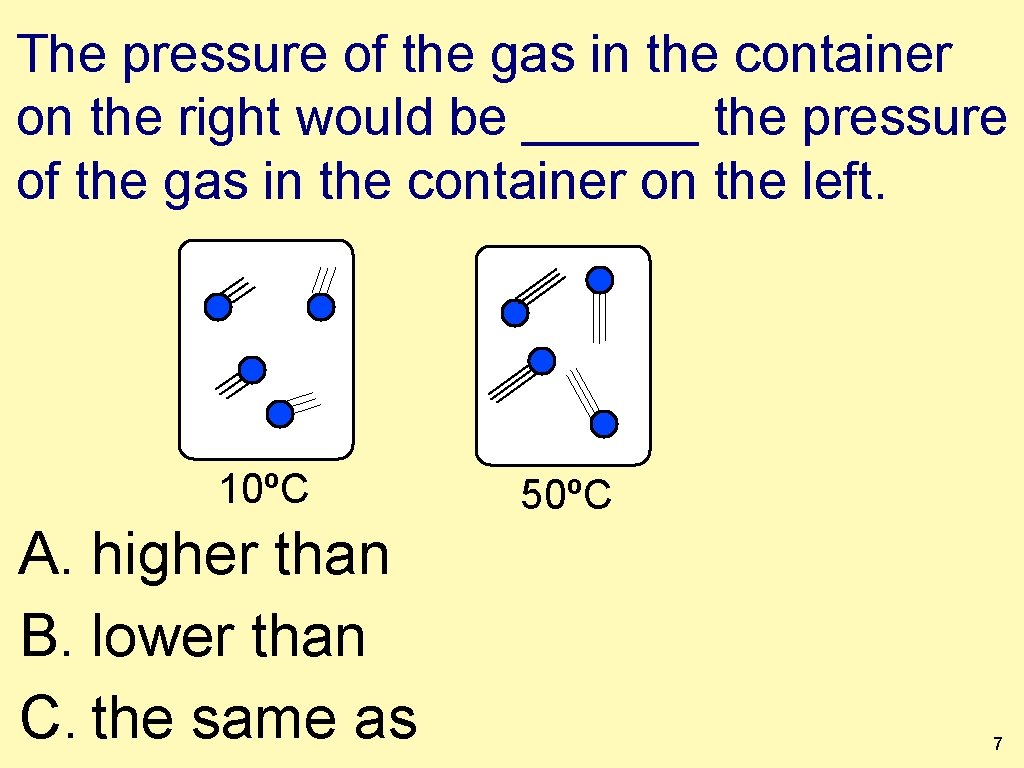
The pressure of the gas in the container on the right would be ______ the pressure of the gas in the container on the left. 10ºC A. higher than B. lower than C. the same as 50ºC 7
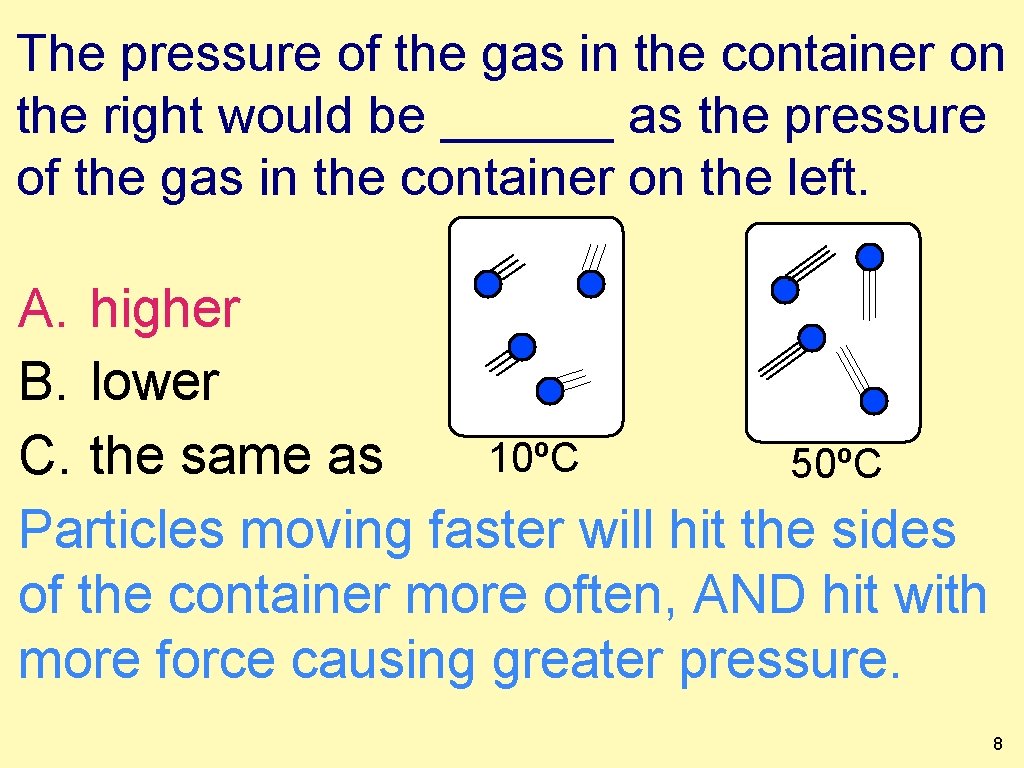
The pressure of the gas in the container on the right would be ______ as the pressure of the gas in the container on the left. A. higher B. lower 10ºC C. the same as 50ºC Particles moving faster will hit the sides of the container more often, AND hit with more force causing greater pressure. 8

Air Pressure • Air molecules cause air pressure • The weight of all the air on top of us causes the pressure. • Since air is a fluid, it pushes “all around, ” not just from the top. • you will study fluid dynamics when you go to physics 1&2 9

Barometer Measuring Air Pressure • Standard air pressure • 760 mm of Hg • 760 torr • 29. 92 inches of Hg • 1 atm • 101. 3 k. Pa 10
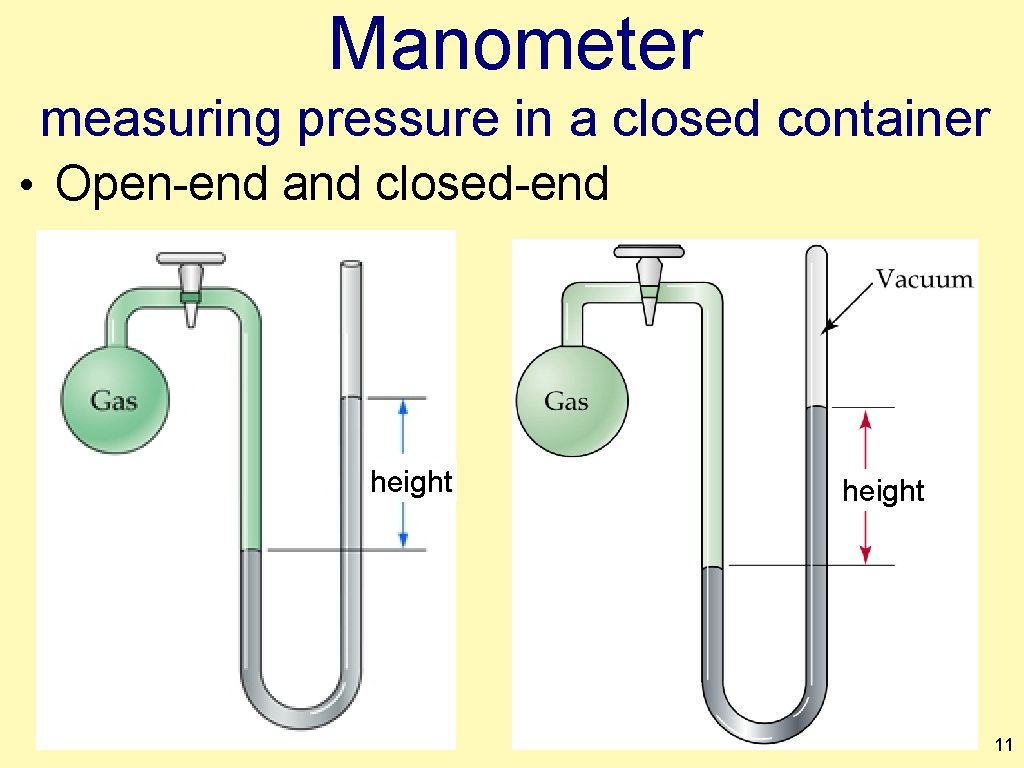
Manometer measuring pressure in a closed container • Open-end and closed-end height 11
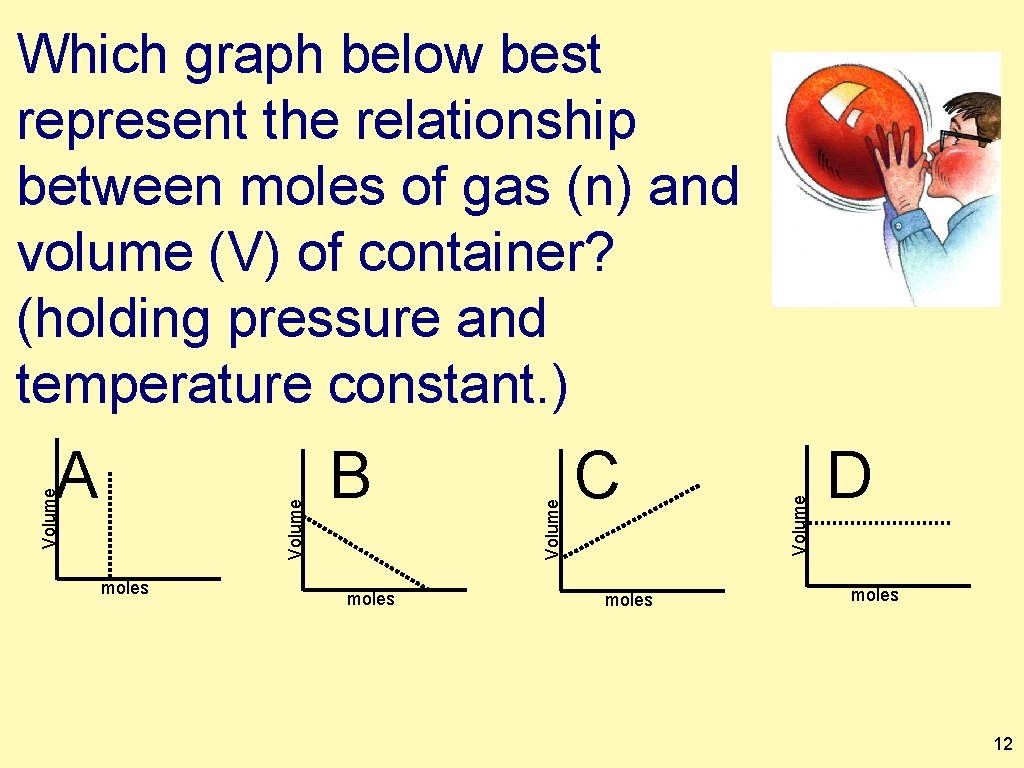
moles B moles C moles Volume A Volume Which graph below best represent the relationship between moles of gas (n) and volume (V) of container? (holding pressure and temperature constant. ) D moles 12
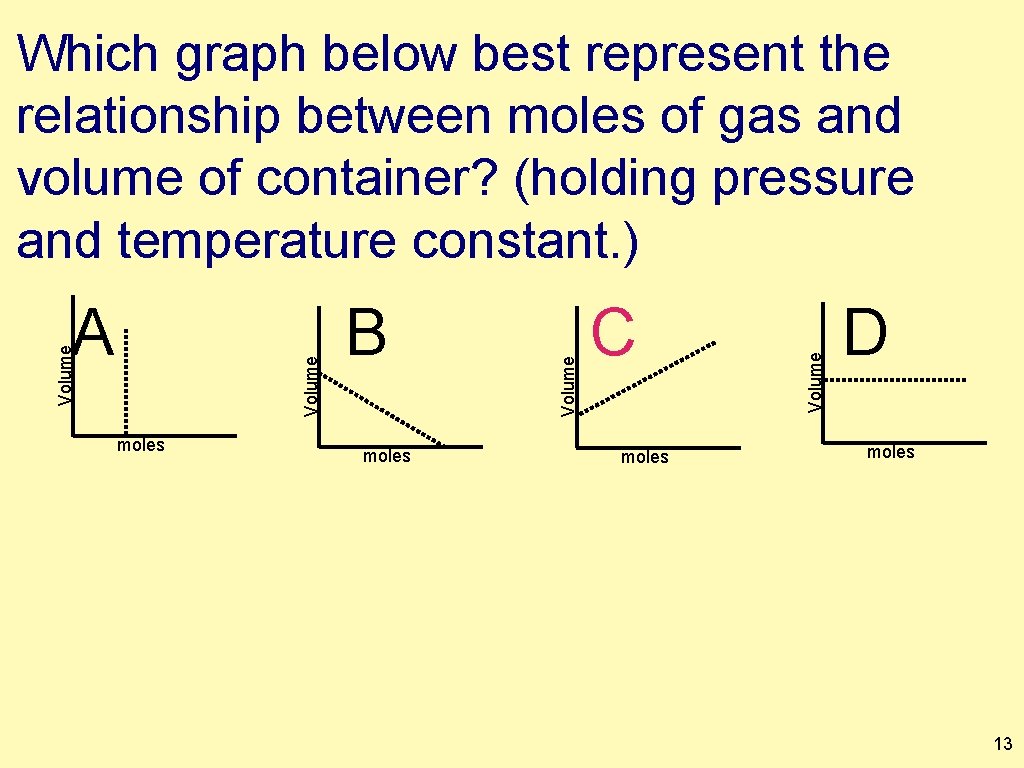
moles B moles C moles Volume A Volume Which graph below best represent the relationship between moles of gas and volume of container? (holding pressure and temperature constant. ) D moles 13

number of molecules and volume • moles and volume • n&V • increase the number of moles, (while pressure & temp remain constant) and the volume will increase. • This is a direct relationship • n is proportional to V • k = a constant 14
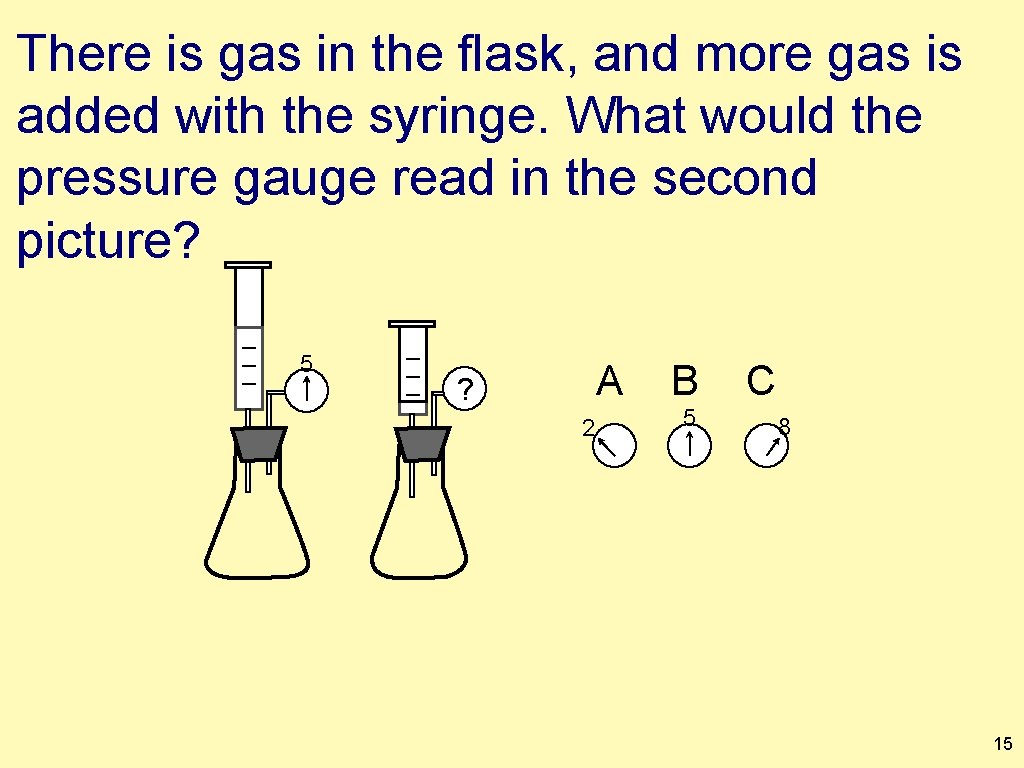
There is gas in the flask, and more gas is added with the syringe. What would the pressure gauge read in the second picture? 5 A ? 2 B 5 C 8 15
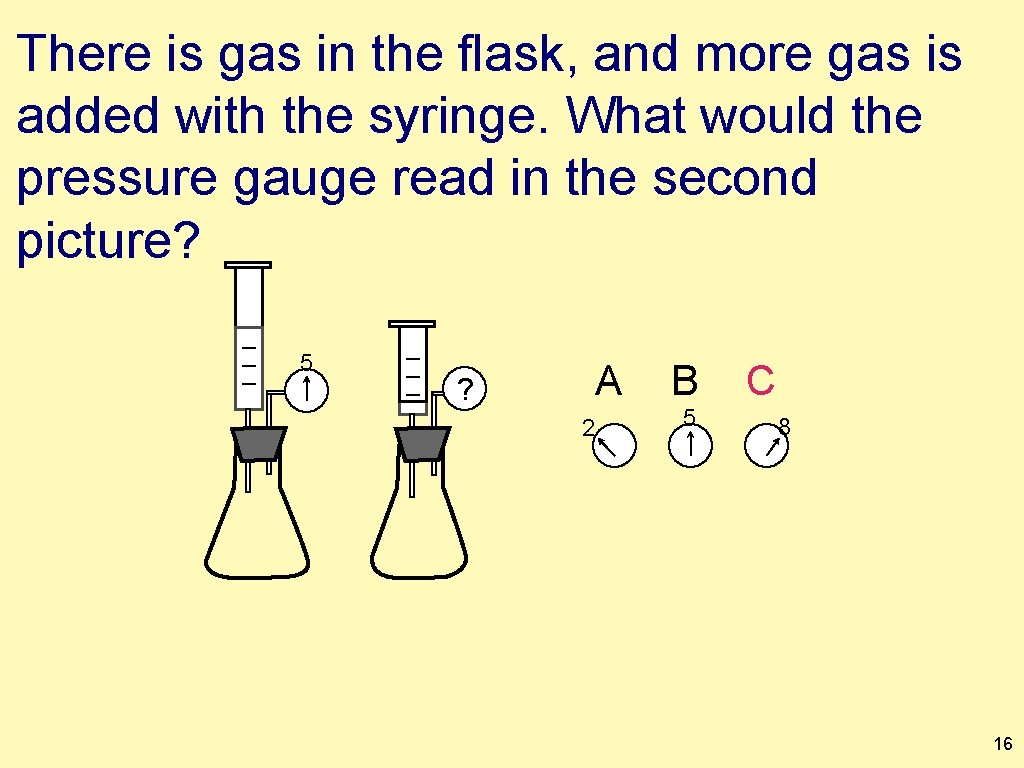
There is gas in the flask, and more gas is added with the syringe. What would the pressure gauge read in the second picture? 5 ? A 2 B 5 C 8 16

number of molecules and pressure • moles and pressure • n&P • increase the number of moles (while maintaining constant volume and temp) and the pressure increases. • this is a direct relationship • n is proportional to P • k = a constant 17

The height of the piston in the container on the right would be ______ the height of the gas in the container on the left. ? ? A. higher than B. lower than C. the same as 18
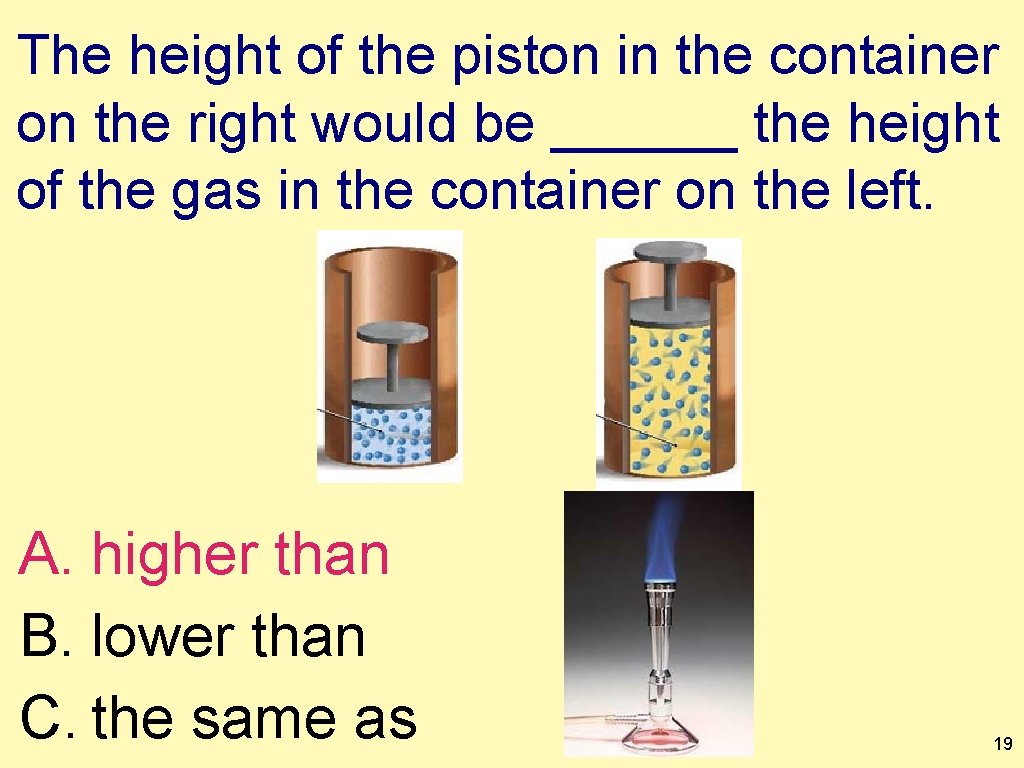
The height of the piston in the container on the right would be ______ the height of the gas in the container on the left. A. higher than B. lower than C. the same as 19

temperature and volume • T&V • increase the temperature (while pressure remains constant) and the volume increases. • this is a direct relationship • V is proportional to T • k = a constant 20

The pressure of the gas in the container on the right would be ______ the pressure of the gas in the container on the left. (Temperature and moles remain constant. ) ? ? A. higher than B. lower than C. the same as 21
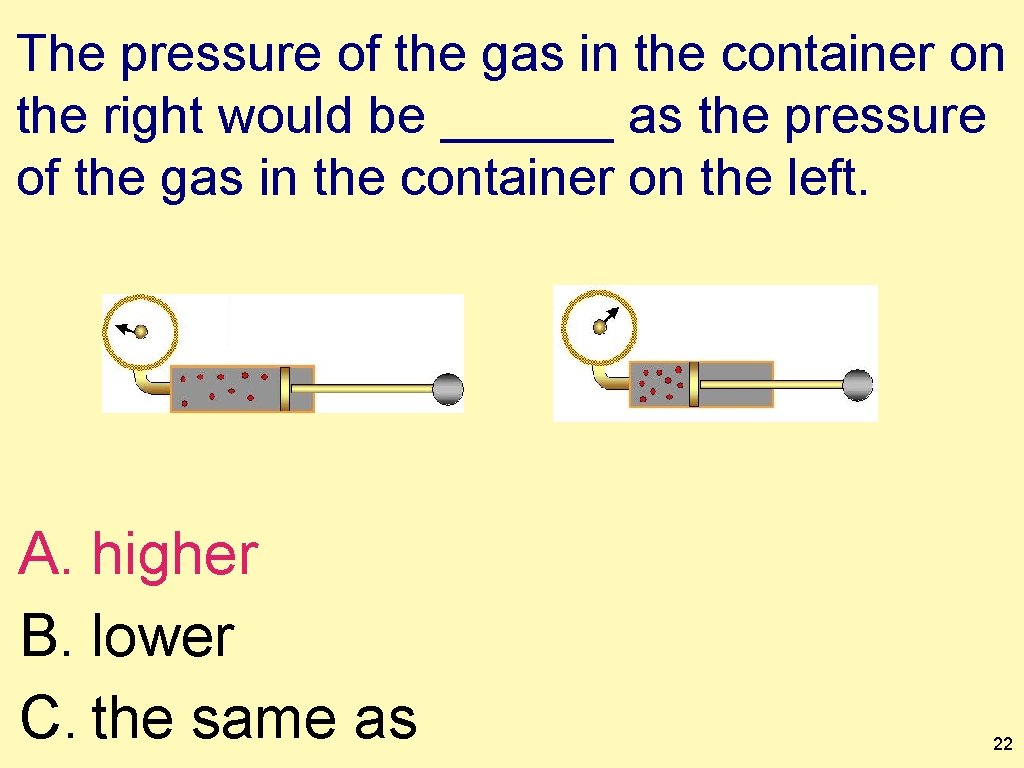
The pressure of the gas in the container on the right would be ______ as the pressure of the gas in the container on the left. A. higher B. lower C. the same as 22
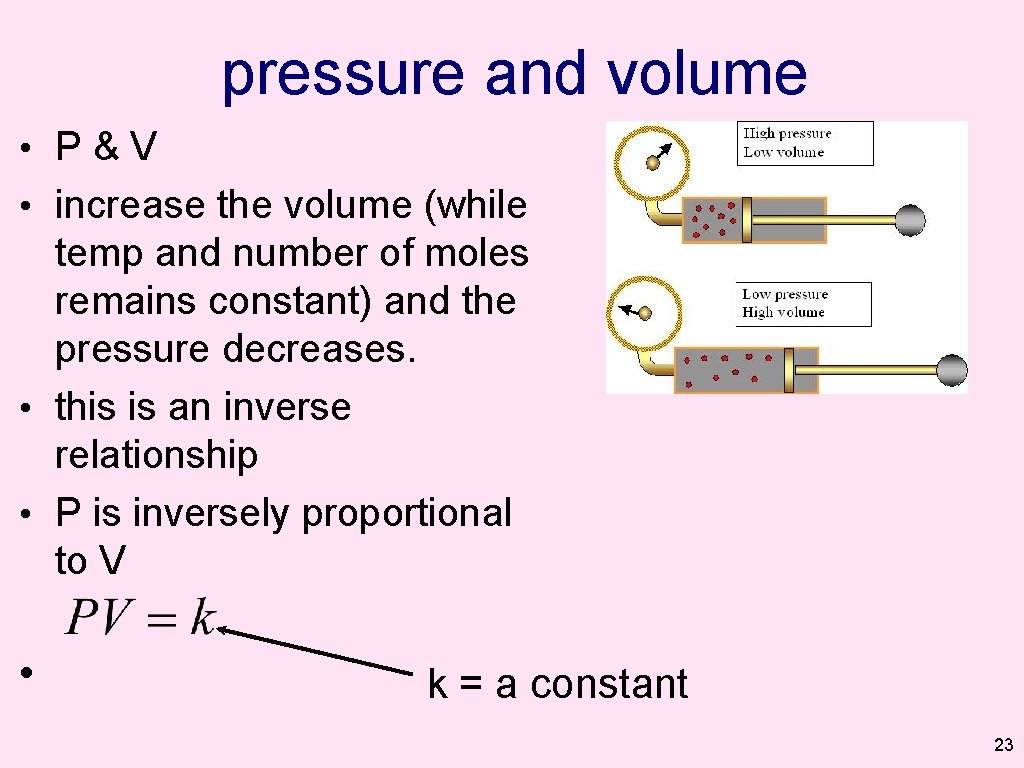
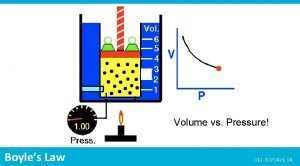
pressure and volume • P&V • increase the volume (while temp and number of moles remains constant) and the pressure decreases. • this is an inverse relationship • P is inversely proportional to V • k = a constant 23
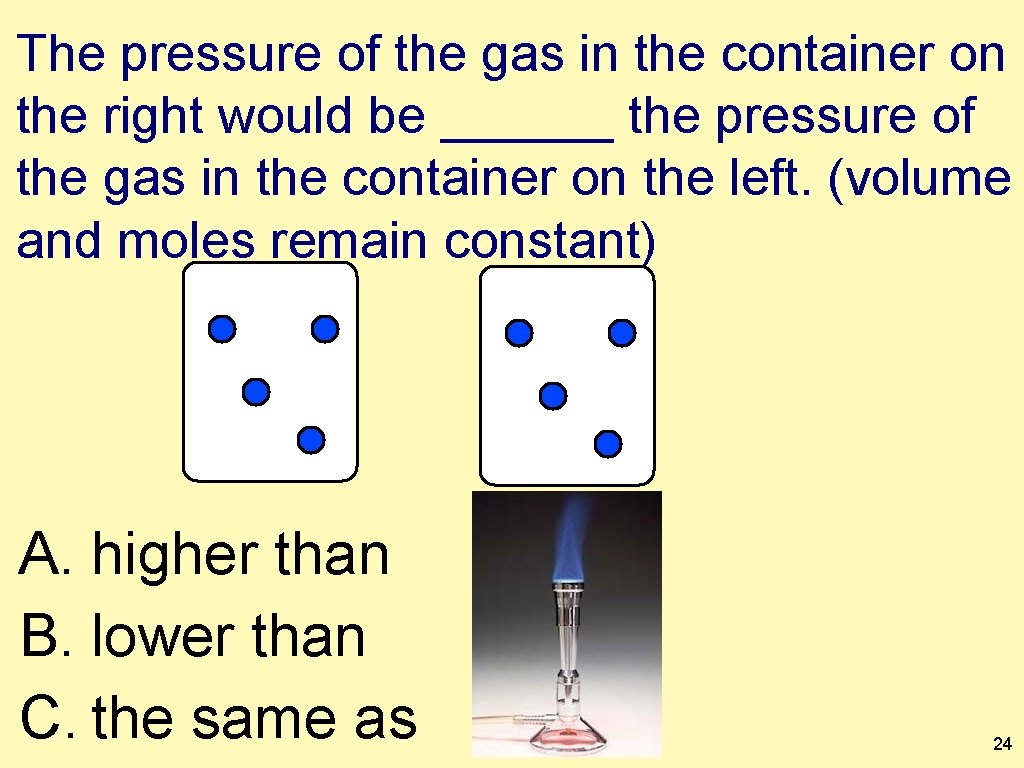
The pressure of the gas in the container on the right would be ______ the pressure of the gas in the container on the left. (volume and moles remain constant) A. higher than B. lower than C. the same as 24
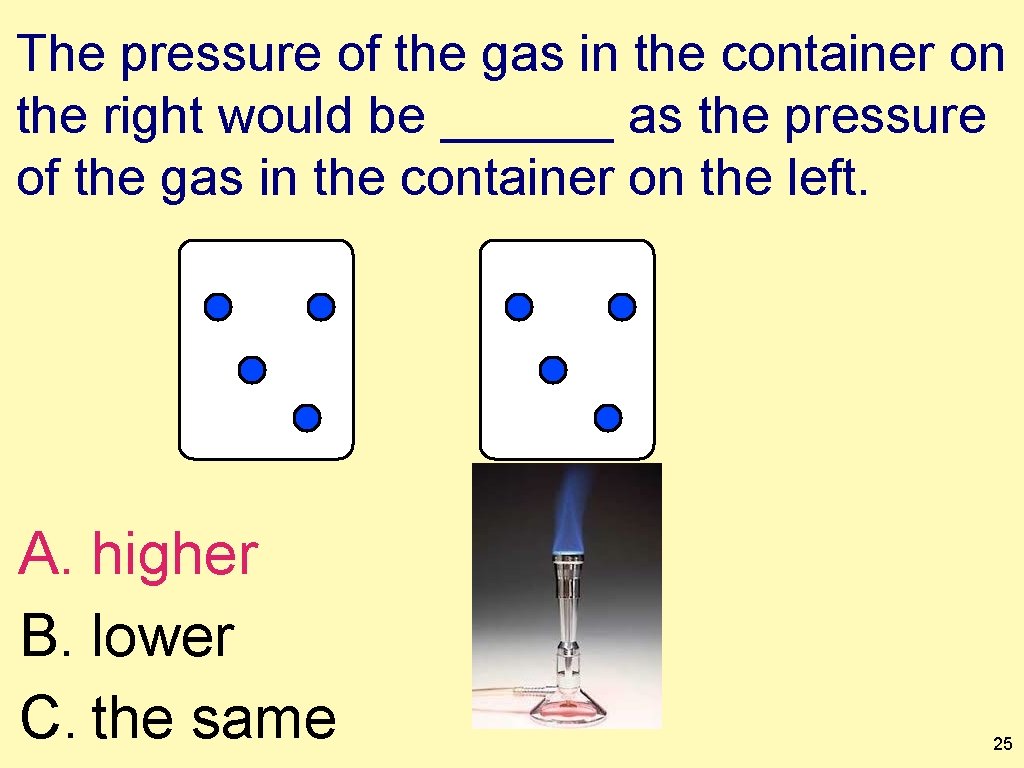
The pressure of the gas in the container on the right would be ______ as the pressure of the gas in the container on the left. A. higher B. lower C. the same 25

pressure and temperature • P&T • increase the temperature (while volume and moles remains constant) and the pressure increases. atm • this is an direct relationship • P is directly proportional to T • k = a constant 26

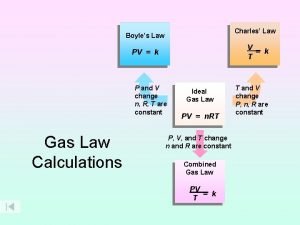
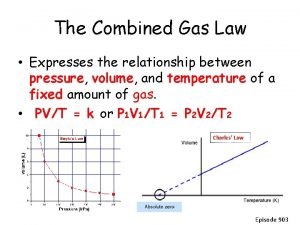
Gas Relationships Together • All the k’s are different V n = k P n = k V T = k P V = k P T = k • Squish the k’s all together, let’s call kcombined, R • and since R = R • The Combined Gas Law 27

The Combined Gas Law The “Before and After” Gas Law • Consider 0. 5 L of gas in sealed syringe in a 50ºC water bath with a pressure of 200 mm. Hg. The syringe is compressed to 0. 1 L held at 50ºC, what will be the new pressure? P 1 V 1 P 2 V 2 = n 1 T 1 n 2 T 2 • 2 L of oxygen gas in a sealed, rigid P 1 V 1 P 2 V 2 = metal cylinder is at 0. 80 atm at 25ºC. n 1 T 1 n 2 To what temperature, in Celsius should the gas be changed to reduce the pressure to 0. 20 atm? 28

The Combined Gas Law The “Before and After” Gas Law • Consider 0. 5 L of gas in sealed syringe in a 50ºC water bath with a pressure of 200 mm. Hg. The syringe is compressed to 0. 1 L held at 50ºC, what will be the new pressure? P 1 V 1 P 2 V 2 = n 1 T 1 n 2 T 2 • 2 L of oxygen gas in a sealed, rigid P 1 V 1 P 2 V 2 = metal cylinder is at 0. 80 atm at 25ºC. n 1 T 1 n 2 To what temperature, in Celsius should the gas be changed to reduce the pressure to 0. 20 atm? 29

pressure and temperature. . . again • Double the temp, does not double the pressure as you might expect. • The pressure only reaches 1. 2 atm, why? ≠ 2 atm = 1. 2 atm 1 atm 30
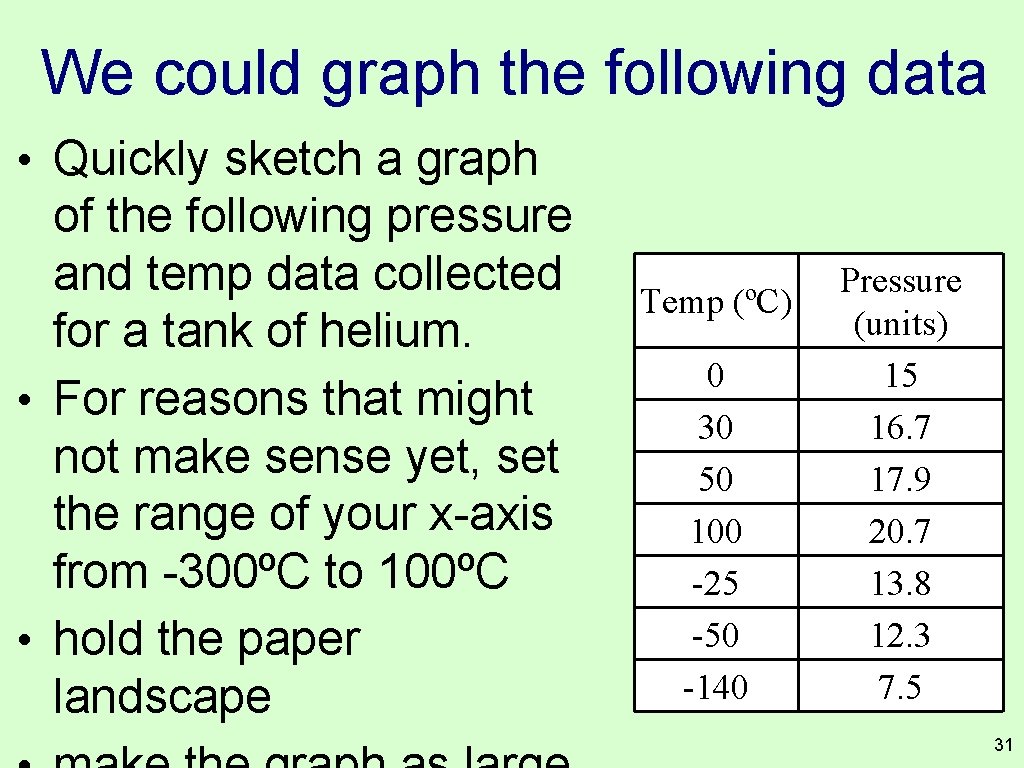
We could graph the following data • Quickly sketch a graph of the following pressure and temp data collected for a tank of helium. • For reasons that might not make sense yet, set the range of your x-axis from -300ºC to 100ºC • hold the paper landscape Temp (ºC) 0 30 50 100 -25 -50 -140 Pressure (units) 15 16. 7 17. 9 20. 7 13. 8 12. 3 7. 5 31

Cool Down. . Slow Down • As gases cool, they slow down and cause • • less pressure. . Slow molecules more = less pressure. . . slower still = even less pressure. . . finally the molecules stop, and don’t cause any pressure and can’t go any slower. this is as slow as the molecules can go, thus this is the coldest the molecules can get. 32
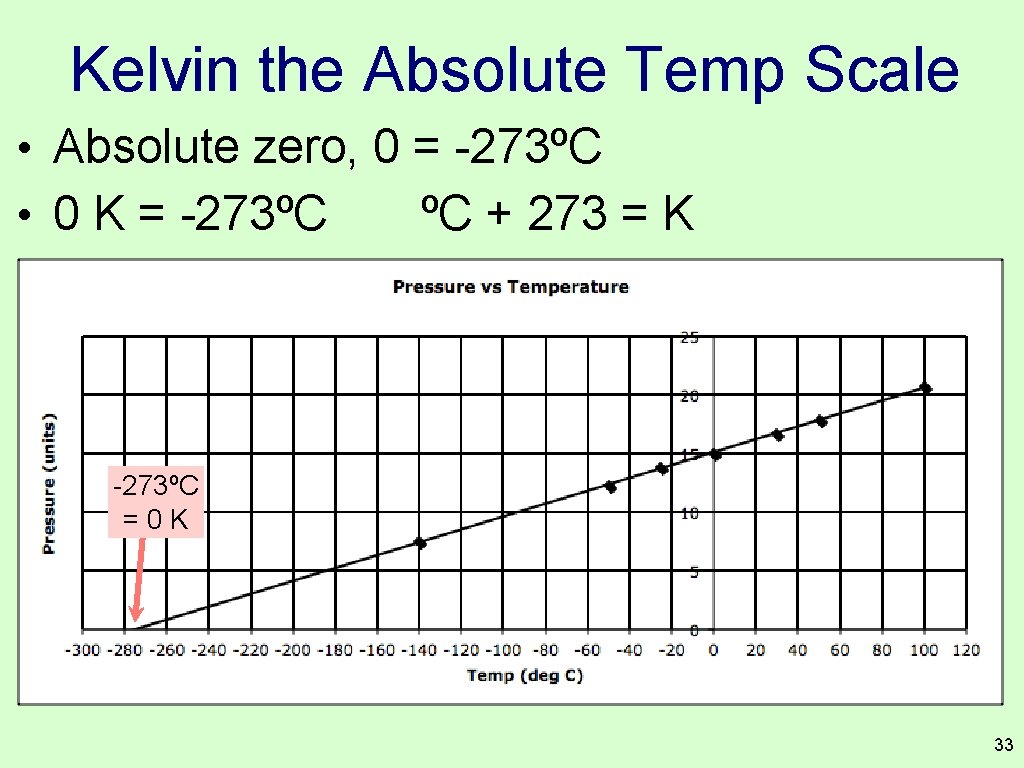
Kelvin the Absolute Temp Scale • Absolute zero, 0 = -273ºC • 0 K = -273ºC ºC + 273 = K -273ºC =0 K 33

pressure and temperature. . . again • Double the temp, does not double the pressure as you might expect. • 50ºC = 323 K, and to double the pressure we must double the Kelvin temp to 646 K = 373ºC 1 atm ≠ 2 atm 34
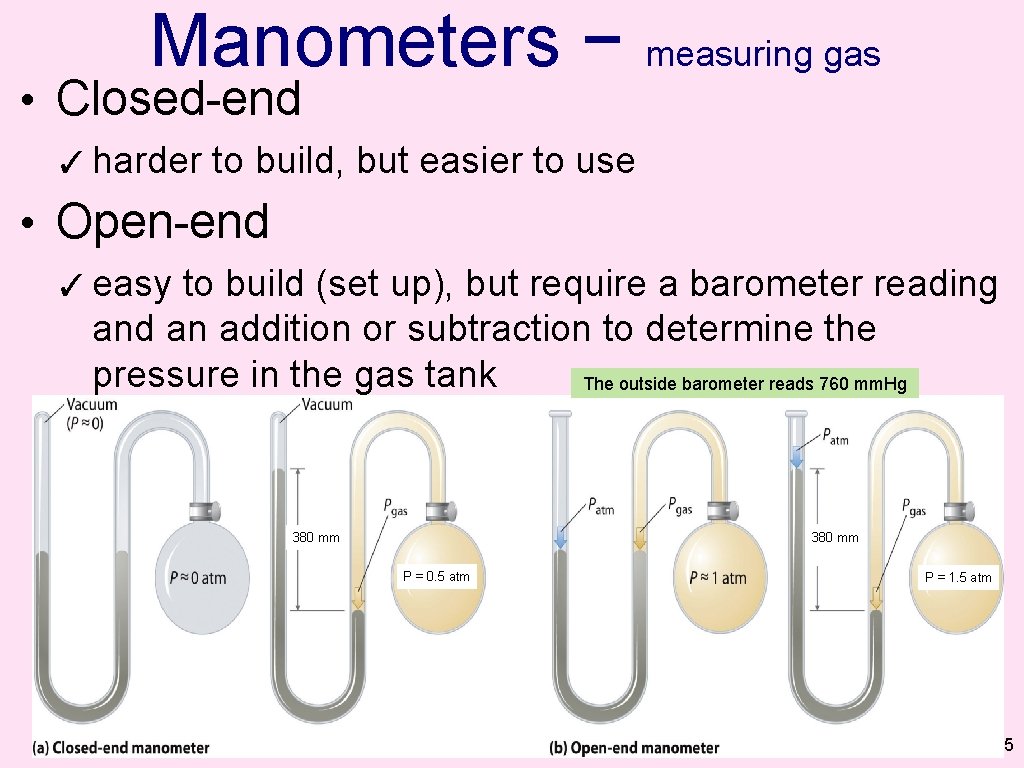
Manometers − measuring gas • Closed-end ✓ harder to build, but easier to use • Open-end ✓ easy to build (set up), but require a barometer reading and an addition or subtraction to determine the pressure in the gas tank The outside barometer reads 760 mm. Hg 380 mm P = 0. 5 atm P = 1. 5 atm 35
Pre LAD D. 1 Gas in a Tube 36

Predict: As you lift the eudiometer up in the cylinder, the vol of the gas in the eudiometer will measure 1. larger in 1 2. larger in 2 3. the same volume 1 ? 2 37

As you lift the eudiometer up in the cylinder, the vol of the gas in the eudiometer is larger in 1. The “weight” of the 1 2 volume of water above the gas in the eudiometer will “squish” the gas into a smaller volume. 2. Thus the volume will be larger in #2 38

As you lift the eudiometer up in the cylinder, the pressure of the gas in the eudiometer will ? 1. stay the same 2. get smaller 3. get larger 39

As you lift the eudiometer up in the cylinder, the pressure of the gas in the eudiometer will 1. 2. stay the same get smaller 3. Since we just learned that the Volume gets larger, the pressure must get smaller Or. . . consider the setup as a manometer, and the gas is “winning” (higher pressure than air) on the left, and the gas on the right is “losing” (lower pressure than air). 40

In the eudiometer pictured, the pressure of the air inside 1. is the same as the air pressure 2. is more than air pressure 3. is less than the air pressure 41

In the eudiometer pictured, the pressure of the air inside 1. is the same as the air pressure 2. is more than air pressure 3. is less than the air pressure 4. The pressure in the eudiometer is the same as the air pressure, however, two gases are causing the equal pressure - gas and water vapor, thus the gas must be less than the outside air pressure. 42

Exactly what is in the gas space of the eudiometer? 1. just the gas that I put in with the hose as demonstrated 2. the gas that I put in and some other gas as well 43

Dalton’s Law of Partial Pressures 44
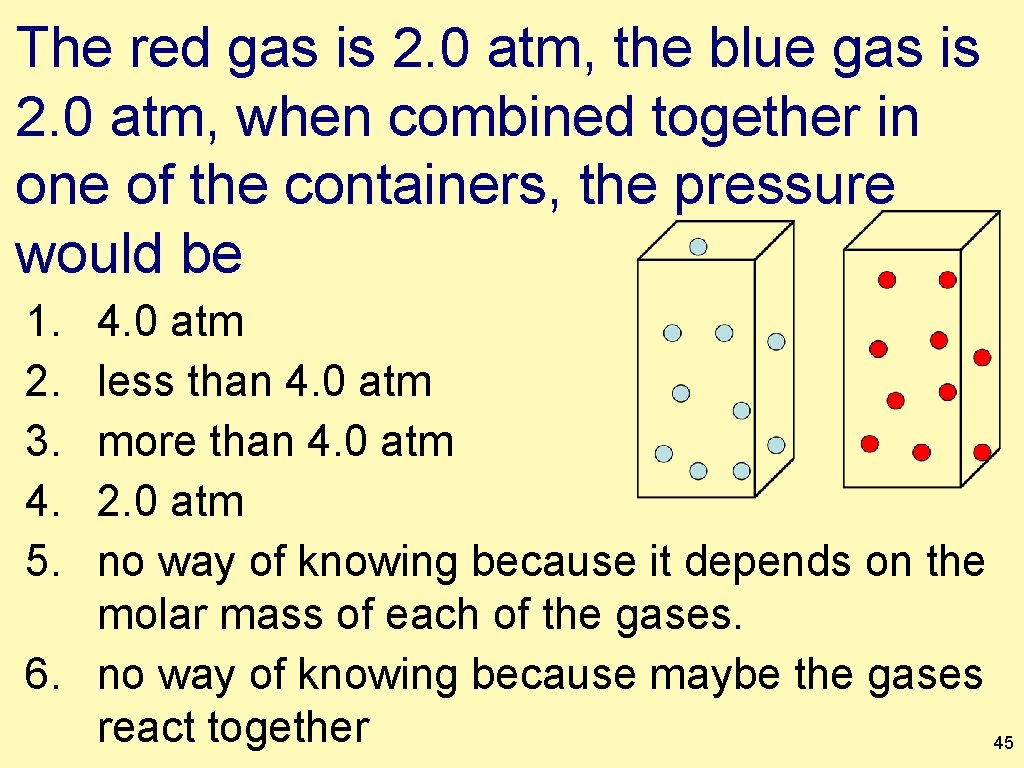
The red gas is 2. 0 atm, the blue gas is 2. 0 atm, when combined together in one of the containers, the pressure would be 1. 2. 3. 4. 5. 4. 0 atm less than 4. 0 atm more than 4. 0 atm 2. 0 atm no way of knowing because it depends on the molar mass of each of the gases. 6. no way of knowing because maybe the gases react together 45
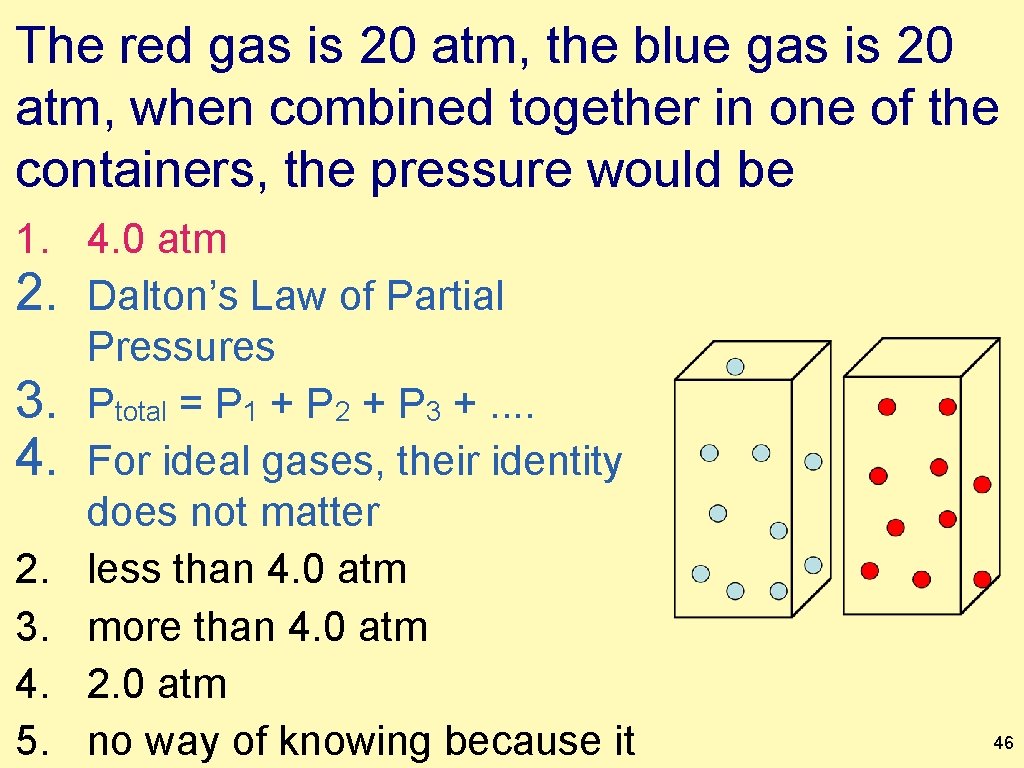
The red gas is 20 atm, the blue gas is 20 atm, when combined together in one of the containers, the pressure would be 1. 4. 0 atm 2. Dalton’s Law of Partial Pressures 3. Ptotal = P 1 + P 2 + P 3 +. . 4. For ideal gases, their identity does not matter 2. less than 4. 0 atm 3. more than 4. 0 atm 4. 2. 0 atm 5. no way of knowing because it 46

A rigid flask contains 0. 3 moles He, 0. 4 moles H 2, and 0. 5 moles Ne. The total pressure is 15 atm. What is the partial pressure of the H 2? (No calculator. ) 1. Input a numeric answer. 2. or work together and discuss with your nearby mates 47 47

A rigid flask contains 0. 3 moles He, 0. 4 moles H 2, and 0. 5 moles Ne. The total pressure is 15 atm. What is the partial pressure of the H 2? (No calculator. ) 1. the mole fraction: • (a percentage without multiplying by 100) 2. 3. Let’s look for this on the formula sheet…. 48
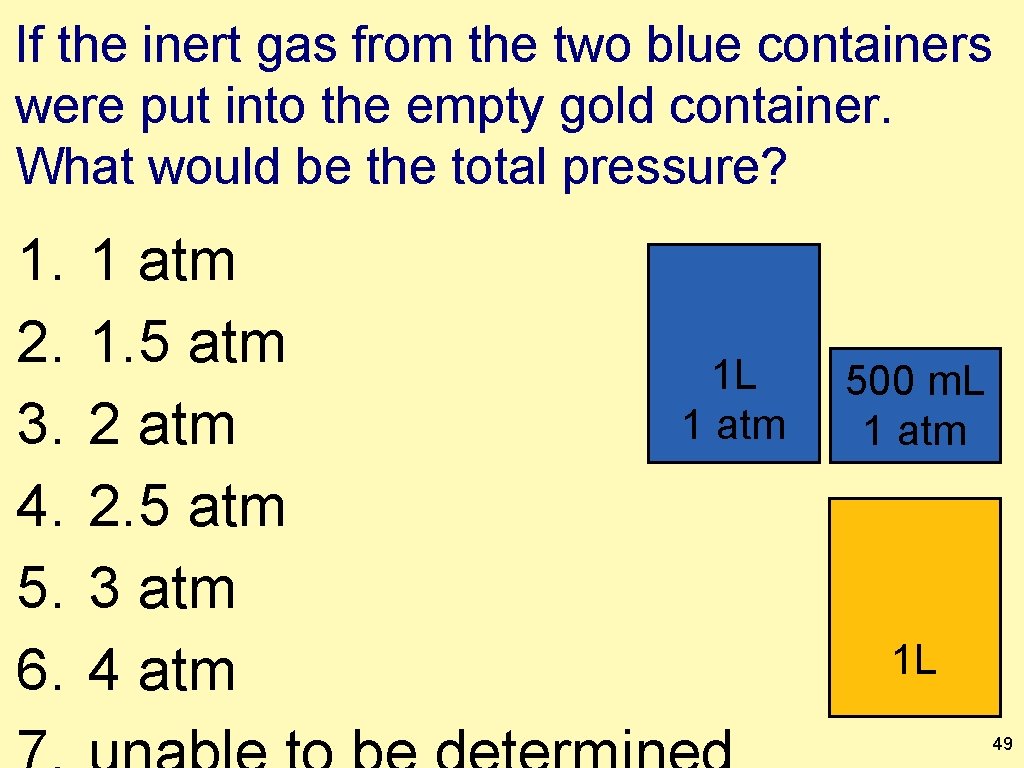
If the inert gas from the two blue containers were put into the empty gold container. What would be the total pressure? 1. 2. 3. 4. 5. 6. 1 atm 1. 5 atm 2. 5 atm 3 atm 4 atm 1 L 1 atm 500 m. L 1 atm 1 L 49
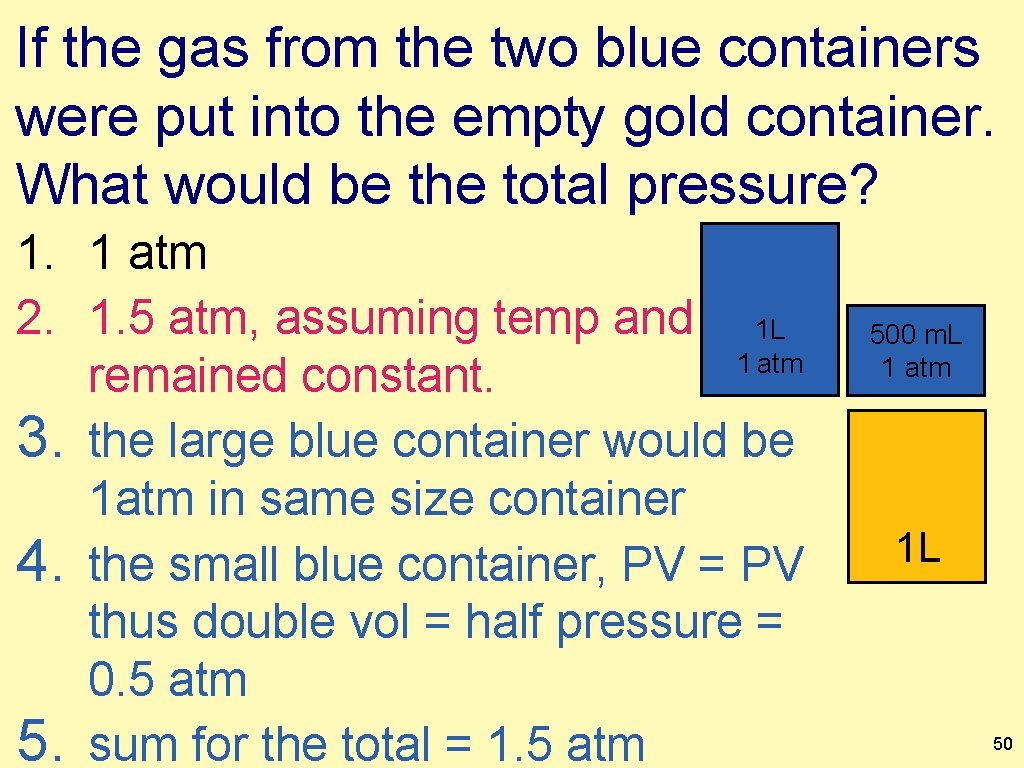
If the gas from the two blue containers were put into the empty gold container. What would be the total pressure? 1. 1 atm 2. 1. 5 atm, assuming temp and 1 L 1 atm remained constant. 3. the large blue container would be 1 atm in same size container 4. the small blue container, PV = PV thus double vol = half pressure = 0. 5 atm 5. sum for the total = 1. 5 atm 500 m. L 1 atm 1 L 50
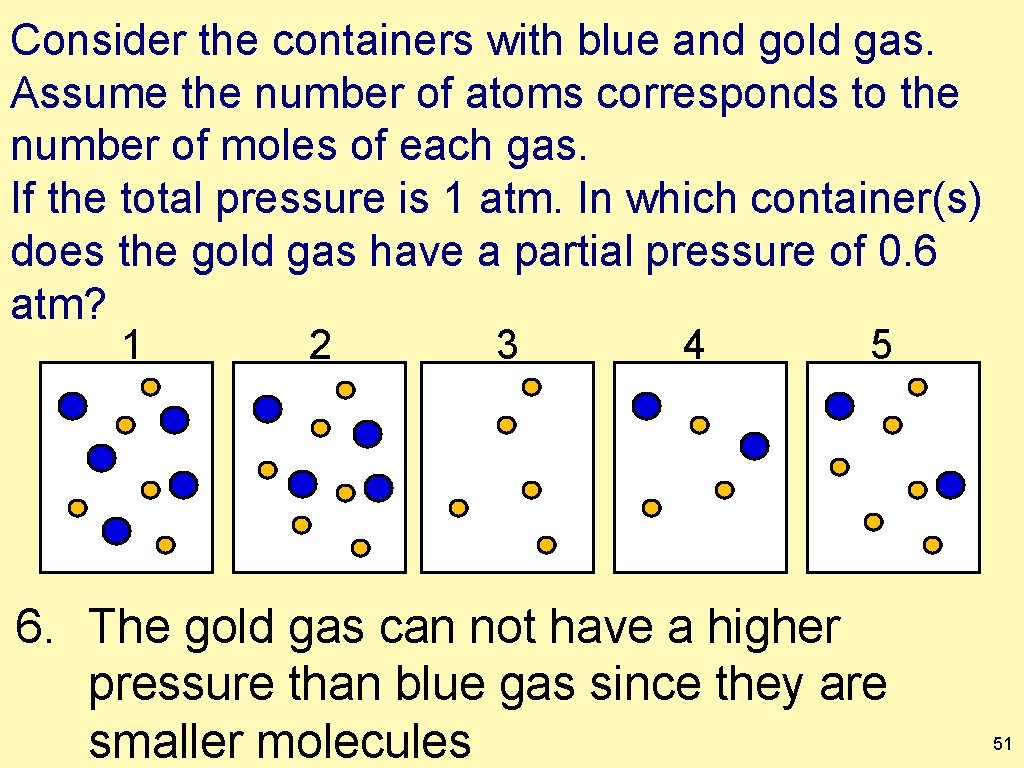
Consider the containers with blue and gold gas. Assume the number of atoms corresponds to the number of moles of each gas. If the total pressure is 1 atm. In which container(s) does the gold gas have a partial pressure of 0. 6 atm? 1 2 3 4 5 6. The gold gas can not have a higher pressure than blue gas since they are smaller molecules 51
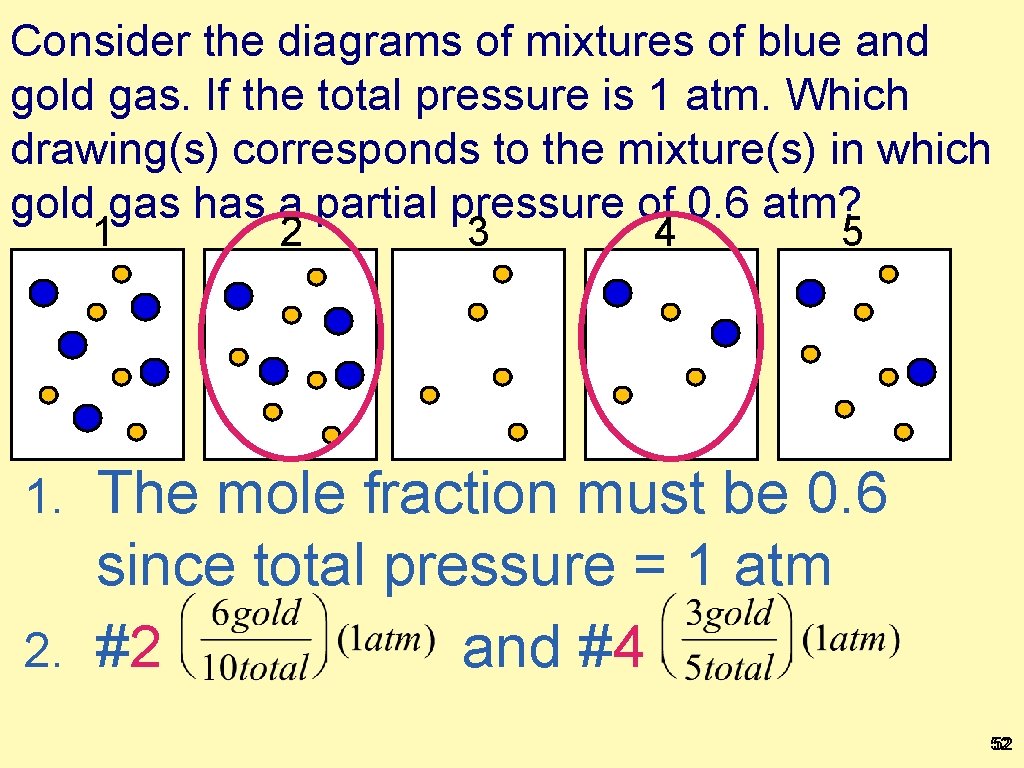
Consider the diagrams of mixtures of blue and gold gas. If the total pressure is 1 atm. Which drawing(s) corresponds to the mixture(s) in which gold gas has a partial pressure of 0. 6 atm? 1 2 3 4 5 The mole fraction must be 0. 6 since total pressure = 1 atm 2. #2 and #4 1. 52 52

Equal masses of three gases were placed in a rigid, sealed 3 L container. The total pressure is 2 atm. Which gas has the highest partial pressure? 1. 2. 3. 4. He Ne Ar All three gases cause the same partial pressure because they all have the same mass. 53

Equal masses of three gases were placed in a rigid, sealed 3 L container. The total pressure is 2 atm. Which gas has the highest partial pressure? 1. He 2. The gas with the lower molar mass will be more moles and thus cause greater pressure. 3. It is a valid assumption that the pressure is low enough and temperature is high enough to allow the gases to behave ideally. 2. Ne 3. Ar 4. All three gases cause the same partial pressure. 54

Equilibrium Vapor Pressure 55

H 2 O as a gas 1. All liquids evaporate. 2. Evaporated liquid causes pressure like any other gas. 3. If the flask is left open the water would all eventually evaporate? 1. Yes 2. No 56

H 2 O as a gas 1. All liquids evaporate. 2. Evaporated liquid causes pressure like any other gas. 3. If the flask is left open the water would all eventually evaporate? 1. Yes ‣ Evaporation is a surface phenomenon. It may take a long time since there is such a small opening in the flask. 57

If the flask is sealed, will all the water evaporate? 1. Yes 2. No 3. Maybe 58

If the flask is sealed, will all the water evaporate? 1. Yes 2. Not likely 3. The image shows a larger amount of water than completely evaporate into the space, thus it is likely to reach equilibrium. The amount of liquid evaporating will be equal to the amount of vapor condensing. 3. Maybe 59

If there is enough liquid water in the flask, equilibrium will result. The resulting pressure in the flask is called the equilibrium vapor pressure. 60

Which flask will achieve a higher equilibrium vapor pressure? 1. Flask A 2. Flask B 3. The same, since it appears that the flasks are the same size and the amount of liquid water appears to be the same. Flask B Flask A Hot 61

Which flask will achieve a higher equilibrium vapor pressure? 1. Flask A 2. Flask B Higher temp cause greater equilibrium vapor pressure. ‣ 3. The same, since it appears that the flasks are the same size and the amount of liquid water appears to be the same. Flask A Hot 62
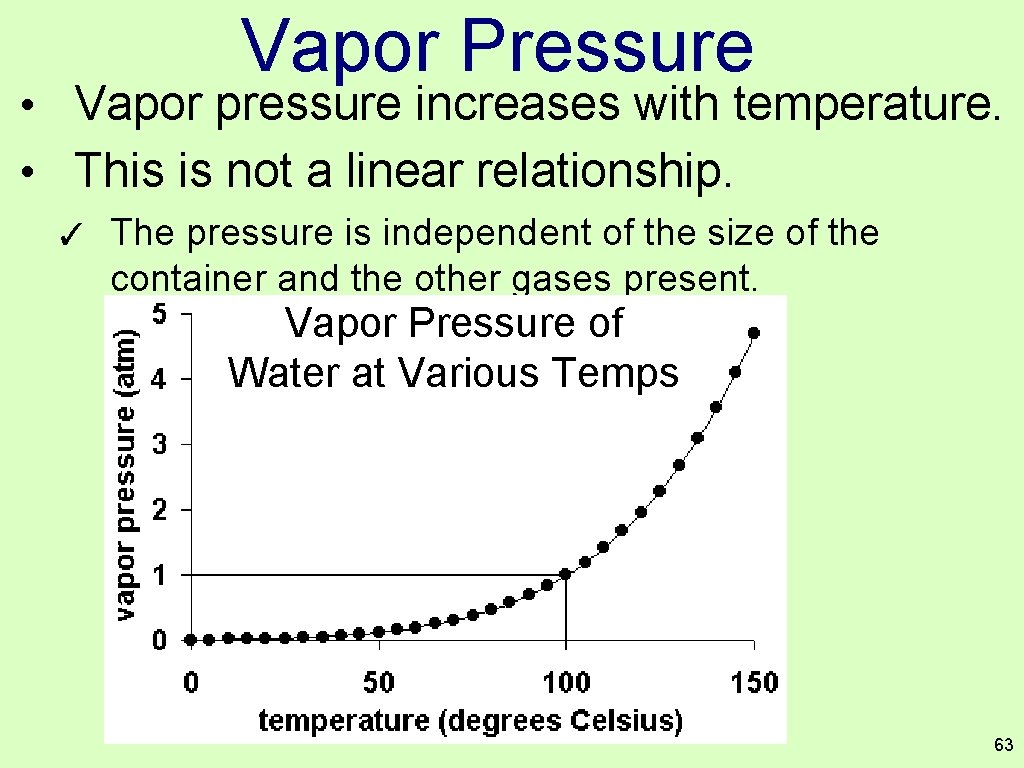
Vapor Pressure • Vapor pressure increases with temperature. • This is not a linear relationship. ✓ The pressure is independent of the size of the container and the other gases present. Vapor Pressure of Water at Various Temps 63
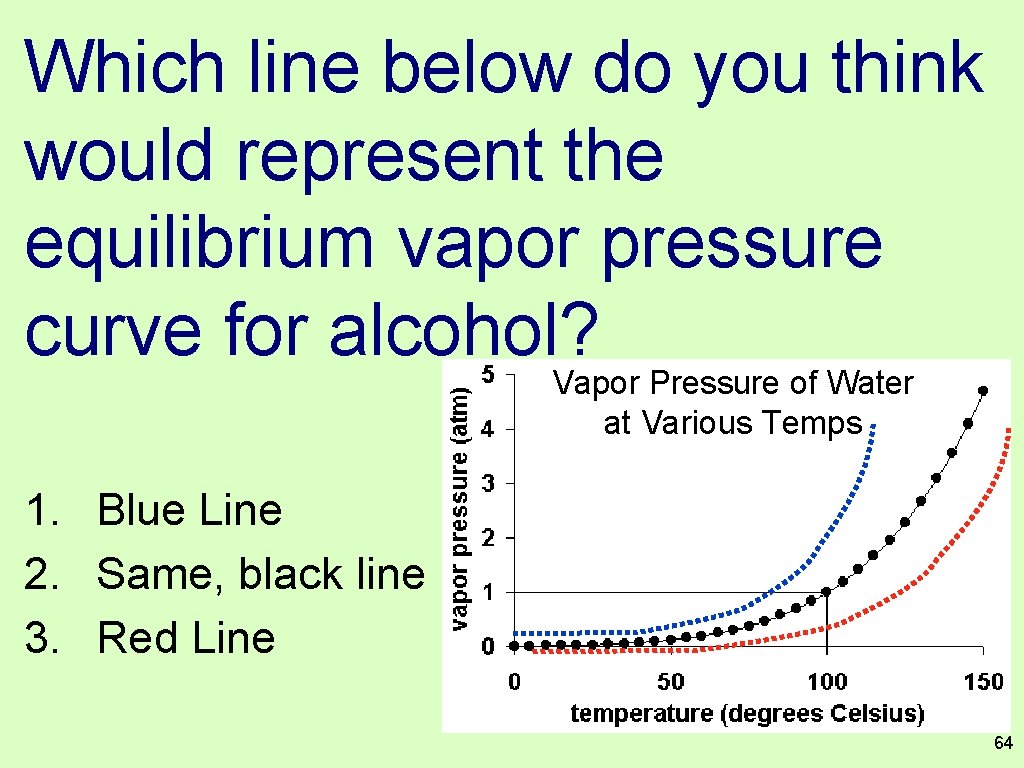
Which line below do you think would represent the equilibrium vapor pressure curve for alcohol? Vapor Pressure of Water at Various Temps 1. Blue Line 2. Same, black line 3. Red Line 64
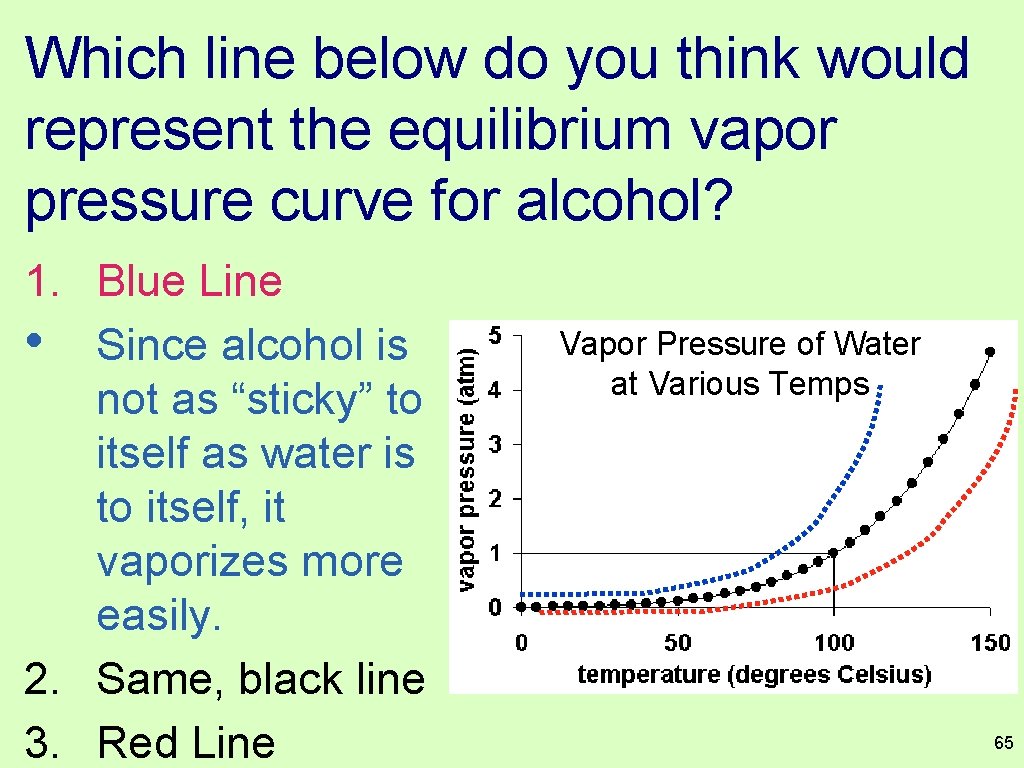
Which line below do you think would represent the equilibrium vapor pressure curve for alcohol? 1. Blue Line • Since alcohol is not as “sticky” to itself as water is to itself, it vaporizes more easily. 2. Same, black line 3. Red Line Vapor Pressure of Water at Various Temps 65
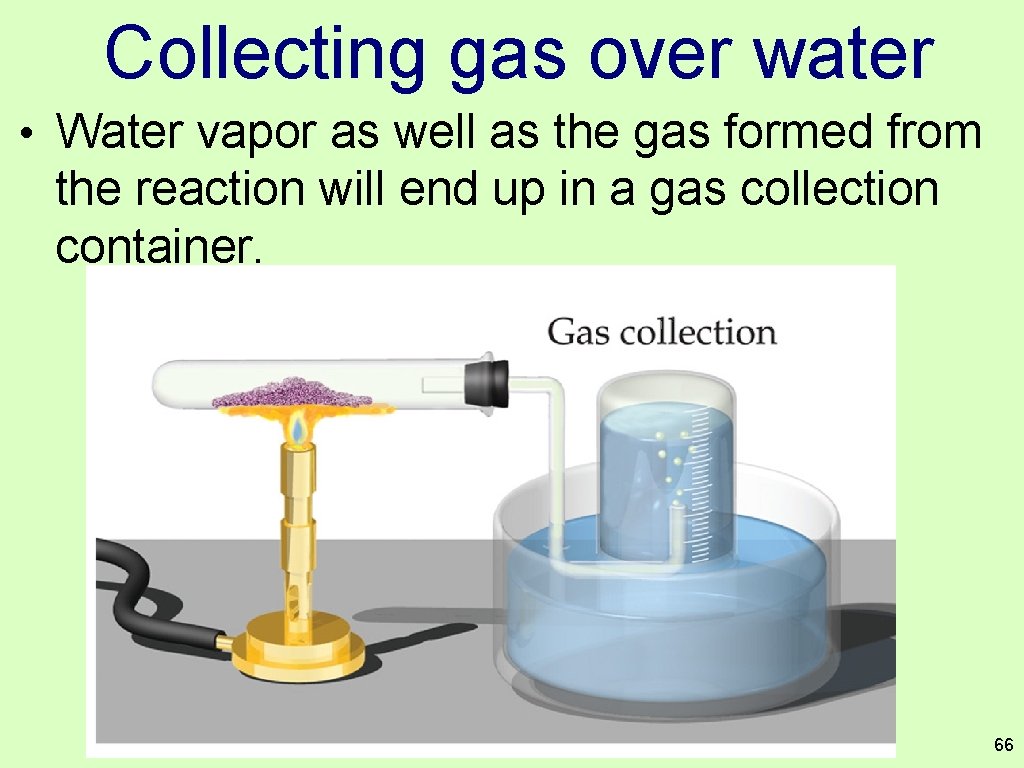
Collecting gas over water • Water vapor as well as the gas formed from the reaction will end up in a gas collection container. 66

Developing the Ideal Gas Law 67

Put some gas in a container at certain conditions. . . air pressure • It just so happens that ✓ 32 g of oxygen, (1 mole) ✓ sealed in a piston cylinder, ✓ plunged into ice water, ✓ barometric air pressure of 760, 22. 4 L • The measured volume of the gas will = 22. 4 L 68
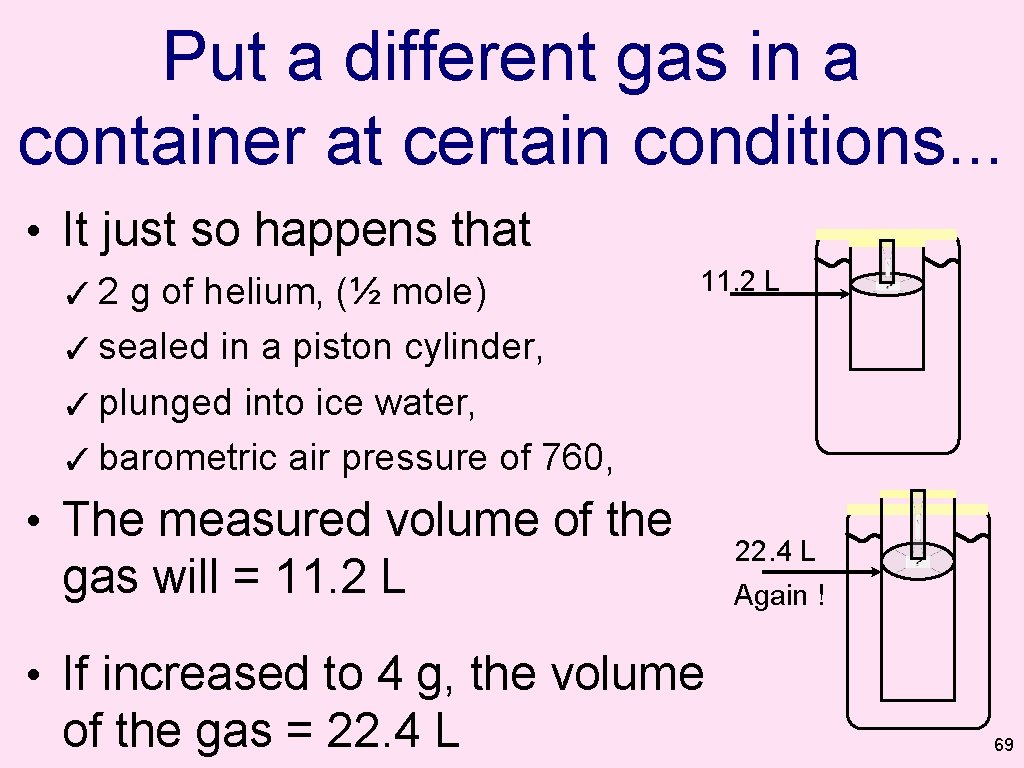
Put a different gas in a container at certain conditions. . . • It just so happens that ✓ 2 g of helium, (½ mole) ✓ sealed in a piston cylinder, ✓ plunged into ice water, ✓ barometric air pressure of 760, 11. 2 L • The measured volume of the gas will = 11. 2 L 22. 4 L Again ! • If increased to 4 g, the volume of the gas = 22. 4 L 69

Just what about gas constant, R? • It just so happens that ✓ 44 g of CO 2, (1 mole) ✓ sealed in a piston cylinder, ✓ plunged into ice water, ✓ barometric air pressure of 760, • The measured volume of the gas will = 22. 4 L, yet again! • Plug any set of these measures values into the Gas Law, and solve for R 70

Just what about gas constant, R? • The units on the constant R are very important. • If we plug the values into the gas law measuring in different units, R will be different. Let’s find R on the Formula Sheet 71

LAD D. 2 Correcting to STP Conditions 72

“Correcting to STP” 42. 0 ml of hydrogen gas was collected at 21. 0ºC over water. The barometer read 757. 5 mm. Hg. Calculate the volume that this hydrogen would occupy at STP? Equilibrium Vapor Pressure 73

“Correcting to STP” • 42. 0 ml of hydrogen gas was collected at 21. 0ºC over water. The barometer read 757. 5 mm. Hg. Calculate the volume that this hydrogen would occupy at STP? Remember, we must first Equilibrium Vapor Pressure determine the gas pressure by compensating for vapor pressure. Then correct the measured conditions to STP conditions. 74

Ideally, Chemistry’s a Gas! Using the Ideal Gas Law 75

The Ideal Gas Law • From data we previously considered: • 1 atm and 0ºC are considered standard conditions (STP). • What is the volume of 1 mole of gas? ✓Depends on the conditions…. • 22. 4 L is the volume of 1 76

Using the Ideal Gas Law • If you have 1500 ml of oxygen gas at a pressure of 624 mm. Hg and at 27. 0ºC, calculate the moles of gas in this container. • How do I know what R value to use? ? ? ✓Look at your units, the units must match. 77

Using the Ideal Gas Law • If you have 1500 ml of oxygen gas at a pressure of 624 mm. Hg and at 27. 0ºC, calculate the moles of gas in this container. • What other questions might someone ask? 78

The Ideal Gas Law • If you have 1500 ml of oxygen gas at a pressure of 624 mm. Hg and at 27. 0ºC, calculate the moles of gas in this container. • What is the mass of the gas? • What is the density of the gas? • What volume of hydrogen at the given conditions would you need to react completely with this oxygen? • What mass of water would be produced? 79

The Ideal Gas Law • If you have 1500 ml of oxygen gas at a pressure of 624 mm. Hg and at 27. 0ºC, calculate the moles of gas in this container. • What is the mass? 80

The Ideal Gas Law • If you have 1500 ml of oxygen gas at a pressure of 624 mm. Hg and at 27. 0ºC, calculate the moles of gas in this container. • What is the mass? • What is the density? 81

The Ideal Gas Law • If you have 1500 ml of oxygen gas at a pressure of 624 mm. Hg and at 27. 0ºC, calculate the moles of gas in this container. • What volume of hydrogen (assume same P, T would you need to react completely with this oxygen? whcih allos us to do stoichiometry with volumes) ✓ 2 H 2 + O 2 → 2 H 2 O • What mass of water would be produced? 82
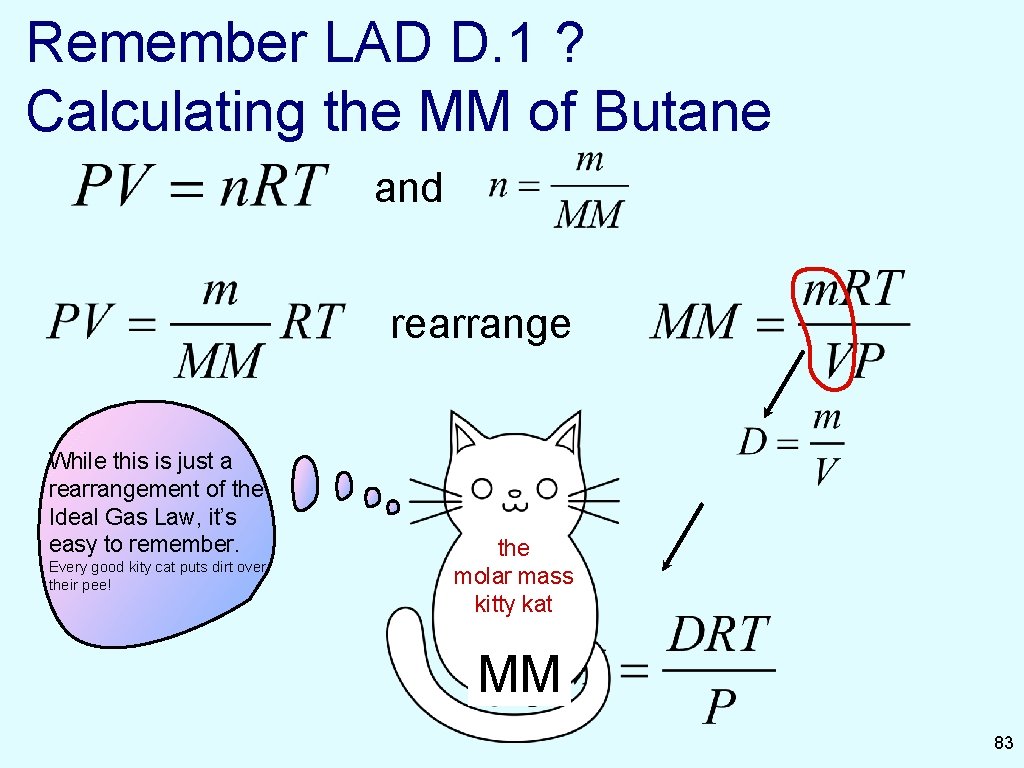
Remember LAD D. 1 ? Calculating the MM of Butane and rearrange While this is just a rearrangement of the Ideal Gas Law, it’s easy to remember. Every good kity cat puts dirt over their pee! the molar mass kitty kat MM 83

Let’s do some algebra. • Determine the molar mass of a Noble gas that has been heated to 300ºC and has a pressure of 25 atm with a density of 21. 3 g/L. 84

Let’s do some algebra. • Determine the molar mass of a Noble gas that has been heated to 300ºC and has a pressure of 25 atm with a density of 21. 3 g/L. 85

Calculate the density of nitrogen gas at STP. 86

Which Equation to Use? • What would be the identity of 0. 896 g of some diatomic gas in a 3. 0 L container that was at 58. 5 mm. Hg of pressure and had a temp of − 50. 0ºC 87

Which Equation to Use? • If you have a 5 L container of air at STP, what is the (partial) pressure of oxygen? • 88

Which Equation to Use? • If you have a 5 L container that is 20% (by mass) hydrogen and 80% nitrogen (by mass) at STP, what is the (partial) pressure of hydrogen? • 89

Which Equation to Use? • O 2 gas in a freely moveable piston container has a volume of 750 ml at 15ºC. If the container is heated to 30ºC what will be the new volume of the gas? 90

When calcium carbonate is heated strongly, carbon dioxide gas is released Ca. CO 3(s) → Ca. O(s) + CO 2(g) What volume of CO 2(g), measured at STP, will be produced if 15. 2 g of Ca. CO 3(s) is heated? 91

Consider the following unbalanced chemical equation for the combustion of propane. C 3 H 8(g) + O 2(g) → CO 2(g) + H 2 O What volume of oxygen gas at 25ºC and 745 atm is needed for the complete combustion of 1. 54 g of propane? 92

Ammonia and gaseous hydrogen chloride combine to form ammonia chloride. NH 3(g) + HCl(g) → NH 4 Cl(s) If 12. 0 L of NH 3(g) at 20. 0ºC and 0. 945 atm is combined with 15. 0 L of HCl(g) at 26ºC and 1. 04 atm, what mass of NH 4 Cl(s) will be produced? Which gas is the limiting reactant? Which gas is present in excess? 93

If water is added to magnesium nitride, ammonia gas is produced when the mixture is heated. Mg 3 N 2(g) + 3 H 2 O(L) → 3 Mg. O(s) + 2 NH 3(g) If 6. 50 g of magnesium nitride is treated with water, what volume of ammonia gas would be collected at 22. 0ºC and 755 mm Hg? 94

Clicker Questions 95

A sealed container is manipulated such that the absolute temp is doubled, and the pressure is tripled. How will the number of moles of molecules be affected? (Do not use a calculator. ) 1. increase 2. decrease 3. stay the same 96 96

A sealed container is manipulated such that the absolute temp is doubled, and the pressure is tripled. How will the number of moles of molecules be affected? (Do not use a calculator. ) 1. increase 2. decrease 3. stay the same 4. The problem tells you that the container is sealed, thus heat and volume changes must be what is used to change the temp and pressure 97 97

A hydrocarbon with an empirical formula of CH 2 has a density of 2. 5 g/L at STP. A possible formula of the hydrocarbon is 1. 2. 3. 4. 5. CH 2 C 2 H 4 C 3 H 6 C 4 H 8 C 5 H 10 98

A hydrocarbon with an empirical formula of CH 2 has a density of 2. 5 g/L at STP. A possible formula of the hydrocarbon is 1. 2. 3. 4. 5. CH 2 C 2 H 4 C 3 H 6 C 4 H 8 C 5 H 10 99

Select the gas with the greatest density at STP. 1. H 2 2. He 3. H 2 O 4. CO 2 5. O 2 6. They all have the same density, since the pressure and temperature conditions are the same. 100

Select the gas with the greatest density at STP. 1. 2. 3. 4. 5. 6. H 2 thus the gas with the larger molar mass will have the larger density when He the conditions are the same for each H 2 O gas. CO 2 They all have the same density, since the conditions are the same. 101
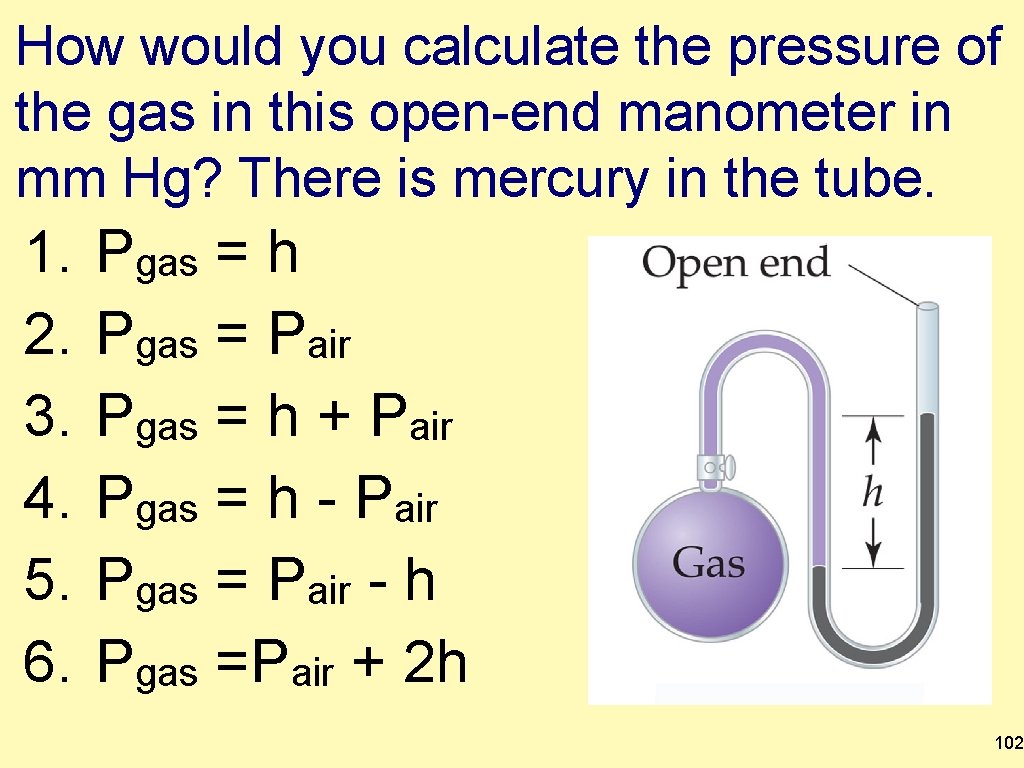
How would you calculate the pressure of the gas in this open-end manometer in mm Hg? There is mercury in the tube. 1. 2. 3. 4. 5. 6. Pgas = h Pgas = Pair Pgas = h + Pair Pgas = h - Pair Pgas = Pair - h Pgas =Pair + 2 h 102
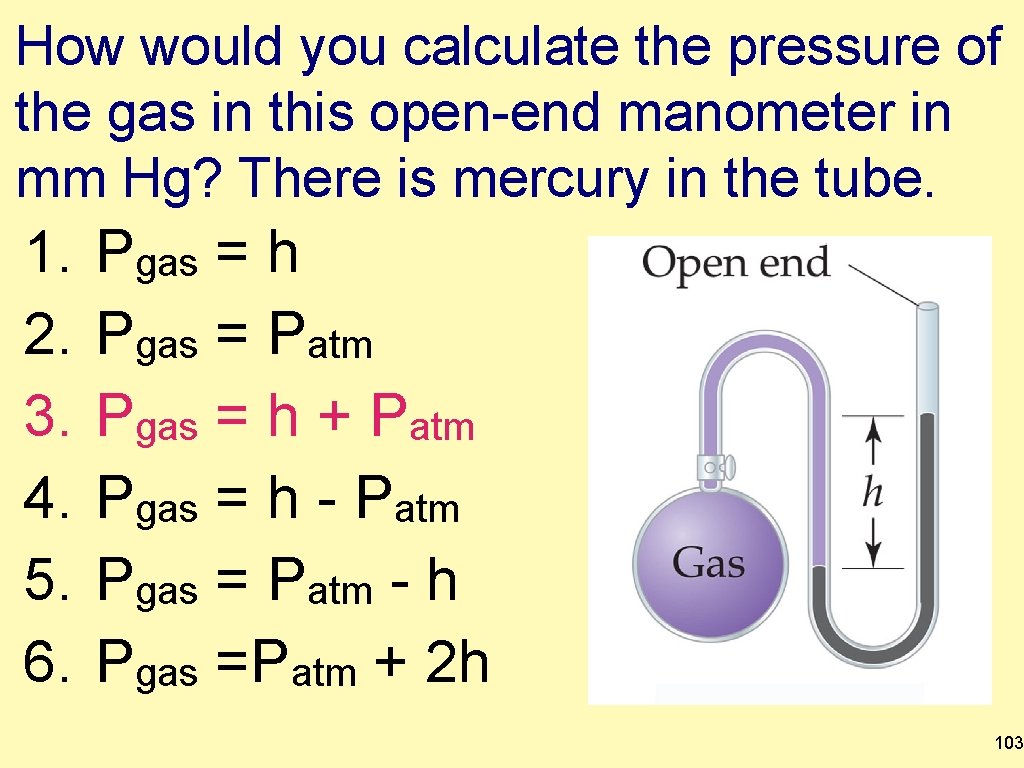
How would you calculate the pressure of the gas in this open-end manometer in mm Hg? There is mercury in the tube. 1. 2. 3. 4. 5. 6. Pgas = h Pgas = Patm Pgas = h + Patm Pgas = h - Patm Pgas = Patm - h Pgas =Patm + 2 h 103
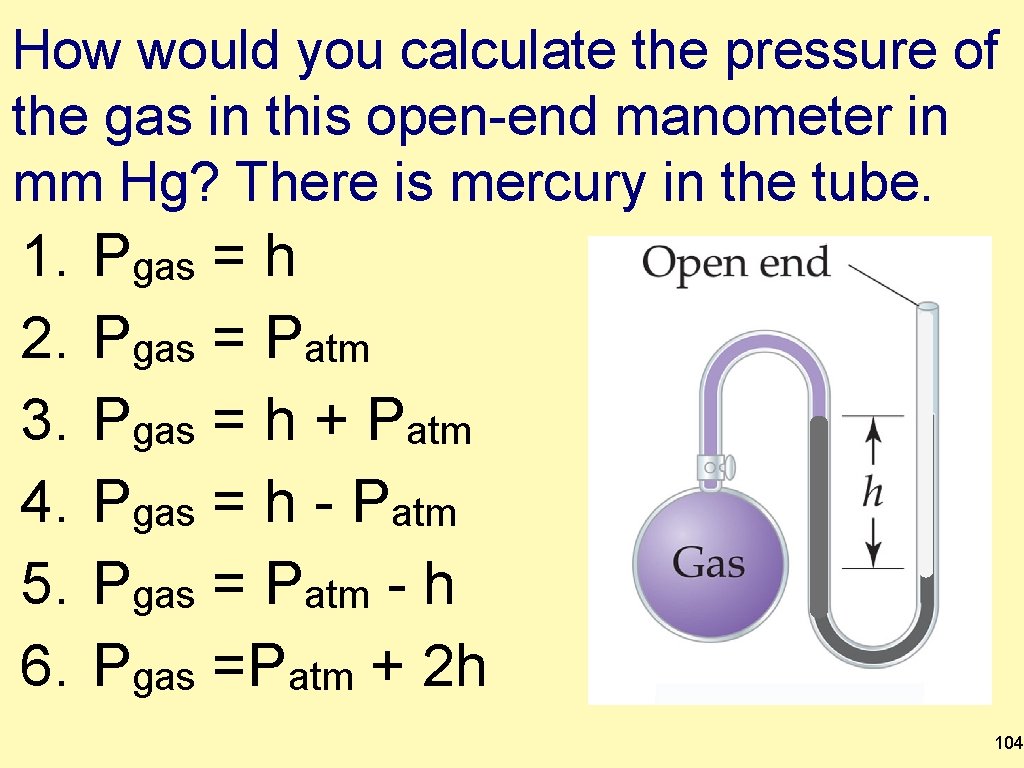
How would you calculate the pressure of the gas in this open-end manometer in mm Hg? There is mercury in the tube. 1. 2. 3. 4. 5. 6. Pgas = h Pgas = Patm Pgas = h + Patm Pgas = h - Patm Pgas = Patm - h Pgas =Patm + 2 h 104
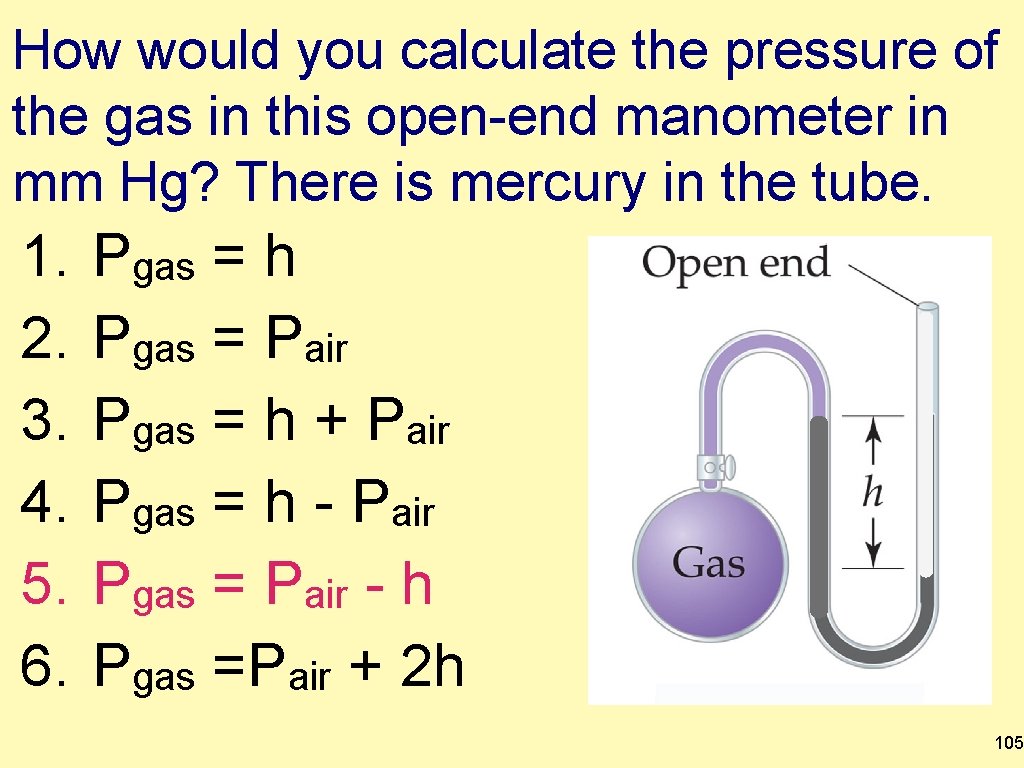
How would you calculate the pressure of the gas in this open-end manometer in mm Hg? There is mercury in the tube. 1. 2. 3. 4. 5. 6. Pgas = h Pgas = Pair Pgas = h + Pair Pgas = h - Pair Pgas = Pair - h Pgas =Pair + 2 h 105
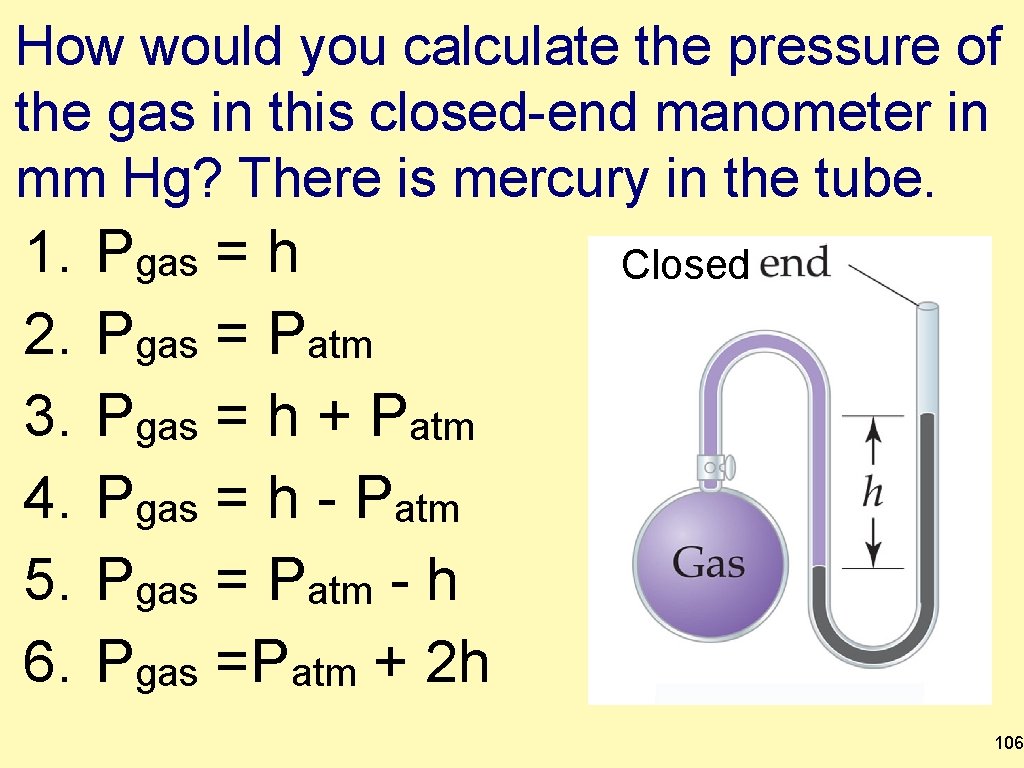
How would you calculate the pressure of the gas in this closed-end manometer in mm Hg? There is mercury in the tube. 1. 2. 3. 4. 5. 6. Pgas = h Pgas = Patm Pgas = h + Patm Pgas = h - Patm Pgas = Patm - h Pgas =Patm + 2 h Closed 106
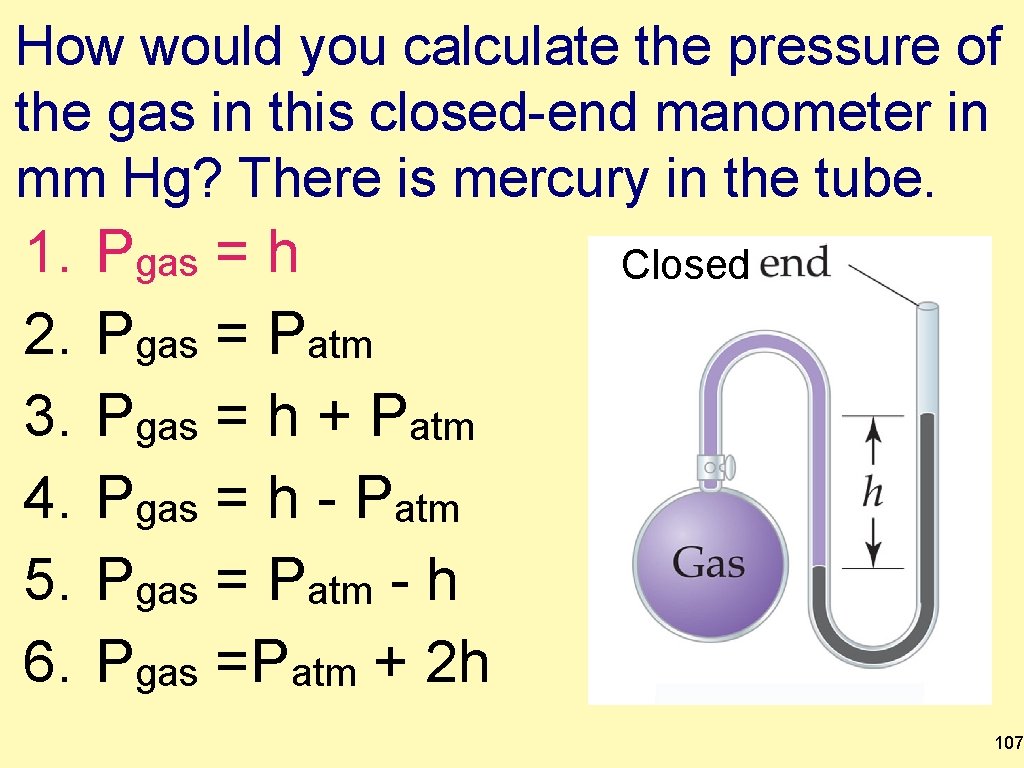
How would you calculate the pressure of the gas in this closed-end manometer in mm Hg? There is mercury in the tube. 1. 2. 3. 4. 5. 6. Pgas = h Pgas = Patm Pgas = h + Patm Pgas = h - Patm Pgas = Patm - h Pgas =Patm + 2 h Closed 107

The temp of a container of He is held constant, and the volume is doubled, thus the pressure changes. If the gas were carbon dioxide instead of helium, the change in pressure would be 1. the same as the He ∆ 2. less than the He ∆ 3. more than the He ∆ 108

The temp of a container of He is held constant, and the volume is doubled, thus the pressure changes. If the gas were carbon dioxide instead of helium, the change in pressure would be 1. the same as the He∆ 2. Avogadro’s Law • At a fixed temperature and pressure, equal volumes of any ideal gas have the same number of moles. • Thus changing volume will result in the same pressure changes regardless of the gas, since it will be same number of molecules. 109

The volume of the container is reduced to half it’s size, but the pressure remains constant. What can be true about the gas inside. Select all that apply. 1. The gas must now weigh half as much. 2. The temp was reduced from 100 to 50. 3. The gas molecules were switched to a gas with half the molar mass. 4. Half of the molecules may have leaked out. 5. No specifics are possible without knowing the identity of the gas. 110

The volume of the container is reduced to half it’s size, but the pressure remains constant. What can be true about the gas inside. Select all that apply. 1. The gas must now weigh half as much. 2. The temp was reduced from 100 to 50 If assumed to be Kelvin 3. The gas molecules were switched to a gas with half the molar mass. 4. Half of the molecules may have leaked out 5. No specifics are possible without knowing the identity of the gas. 111

The y-axis could be labeled 1. 2. 3. 4. pressure ? temperature moles any of the above Volume 112

The y-axis could be labeled 1. pressure 2. inverse relationship ? 2. temperature 3. moles 4. any of the above Volume 113
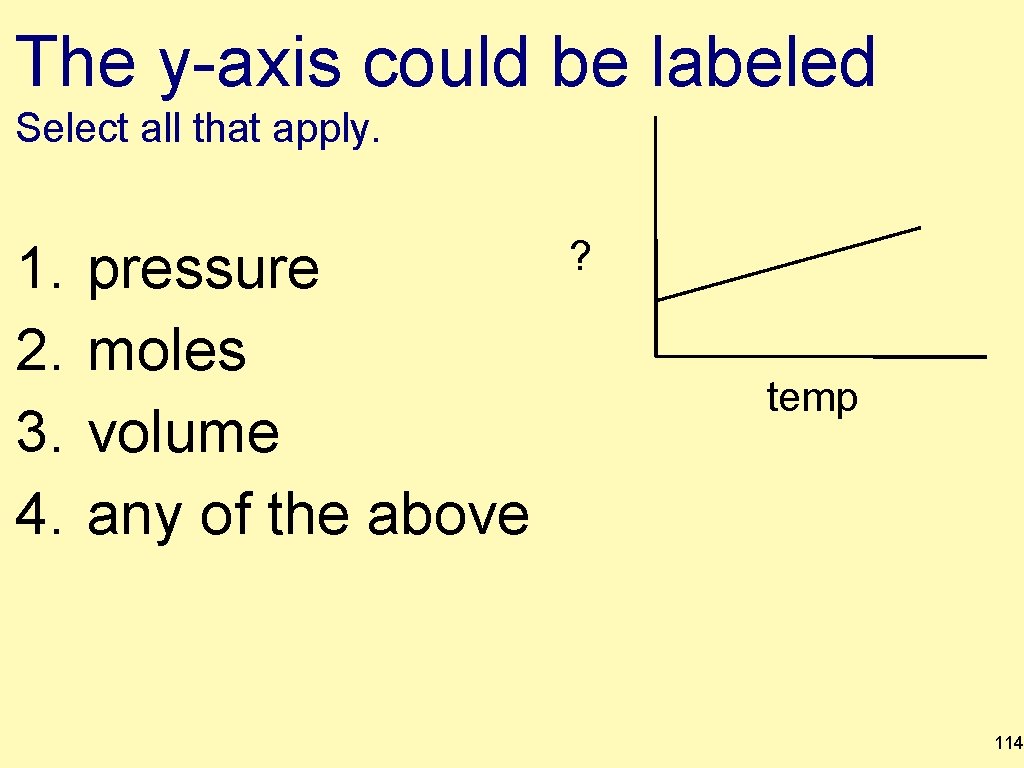
The y-axis could be labeled Select all that apply. 1. 2. 3. 4. pressure moles volume any of the above ? temp 114
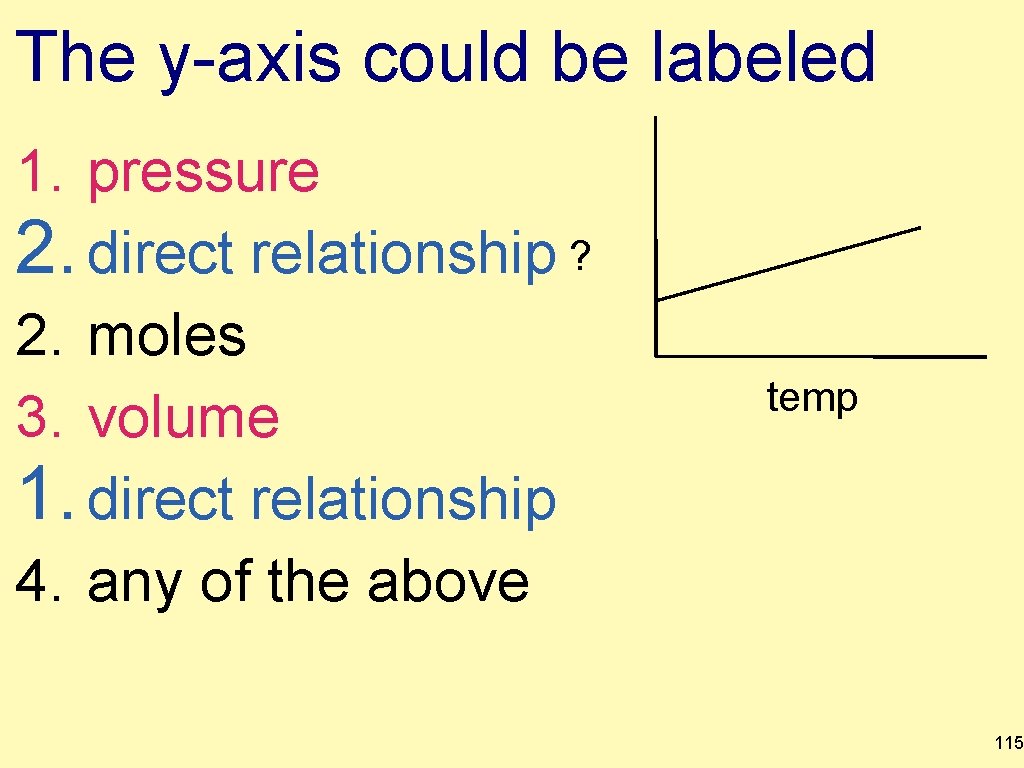
The y-axis could be labeled 1. pressure 2. direct relationship ? 2. moles 3. volume 1. direct relationship 4. any of the above temp 115

What is the density of CO 2 in a 4 L container at 1 atm and 273 K closest to? No calculator 1. 2. 3. 4. 5. 6. 88 g/L 44 g/L 22 g/L 11 g/L 5 g/L 2 g/L 116

What is the density of CO 2 in a 4 L container at 1 atm and 273 K closest to? No calculator 1. 2. 3. 4. 5. 6. 88 g/L 44 g/L 22 g/L 11 g/L 5 g/L 2 g/L 117

When nitrogen gas and hydrogen gas react, ammonia gas results. Write a balanced equation for this reaction. Is this picture stoichiometrically correct? During the course of the reaction, 1. 2. 3. 4. the pressure increases. the pressure decreases. the pressure stays the same. can not be determined 118

When nitrogen gas and hydrogen gas react, ammonia gas results. Write a balanced equation for this reaction. Is this picture stoichiometrically correct? During the course of the reaction, 1. 2. 3. the pressure increases. the pressure decreases. the pressure stays the same. 119

Problem Types Gas Laws 120

An excess of Al(s) is added to 100. m. L of 0. 400 M HCl. At 0˚C and 1 atm pressure, what volume of H 2 gas can be obtained? Calculators, yes 121

An excess of Al(s) is added to 100. m. L of 0. 400 M HCl. At 0˚C and 1 atm pressure, what volume of H 2 gas can be obtained? 1. 2 Al + 6 HCl → 2 Al. Cl 3 + 3 H 2 Woo Hoo STP 122

A 4. 0 L container that holds 3. 0 g of gas at STP will contain which gas? 1. 2. 3. 4. 5. O 2 NH 3 C 2 H 6 CO 2 SO 2 123
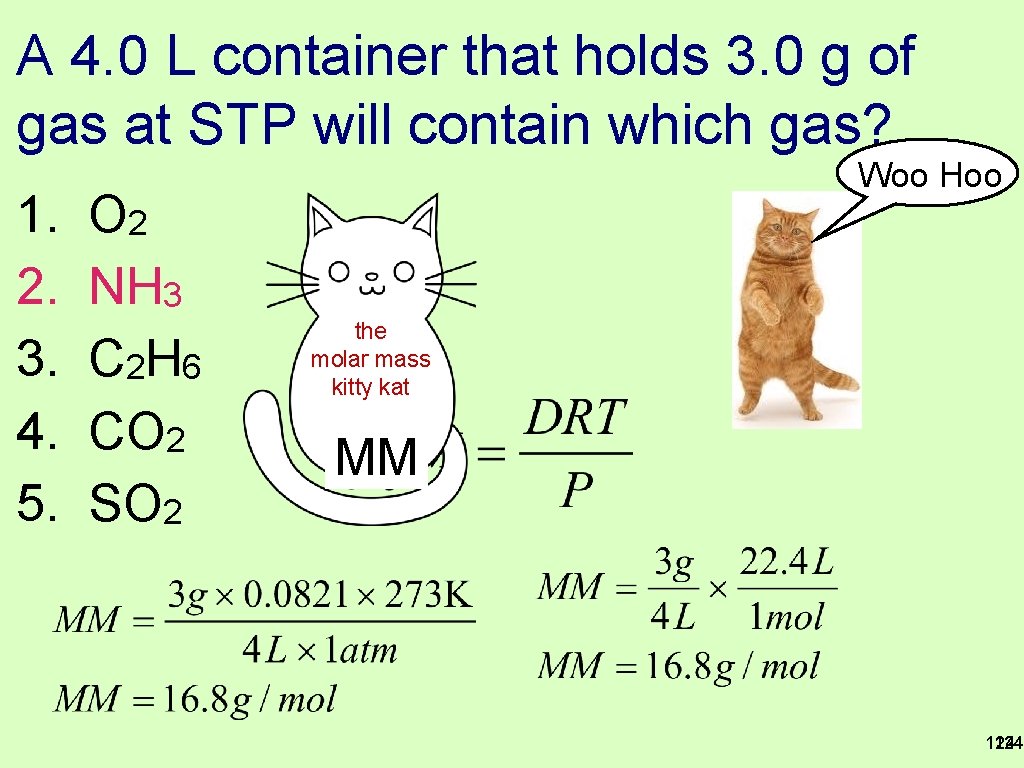
A 4. 0 L container that holds 3. 0 g of gas at STP will contain which gas? 1. 2. 3. 4. 5. O 2 NH 3 C 2 H 6 CO 2 SO 2 Woo Hoo the molar mass kitty kat MM 124

A flask that has a total pressure of 500 mm. Hg contains 0. 40 mole of SO 2, 0. 50 mole of N 2, and 0. 10 mole of He. Determine the partial pressure of SO 2 1. Report your answer to the 1’s place NO Calculators 125

A flask that has a total pressure of 500 mm. Hg contains 0. 40 mole of SO 2, 0. 50 mole of N 2, and 0. 10 mole of He. Determine the partial pressure of SO 2 1. Report your answer to the 1’s place 2. 1 mole of gas in total 3. SO is 40% of the gas, thus 40% of 500 = 200 mm NO Calculators 126

NH 4 NO 3(s) → N 2 O(g) + 2 H 2 O(g) When 8. 0 g of ammonium nitrate is sealed in a 2. 0 L flask and heated to 300. ºC, it completely decomposes. Calculate the total pressure in the flask. yes calculator 127

NH 4 NO 3(s) → N 2 O(g) + 2 H 2 O(g) When 8. 0 g of ammonium nitrate is sealed in a 2. 0 L flask and heated to 300. ºC, it completely decomposes. Calculate the total pressure in the 1. input your answer flask. 128

When the stopcock is opened, and the two gases mix, the temperature remains constant, calculate the total pressure in the apparatus? O 2 4 L 0. 5 atm N 2 1 L 1. 5 atm 1. Input your answer 129

When the stopcock is opened, and the two gases mix, the temperature remains constant, calculate the total pressure in the apparatus? O 2 4 L 0. 5 atm N 2 1 L 1. 5 atm 1. 0. 7 atm 130
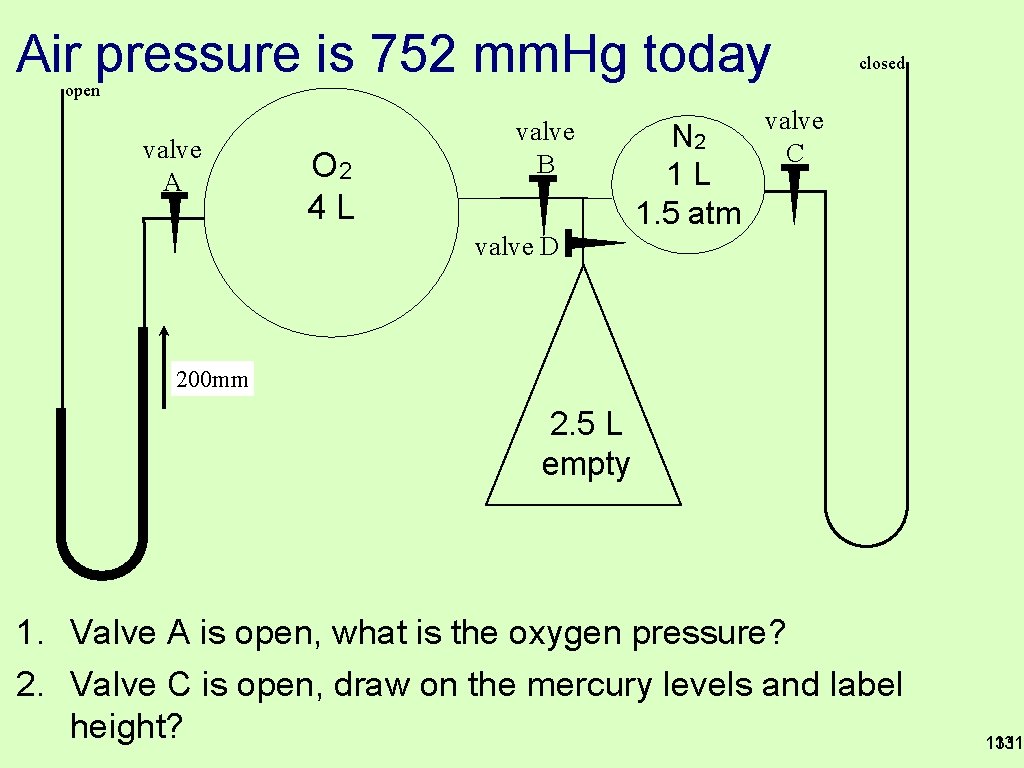
Air pressure is 752 mm. Hg today closed open valve A O 2 4 L valve B N 2 1 L 1. 5 atm valve C valve D 200 mm 2. 5 L empty 1. Valve A is open, what is the oxygen pressure? 2. Valve C is open, draw on the mercury levels and label height? 131
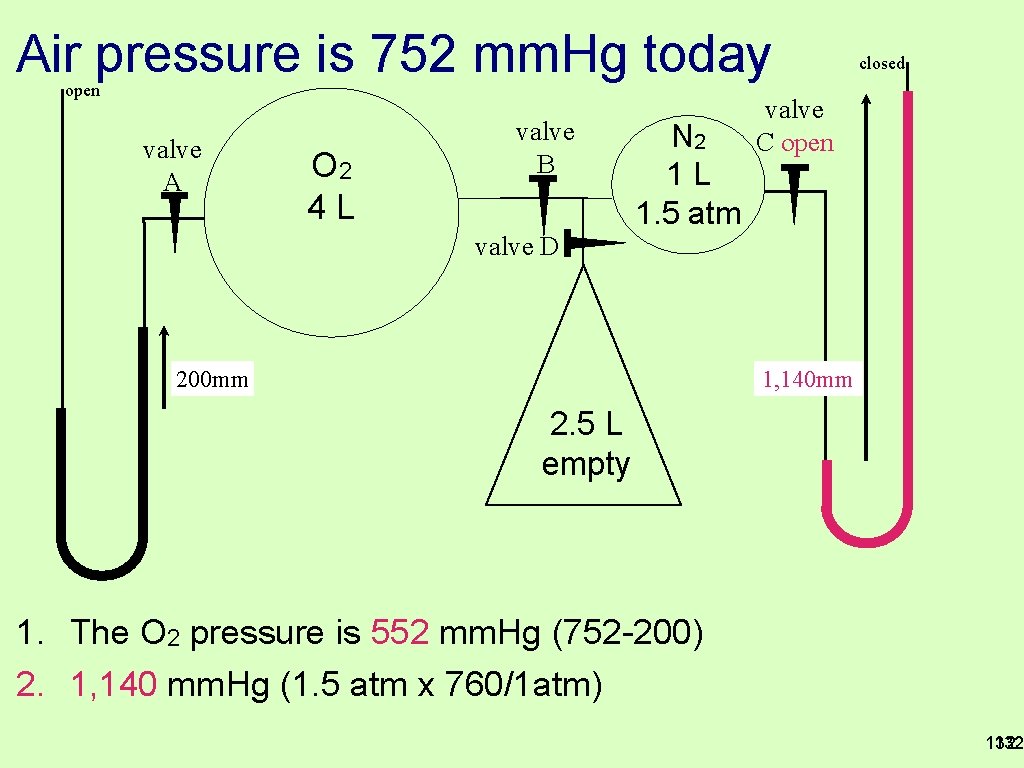
Air pressure is 752 mm. Hg today open valve A O 2 4 L valve B N 2 1 L 1. 5 atm closed valve C open valve D 1, 140 mm 200 mm 2. 5 L empty 1. The O 2 pressure is 552 mm. Hg (752 -200) 2. 1, 140 mm. Hg (1. 5 atm x 760/1 atm) 132
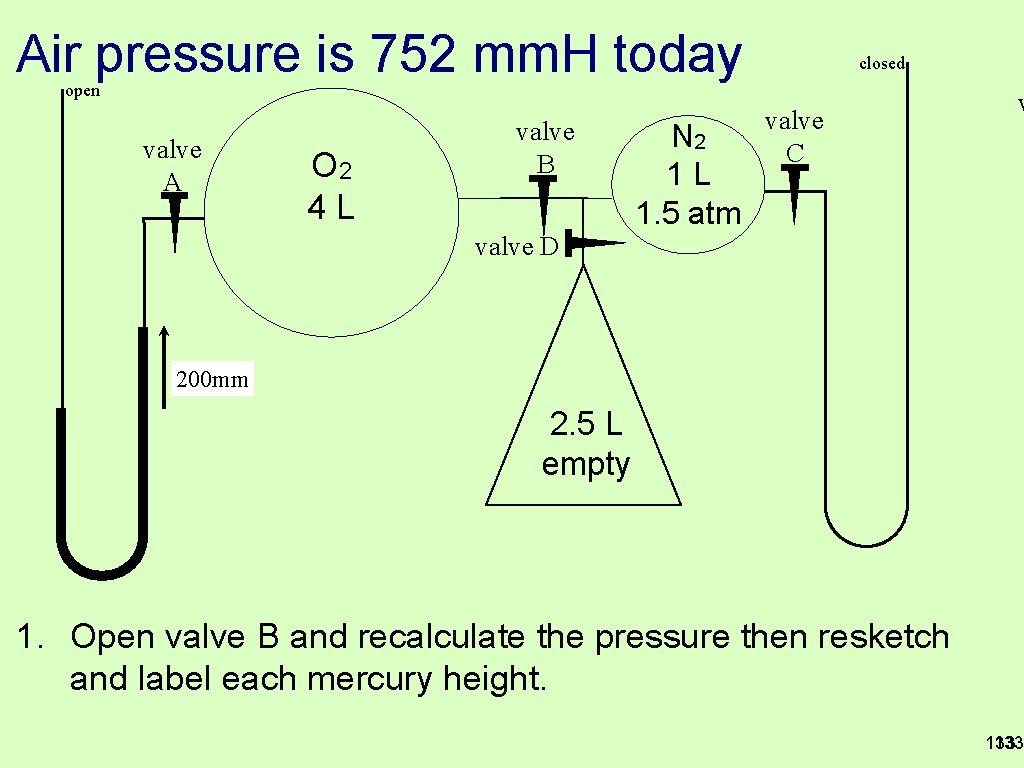
Air pressure is 752 mm. H today closed open valve A O 2 4 L valve B N 2 1 L 1. 5 atm valve C v valve D 200 mm 2. 5 L empty 1. Open valve B and recalculate the pressure then resketch and label each mercury height. 133
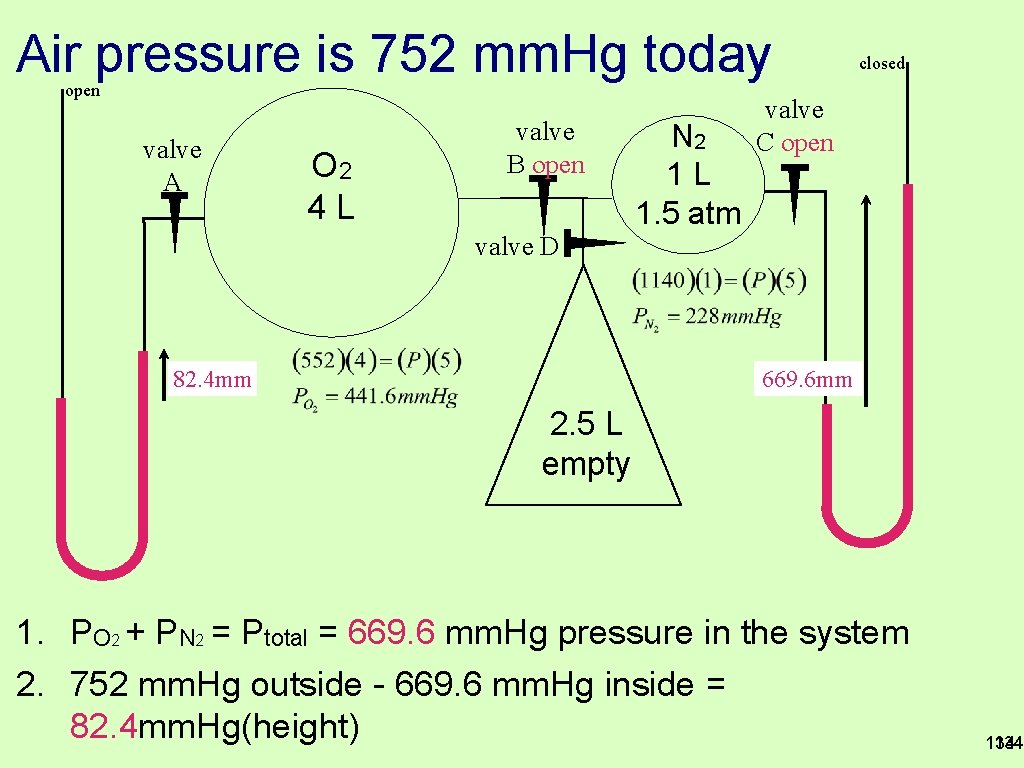
Air pressure is 752 mm. Hg today open valve A O 2 4 L valve B open N 2 1 L 1. 5 atm closed valve C open valve D 669. 6 mm 82. 4 mm 2. 5 L empty 1. PO 2 + PN 2 = Ptotal = 669. 6 mm. Hg pressure in the system 2. 752 mm. Hg outside - 669. 6 mm. Hg inside = 82. 4 mm. Hg(height) 134
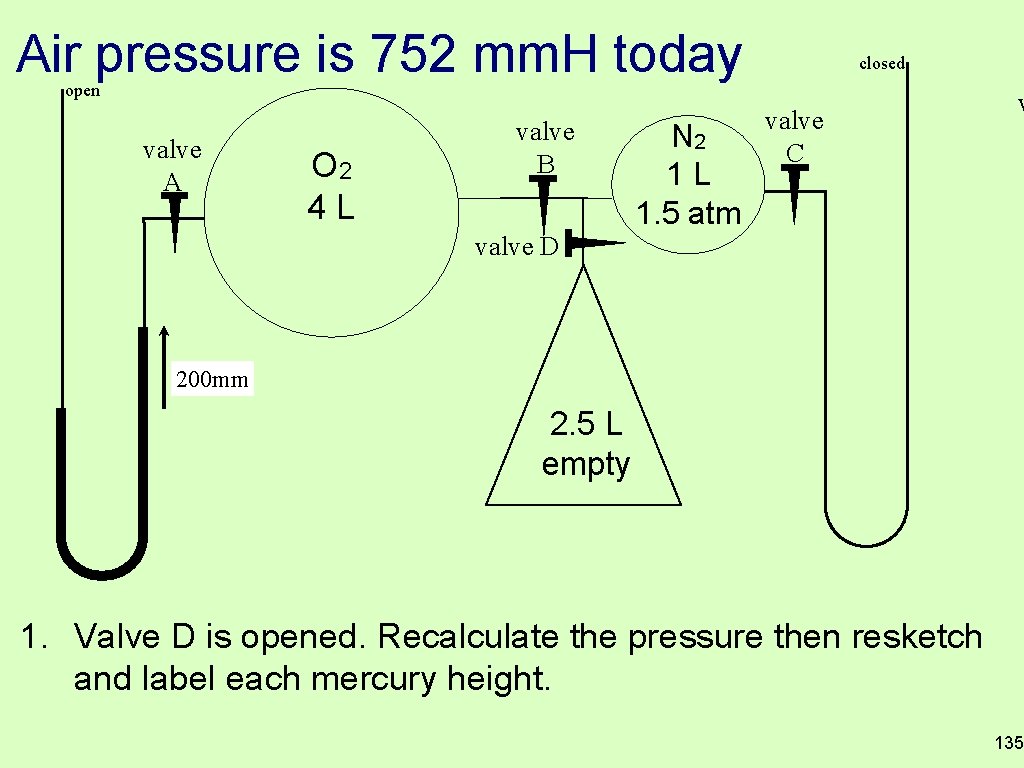
Air pressure is 752 mm. H today closed open valve A O 2 4 L valve B N 2 1 L 1. 5 atm valve C v valve D 200 mm 2. 5 L empty 1. Valve D is opened. Recalculate the pressure then resketch and label each mercury height. 135
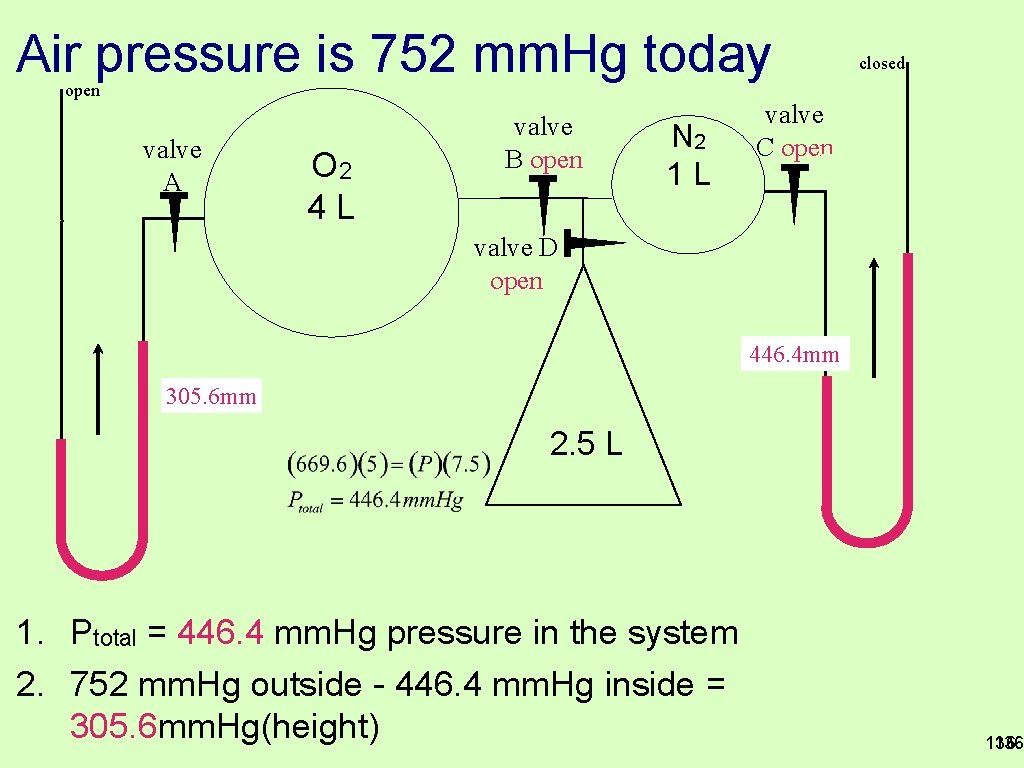
Air pressure is 752 mm. Hg today open valve A O 2 4 L valve B open N 2 1 L closed valve C open valve D open 446. 4 mm 305. 6 mm 2. 5 L 1. Ptotal = 446. 4 mm. Hg pressure in the system 2. 752 mm. Hg outside - 446. 4 mm. Hg inside = 305. 6 mm. Hg(height) 136

Iron oxide and carbon monoxide react together according to the equation shown below. If you had 112 L of CO at STP and 240 g of Fe 2 O 3 what volume of CO 2 can you produce at STP? 3 CO(g) + Fe 2 O 3(g) → 2 Fe(s) + 6 CO 2(g) 137

If 3. 21 mole of a gas occupies 56. 2 L at some temperature and pressure, 5. 29 mole of this gas will occupy what volume under these same conditions. 1. 2. 3. 4. 5. 606 m. L 1. 65 L 34. 1 L 92. 6 L You can not determine the volume without knowing the temperature and pressure. 138

If 3. 21 mole of a gas occupies 56. 2 L at some temperature and pressure, 5. 29 mole of this gas will occupy what volume under these same conditions. 1. 2. 3. 4. 5. 606 m. L 1. 65 L 34. 1 L 92. 6 L You can not determine the volume without knowing the temperature and pressure. 139

If 50. 0 g of a gas occupies 10. 0 L at STP, 150 g of this gas will occupy what volume at STP? 1. 2. 3. 4. 5. 6. 7. Woo Hoo 3. 92 L 50. 8 L 12. 9 L 25. 5 L 5. 08 L 30. 0 L cannot be determined without knowing who the gas is. 140

According to kinetic molecular theory, molecules of different gases at the same temperature always have the same. (Select as many as apply. ) 1. average kinetic energy 2. molecular mass 3. pressure 4. molecular speed 5. average density 141

According to kinetic molecular theory, molecules of different gases at the same temperature always have the same. (Select as many as apply. ) 1. average kinetic energy ✴temperature is proportional to kinetic energy since temperature is merely a measure of the “wiggle” of molecules. 2. molecular mass 3. pressure 4. molecular speed 142 5. average density

L Avogadro’s Hypothesis in 1811 1. 5 atm 2 L 22º Same Pressure? 2 L 22º 1. 5 atm 22º How many atoms? 1. 5 atm 22º 3 L 2 atm 15º How many atoms? 2 L 2 atm 15º 143

L Avogadro’s Hypothesis in 1811 1. 5 atm 2 L 22º Same Pressure? 2 L 22º 1. 5 atm 22º How many atoms? 1. 5 atm 22º 3 L 2 atm 15º The law is named after Amedeo Avogadro who, in 1811, hypothesized that two given samples of an ideal gas, of the same volume and at the same temperature and pressure, contain the same number of molecules. How many atoms? 2 L 2 atm 15º 144

When 100 ml of nitrogen gas is added to 150 ml of hydrogen gas and reacted to form ammonia, as in the equation below, at a constant temperature and constant pressure, what volume of ammonia will be produced? N 2(g) + 3 H 2(g) → 2 NH 3(g) 1. 2. 3. 4. 5. 6. 350 ml 200 ml 150 ml 100 ml Can not determine the volume without more temperature and pressure information. 145

When 100 ml of nitrogen gas is added to 150 ml of hydrogen gas and reacted to form ammonia, as in the equation below, at a constant temperature and constant pressure, what volume of ammonia will be produced? N 2(g) + 3 H 2(g) → 2 NH 3(g) 100 ml 150 ml limits 1. 2. 3. 4. 5. 350 ml 200 ml 150 ml 100 ml At constant conditions, the volume of a gas is proportional to the number of moles of gas. At constant conditions we can “do stoichiometry” with volumes. We do not need to be in moles. 146

What is the temperature of 0. 444 mole of CO gas in a 11. 8 L container at 889 torr. 1. 2. 3. 4. 5. 106ºC 73ºC 14ºC 32ºC 379ºC 147

In a 3. 0 L container at 20ºC that contains 5 g of gas, which gas will have the greatest pressure? 1. 2. 3. 4. 5. He Ne Ar Kr They will all have the same pressure because they are all monatomic noble gases. 148

The volume of a 2. 49 g sample of gas was 752 ml at 1. 98 atm and 62°C. The gas is 1. 2. 3. 4. 5. SO 2 SO 3 NH 3 NO 2 Ne 149

Determine the molar mass of a gas that has a density of 6. 7 g/L at STP. 1. 2. 3. 4. 5. 496 g/mole 150 g/mole 73. 0 g/mole 3. 35 g/mole 0. 298 g/mole 150

Automobile air bags use the decomposition of sodium azide as their sources of gas for rapid inflation, represented in the reaction below. What mass of Na. N 3 is required to provide 40. 0 L of N 2 at 25°C and 763 torr? 2 Na. N 3(s) → 2 Na(s) + 3 N 2(g) 1. 2. 3. 4. 5. 16. 4 g 1. 09 g 160 g 71. 1 g 107 g 151

Potassium chlorate can be decomposed to produce oxygen gas in the laboratory by the reaction below. What volume of dry oxygen gas at 25°C and 1. 00 atm pressure is produced by the decomposition of 7. 5 g of KCl. O 3(s)? 2 KCl. O 3(s) → 2 KCl(s) + 3 O 2(g) 1. 2. 3. 4. 5 L 7. 5 L 2. 2 L 3. 7 L 11 L 152

Determine the total pressure in a 12. 2 L vessel that contains 2. 34 g of carbon dioxide, 1. 73 g of sulfur dioxide, and 3. 33 g of argon, at 42°C. 1. 2. 3. 4. 5. 263 mm. Hg 134 mm. Hg 395 mm. Hg 116 mm. Hg 0. 347 mm. Hg 153

A gas mixture of Ne and Ar has a total pressure of 4. 00 atm and contains 16. 0 moles of gas. If the partial pressure of Ne is 2. 75 atm, how many moles of Ar are in the mixture? 1. 2. 3. 4. 5. 00 moles 6. 75 moles 9. 25 moles 11. 0 moles 12. 0 moles 154

According to the kinetic molecular theory, if the temperature of a gas were raised from 100°C to 200ºC, the average kinetic energy if the gas will 1. 2. 3. 4. 5. 6. double increase by a factor of 1. 27 increase by a factor of 100 decrease by half decrease by a factor of 100 remain the same 155

Speed of Gases How fast do they go? What affects their speed? 156
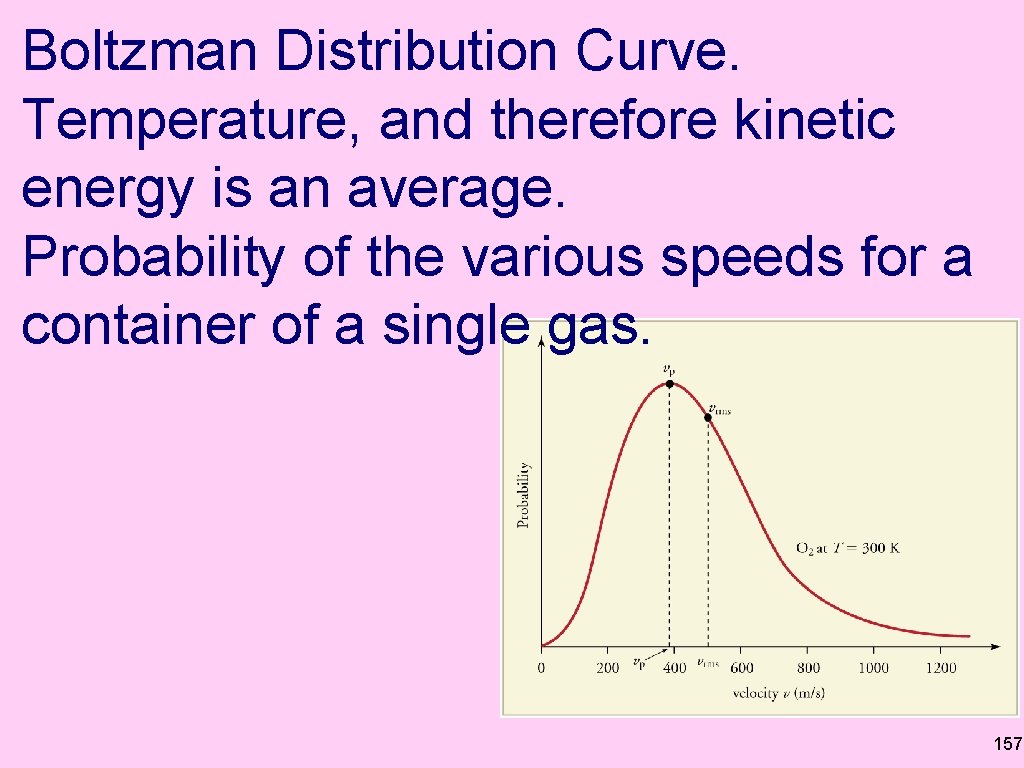
Boltzman Distribution Curve. Temperature, and therefore kinetic energy is an average. Probability of the various speeds for a container of a single gas. 157
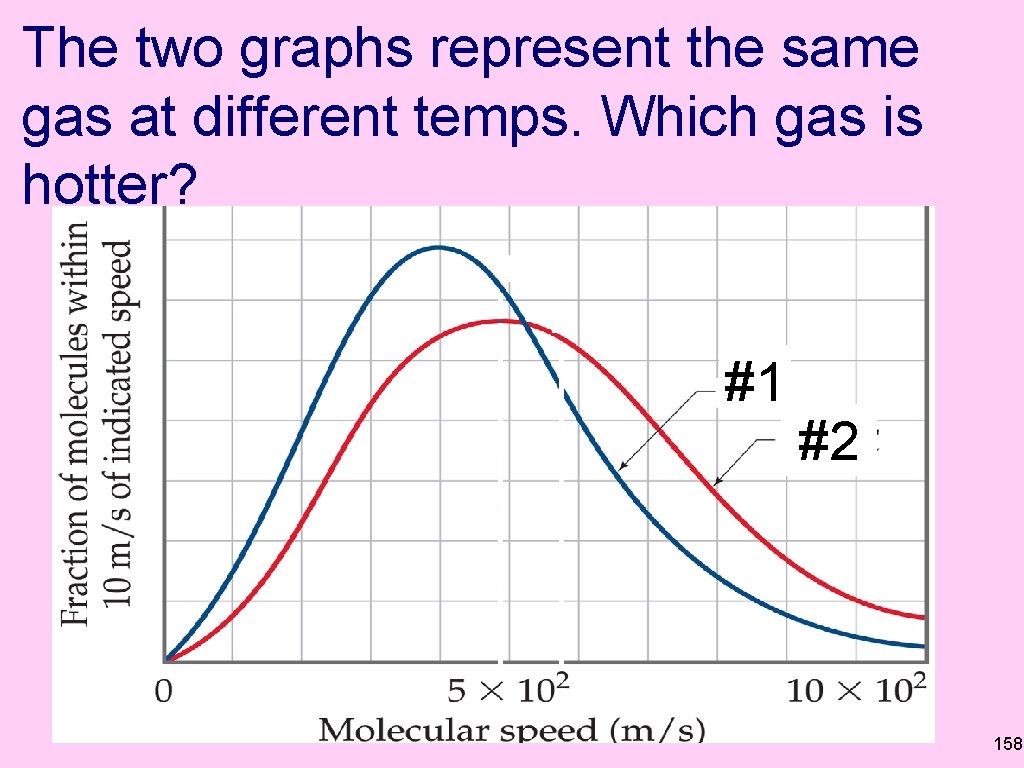
The two graphs represent the same gas at different temps. Which gas is hotter? #1 #2 158
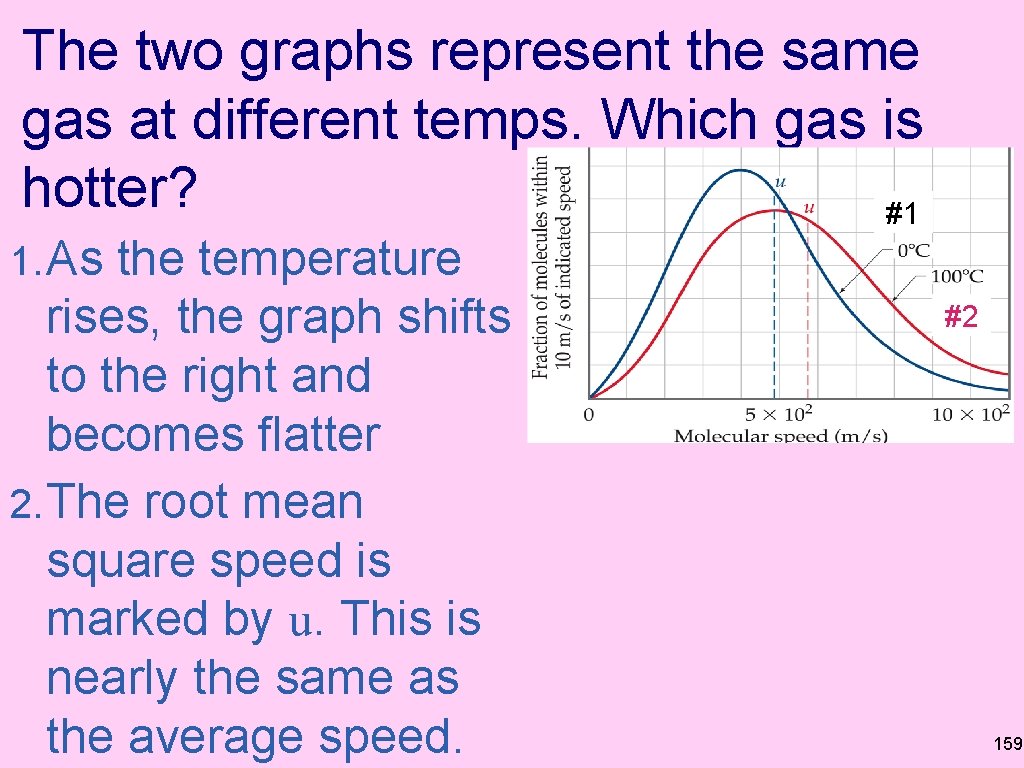
The two graphs represent the same gas at different temps. Which gas is hotter? #1 1. As the temperature rises, the graph shifts to the right and becomes flatter 2. The root mean square speed is marked by u. This is nearly the same as the average speed. #2 159
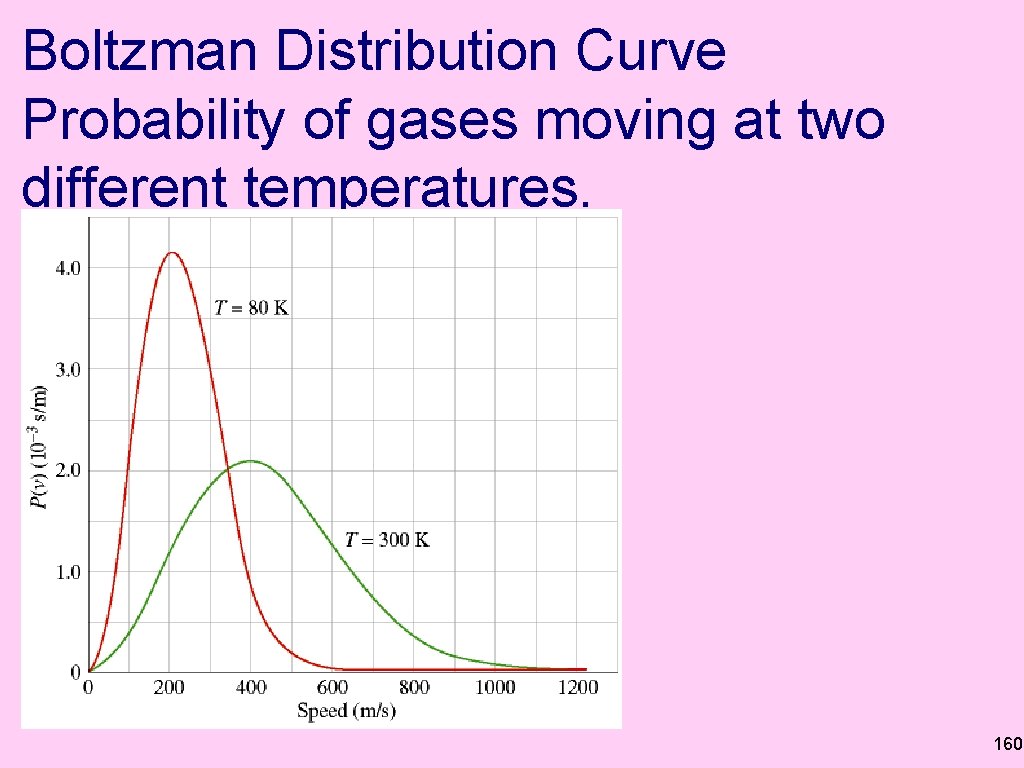
Boltzman Distribution Curve Probability of gases moving at two different temperatures. 160

The white powder will form a ring 1. 2. 3. 4. 5. closer to the NH 3 closer to the HCl in the middle wherever by chance no way of knowing 161

The white powder will form a ring 1. closer to the NH 3 2. Graham’s Law of effusion says that the rates of effusion for two gases is inversely proportional to the square roots of their molar masses 3. Check your blue sheets for an equation (Though you may be asked this on MC as well. ) 2. closer to the HCl 3. in the middle 4. wherever by chance 5. no way of knowing 162

Compare the speed of helium compared to carbon dioxide. 1. He is faster 2. CO 2 is faster 3. He and CO 2 travel at the same speed 163

Compare the speed of helium compared to carbon dioxide. 1. He is faster 2. CO 2 is faster 3. Graham’s Law of effusion says that the rates of effusion for two gases is inversely proportional to their molar masses 164

Since T α KE = • • 2 ½mv If gases are at the same temp and therefore have the same average KE, the equation above indicates that lighter gases (smaller molar mass) must have a larger speed (v). The bottom line? Low MM gasses go fast ✓ High MM gases go slower ✓ 165
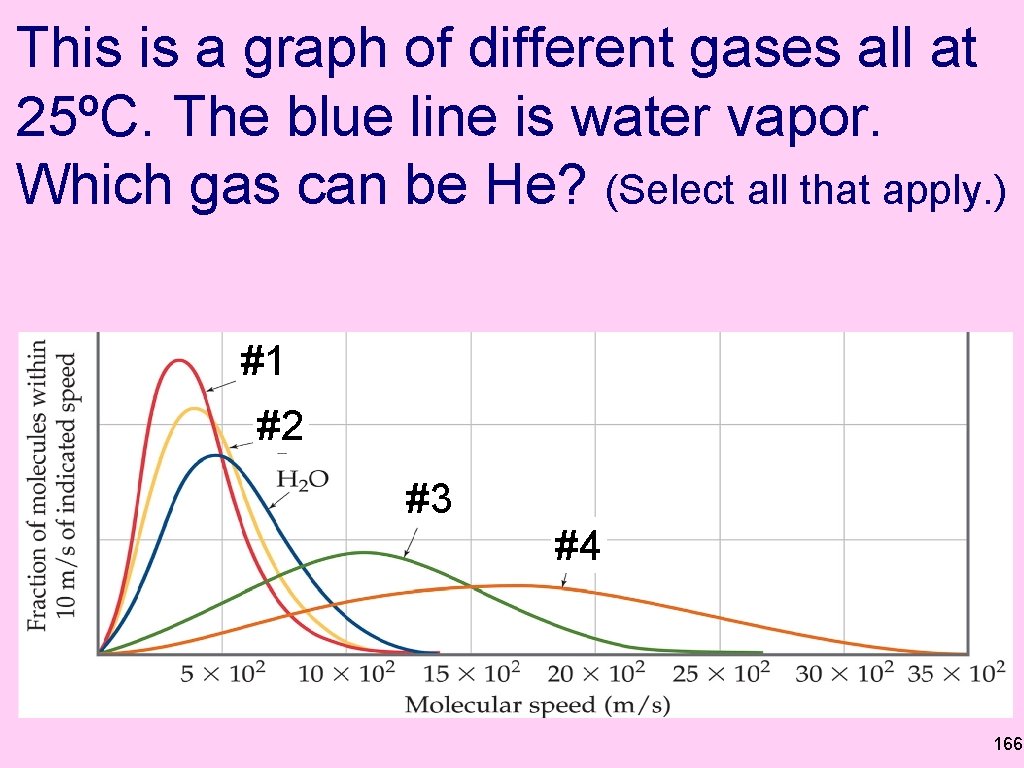
This is a graph of different gases all at 25ºC. The blue line is water vapor. Which gas can be He? (Select all that apply. ) #1 #2 #3 #4 166
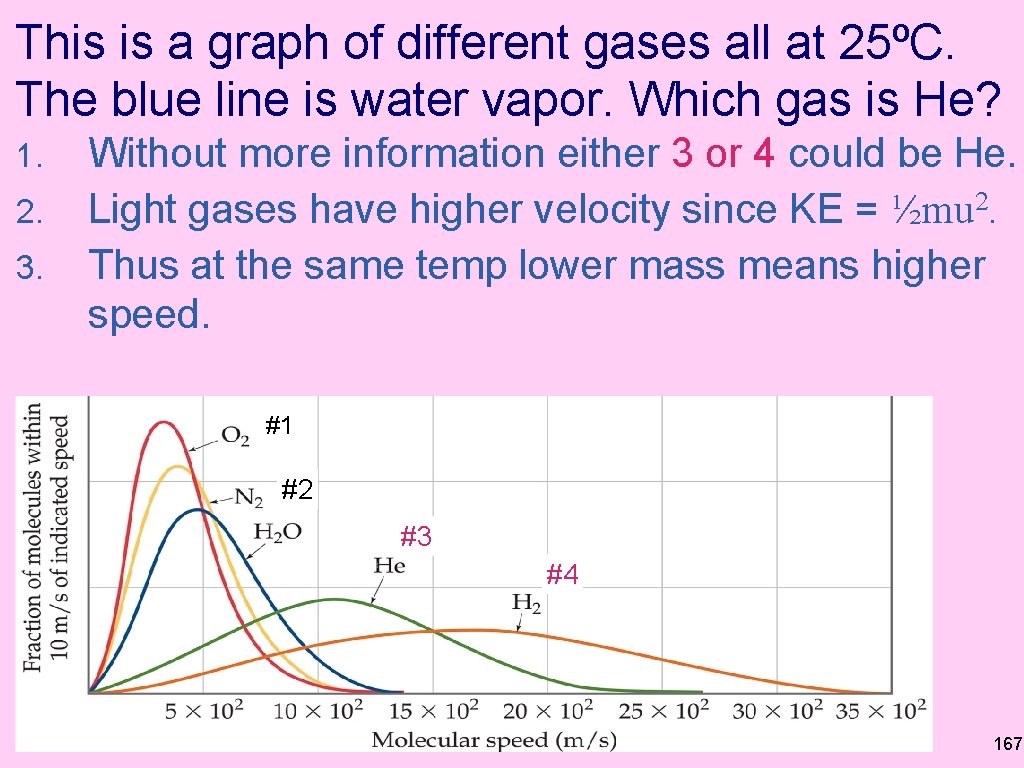
This is a graph of different gases all at 25ºC. The blue line is water vapor. Which gas is He? 1. 2. 3. Without more information either 3 or 4 could be He. Light gases have higher velocity since KE = ½mu 2. Thus at the same temp lower mass means higher speed. #1 #2 #3 #4 167

Select the gas that has an average speed closest to Ne. All gases at same temp & pressure. 1. 2. 3. 4. 5. 6. H 2 He H 2 O CO 2 They all have the same speed, since the conditions are the same. 168

Select the gas that has an average speed closest to Ne at STP. 1. 2. 3. 4. 5. 6. H 2 • the mass of the gas will affect the speed. He • thus the gas with the most similar H 2 O molar mass will be the gas with the closest speed. CO 2 They all have the same speed, since the conditions are the same. 169

Select the gas that has the highest average kinetic energy. All gases at STP. 1. 2. 3. 4. 5. 6. 7. H 2 He H 2 O CO 2 All of these gases have the same KE None of the gases have kinetic energy since they are all at 0ºC. 170

Select the gas that has the highest kinetic energy at STP. 1. 2. 3. 4. 5. H 2 He H 2 O CO 2 • T α KE O 2 6. All of the gases have the same kinetic energy since they are at the same temperature. 6. None of the gases have kinetic energy 171

Effusion and Diffusion • Effusion ✓The escape of gas molecules through a tiny hole into an evacuated space. • Diffusion ✓The spread of one substance throughout a space or throughout a second substance. 172

What’s so Ideal about the Ideal Gas Law? PV = n. RT 173

The “Ideal” Gas Law 1. Kinetic Molecular Theory the temperature of a gas is propotional to its kinetic energy. 2. KMT is dependent upon a gas behaving as if the gas particles have NO IMFs. 3. KMT also considers gases to be “point masses. ” That is to say, the size of each atom is insignificant. • If the statements above are true, the gases will behave “ideally. ” nd rd • However, we know the 2 & 3 statements aren’t always so perfectly true. 174
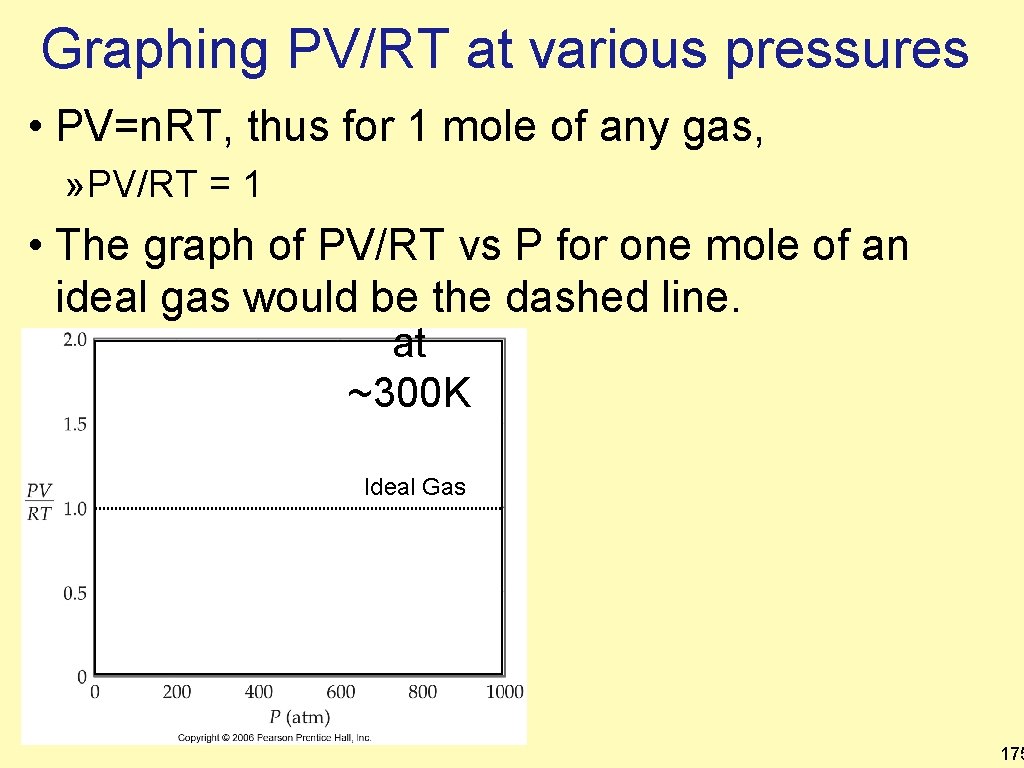
Graphing PV/RT at various pressures • PV=n. RT, thus for 1 mole of any gas, » PV/RT = 1 • The graph of PV/RT vs P for one mole of an ideal gas would be the dashed line. at ~300 K Ideal Gas 175
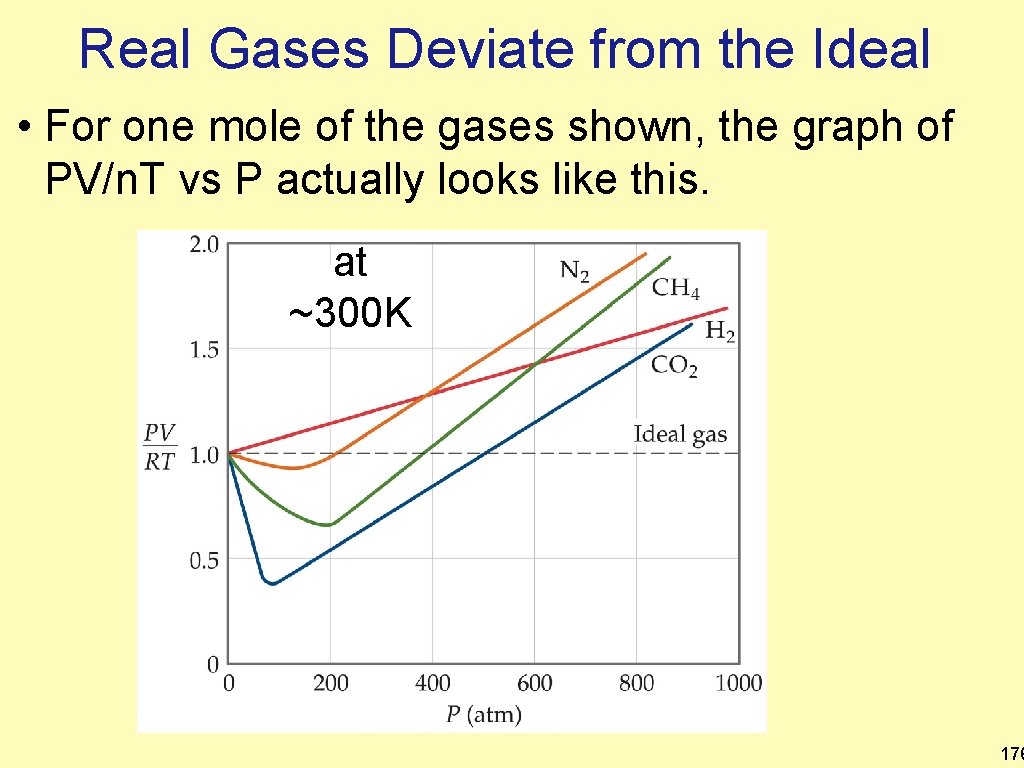
Real Gases Deviate from the Ideal • For one mole of the gases shown, the graph of PV/n. T vs P actually looks like this. at ~300 K 176
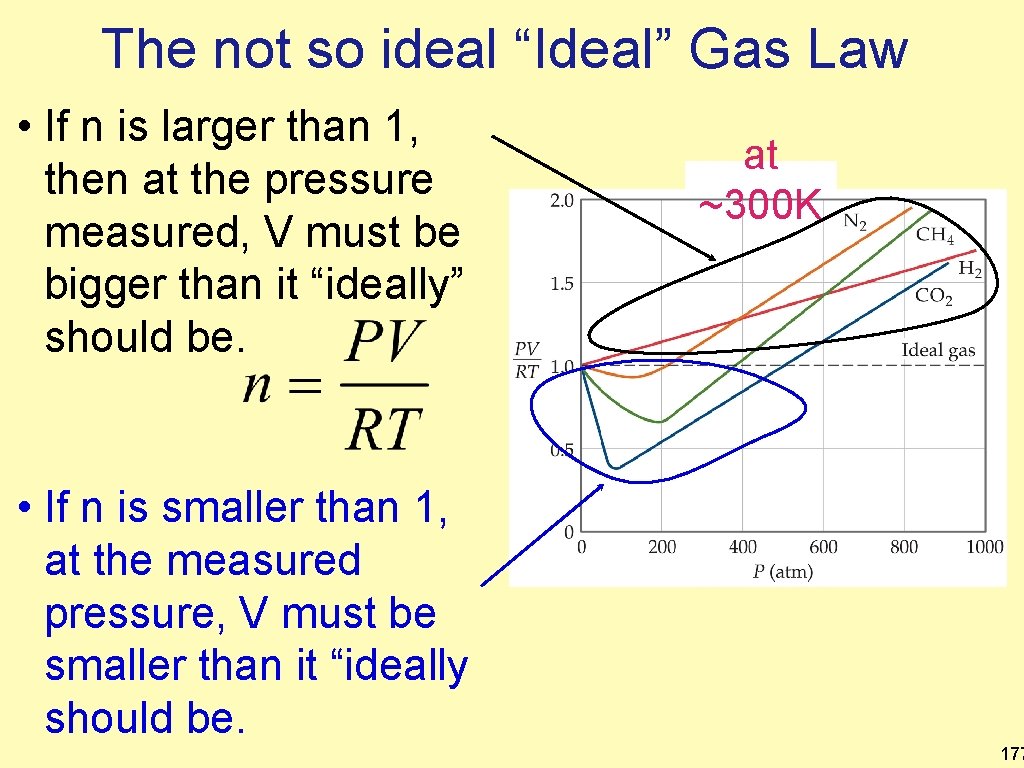
The not so ideal “Ideal” Gas Law • If n is larger than 1, then at the pressure measured, V must be bigger than it “ideally” should be. at ~300 K • If n is smaller than 1, at the measured pressure, V must be smaller than it “ideally should be. 177
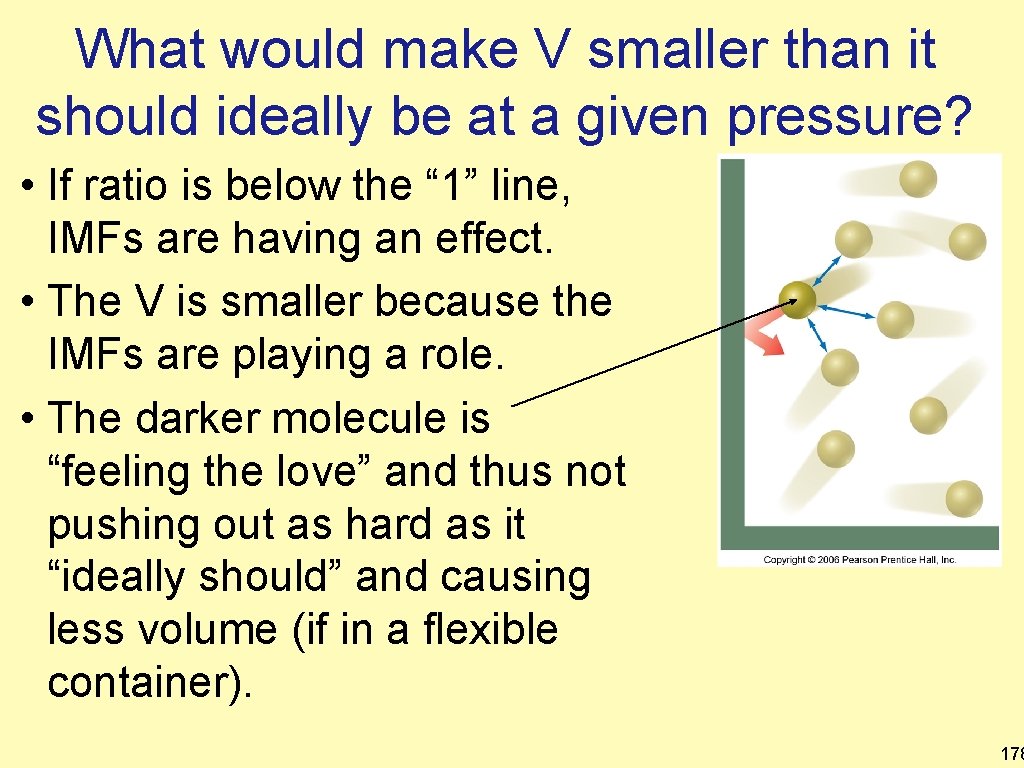
What would make V smaller than it should ideally be at a given pressure? • If ratio is below the “ 1” line, IMFs are having an effect. • The V is smaller because the IMFs are playing a role. • The darker molecule is “feeling the love” and thus not pushing out as hard as it “ideally should” and causing less volume (if in a flexible container). 178

What would make V larger than it should ideally be at a given pressure? • Usually molecules are far apart and their personal particle size is irrelevant, however when “cramped” together, the particle’s personal size begins to matter. • At higher pressures, the volume of each individual molecule begins to matter and actually takes up 179
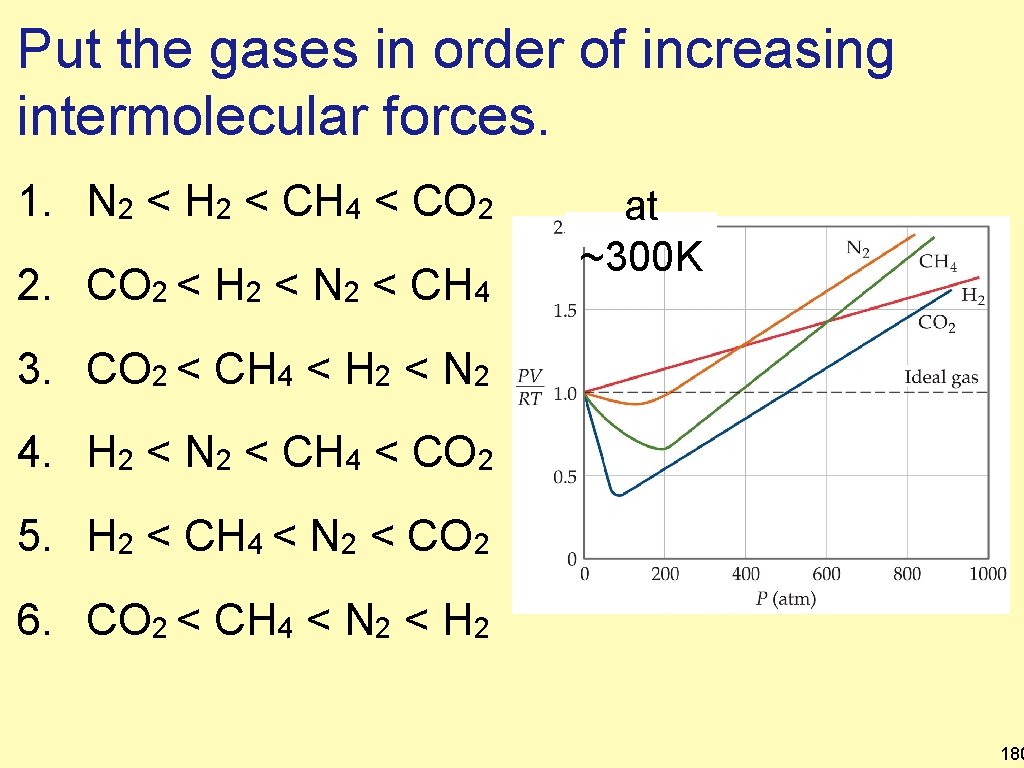
Put the gases in order of increasing intermolecular forces. 1. N 2 < H 2 < CH 4 < CO 2 2. CO 2 < H 2 < N 2 < CH 4 at ~300 K 3. CO 2 < CH 4 < H 2 < N 2 4. H 2 < N 2 < CH 4 < CO 2 5. H 2 < CH 4 < N 2 < CO 2 6. CO 2 < CH 4 < N 2 < H 2 180
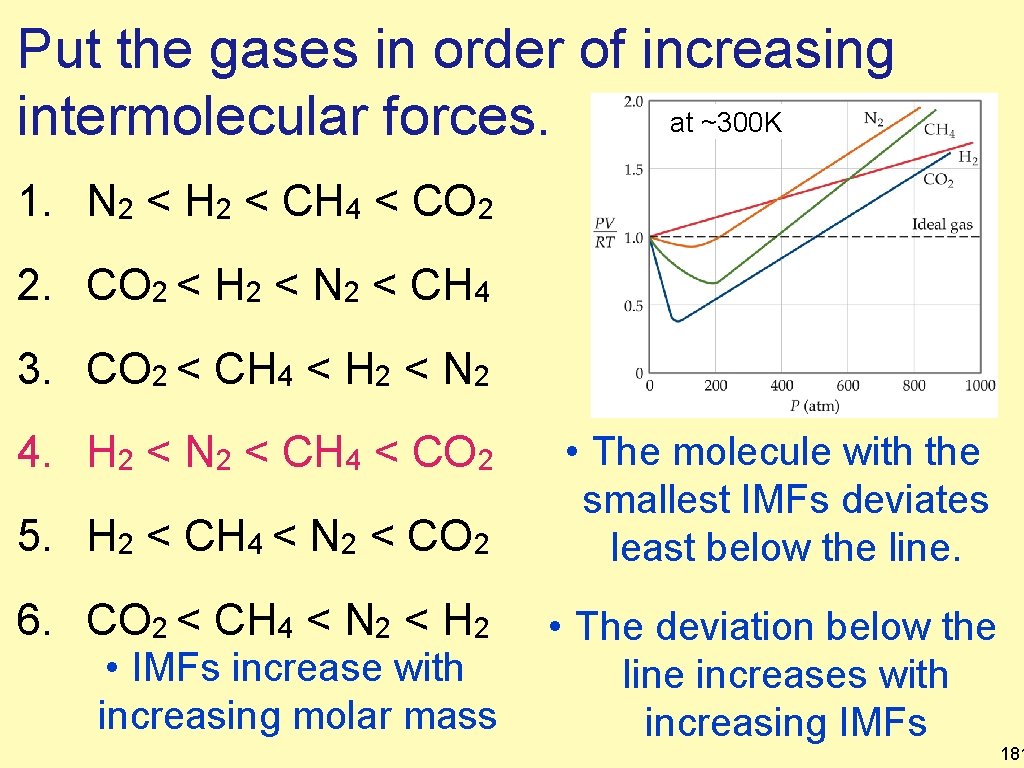
Put the gases in order of increasing at ~300 K intermolecular forces. 1. N 2 < H 2 < CH 4 < CO 2 2. CO 2 < H 2 < N 2 < CH 4 3. CO 2 < CH 4 < H 2 < N 2 4. H 2 < N 2 < CH 4 < CO 2 5. H 2 < CH 4 < N 2 < CO 2 6. CO 2 < CH 4 < N 2 < H 2 • IMFs increase with increasing molar mass • The molecule with the smallest IMFs deviates least below the line. • The deviation below the line increases with increasing IMFs 181
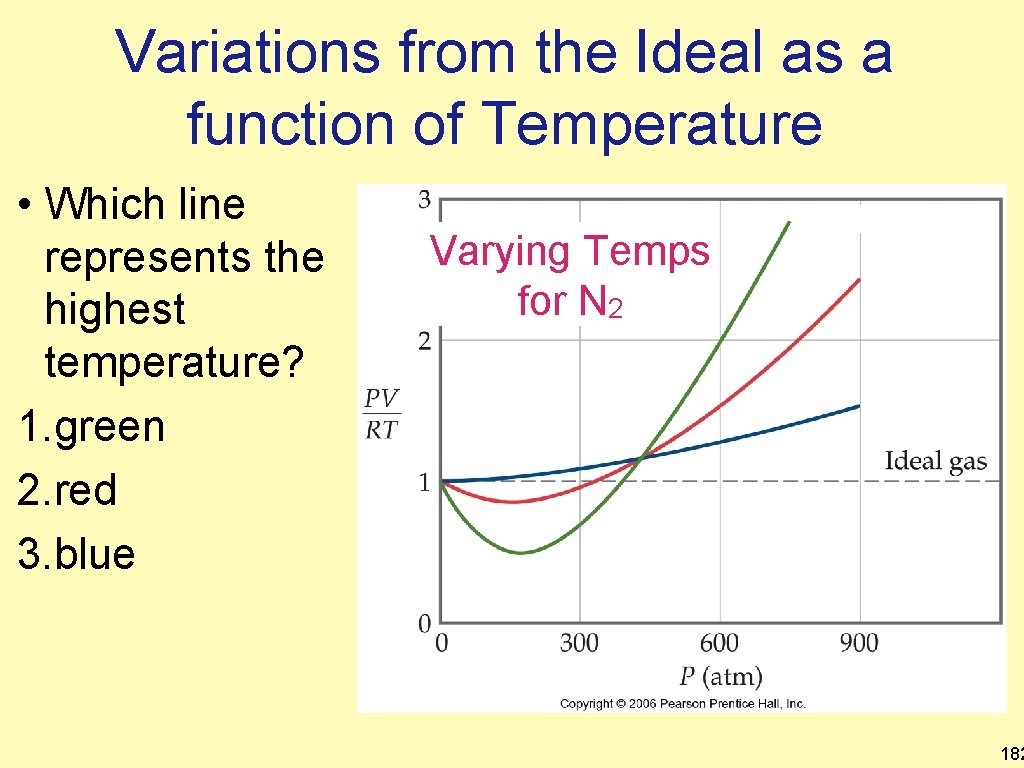
Variations from the Ideal as a function of Temperature • Which line represents the highest temperature? 1. green 2. red 3. blue Varying Temps for N 2 182
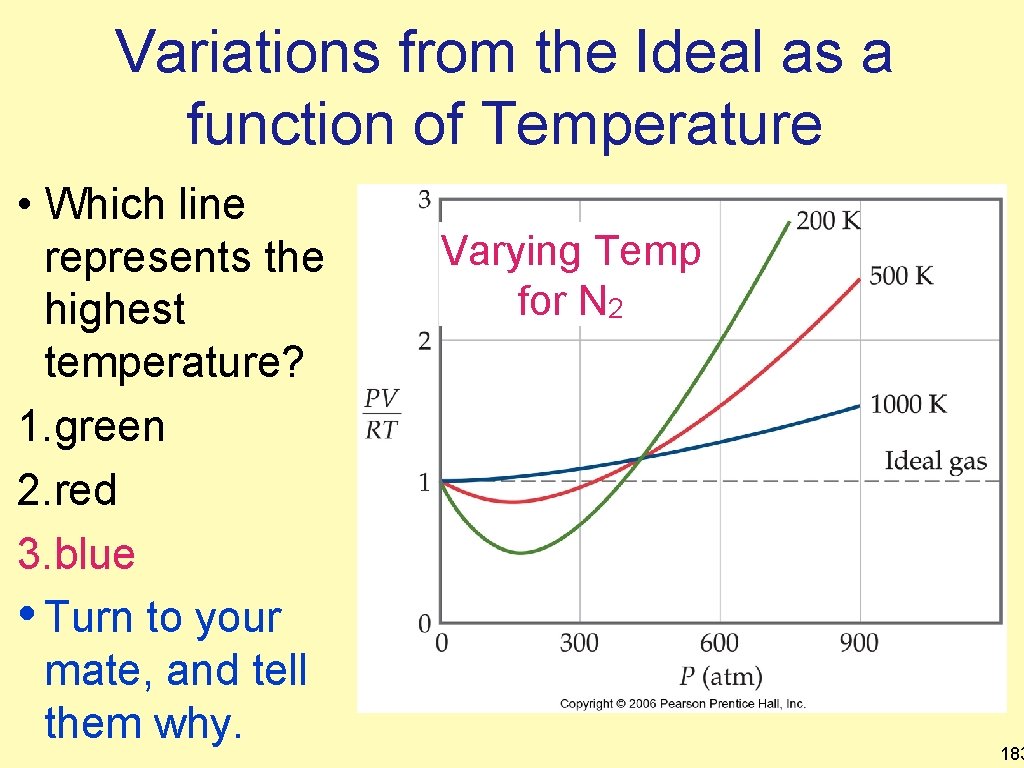
Variations from the Ideal as a function of Temperature • Which line represents the highest temperature? 1. green 2. red 3. blue • Turn to your mate, and tell them why. Varying Temp for N 2 183

Variations from the Ideal as a function of Temperature • • Remember that deviations below the line arise from intermolecular forces. At higher temperatures, IMFs have less effect, thus the line does not dip below as much or at all. Varying Temps for N 2 184

185

The van der Waals Equation Predicting Real Gas Pressures • • • written this way in your correction fortext: correction for IMFs volume of molecules 186
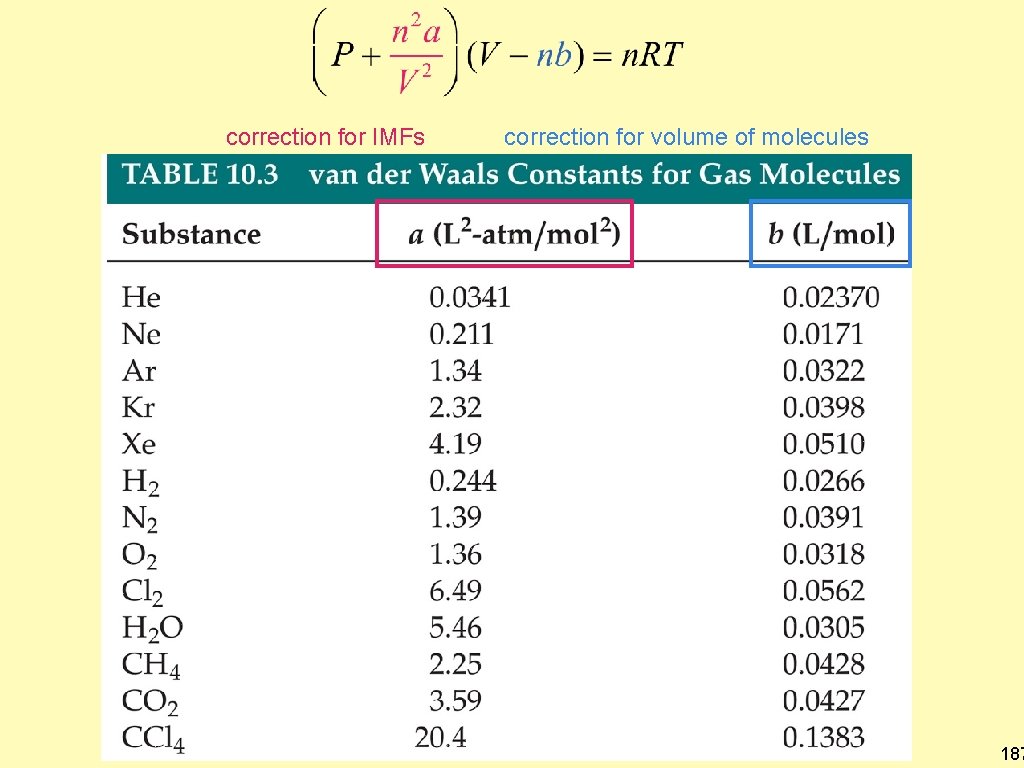
correction for IMFs correction for volume of molecules 187
- Slides: 187