14 2 The Gas Laws This hot air



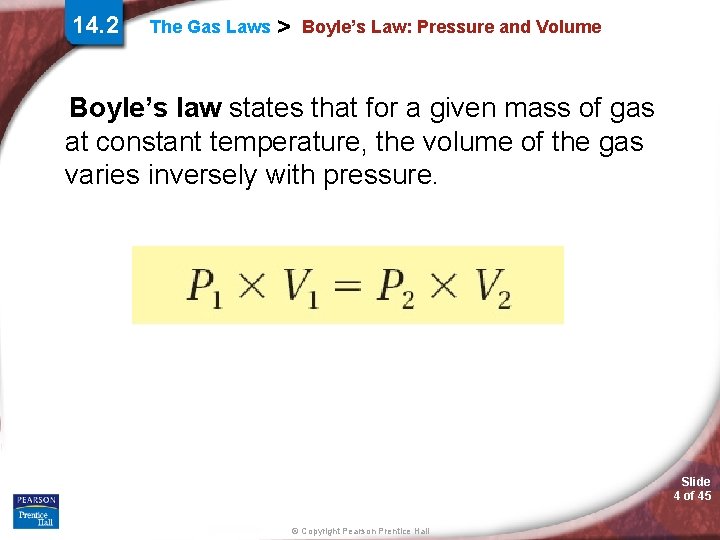







































- Slides: 45

14. 2 The Gas Laws This hot air balloon was designed to carry a passenger around the world. You will study some laws that will allow you to predict gas behavior under specific conditions, such as in a hot air balloon. Slide 1 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Boyle’s Law: Pressure and Volume How are the pressure, volume, and temperature of a gas related? Slide 2 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Boyle’s Law: Pressure and Volume If the temperature is constant, as the pressure of a gas increases, the volume decreases. Slide 3 of 45 © Copyright Pearson Prentice Hall
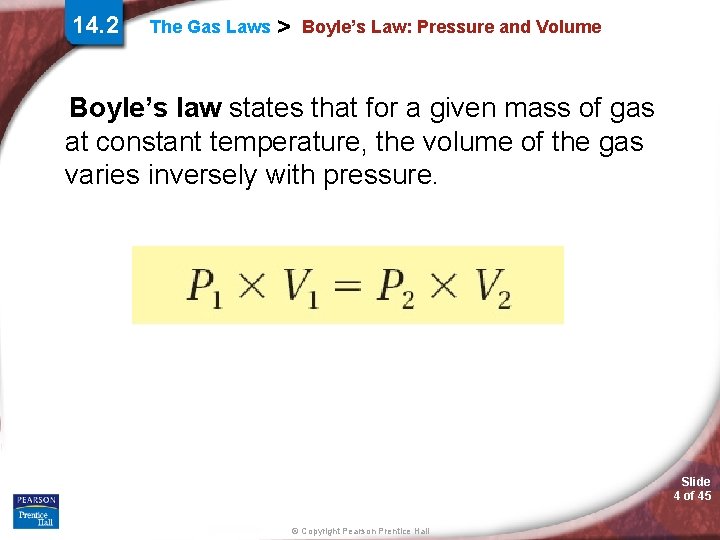
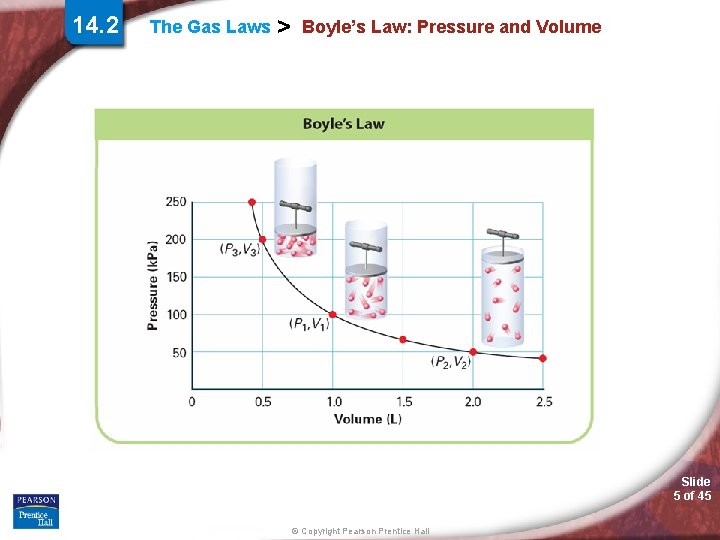
14. 2 The Gas Laws > Boyle’s Law: Pressure and Volume Boyle’s law states that for a given mass of gas at constant temperature, the volume of the gas varies inversely with pressure. Slide 4 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Boyle’s Law: Pressure and Volume Slide 5 of 45 © Copyright Pearson Prentice Hall

The Gas Laws > Boyle’s Law: Pressure and Volume Simulation 15 Examine the relationship between gas, volume and pressure. Slide 6 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 1 Slide 7 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 1 Slide 8 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 1 Slide 9 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 1 Slide 10 of 45 © Copyright Pearson Prentice Hall

Practice Problems for Sample Problem 14. 1 Problem Solving 14. 8 Solve Problem 8 with the help of an interactive guided tutorial. © Copyright Pearson Prentice Hall Slide 11 of 45

14. 2 The Gas Laws > Charles’s Law: Temperature and Volume As the temperature of an enclosed gas increases, the volume increases, if the pressure is constant. Slide 12 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Charles’s Law: Temperature and Volume As the temperature of the water increases, the volume of the balloon increases. Slide 13 of 45 © Copyright Pearson Prentice Hall

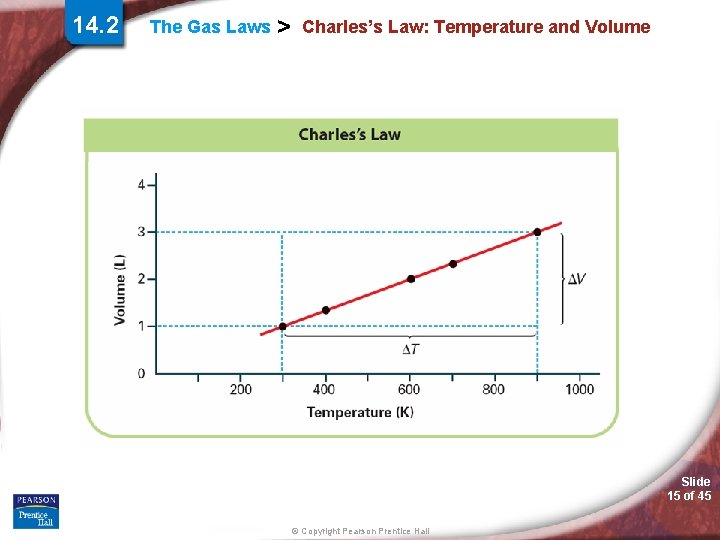
14. 2 The Gas Laws > Charles’s Law: Temperature and Volume Charles’s law states that the volume of a fixed mass of gas is directly proportional to its Kelvin temperature if the pressure is kept constant. Slide 14 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Charles’s Law: Temperature and Volume Slide 15 of 45 © Copyright Pearson Prentice Hall

The Gas Laws > Charles’s Law: Temperature and Volume Simulation 16 Examine the relationship between gas volume and temperature. Slide 16 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 2 Slide 17 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 2 Slide 18 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 2 Slide 19 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 2 Slide 20 of 45 © Copyright Pearson Prentice Hall

Practice Problems for Sample Problem 14. 2 Problem Solving 14. 10 Solve Problem 10 with the help of an interactive guided tutorial. © Copyright Pearson Prentice Hall Slide 21 of 45

14. 2 The Gas Laws > Gay-Lussac’s Law: Pressure and Temperature As the temperature of an enclosed gas increases, the pressure increases, if the volume is constant. Slide 22 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Gay-Lussac’s Law: Pressure and Temperature When a gas is heated at constant volume, the pressure increases. Slide 23 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Gay-Lussac’s Law: Pressure and Temperature Gay-Lussac’s law states that the pressure of a gas is directly proportional to the Kelvin temperature if the volume remains constant. Slide 24 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > Gay-Lussac’s Law: Pressure and Temperature A pressure cooker demonstrates Gay. Lussac’s Law. Slide 25 of 45 © Copyright Pearson Prentice Hall

The Gas Laws > Gay-Lussac’s Law: Pressure and Temperature Simulation 17 Examine the relationship between gas pressure and temperature. Slide 26 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 3 Slide 27 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 3 Slide 28 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 3 Slide 29 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 3 Slide 30 of 45 © Copyright Pearson Prentice Hall

Practice Problems for Sample Problem 14. 3 Problem Solving 14. 12 Solve Problem 12 with the help of an interactive guided tutorial. © Copyright Pearson Prentice Hall Slide 31 of 45

14. 2 The Gas Laws > The Combined Gas Law When is the combined gas law used to solve problems? Slide 32 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > The Combined Gas Law The combined gas law describes the relationship among the pressure, temperature, and volume of an enclosed gas. Slide 33 of 45 © Copyright Pearson Prentice Hall

14. 2 The Gas Laws > The Combined Gas Law The combined gas law allows you to do calculations for situations in which only the amount of gas is constant. Slide 34 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 4 Slide 35 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 4 Slide 36 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 4 Slide 37 of 45 © Copyright Pearson Prentice Hall

SAMPLE PROBLEM 14. 4 Slide 38 of 45 © Copyright Pearson Prentice Hall

Practice Problems for Sample Problem 14. 4 Problem Solving 14. 14 Solve Problem 14 with the help of an interactive guided tutorial. © Copyright Pearson Prentice Hall Slide 39 of 45

14. 2 The Gas Laws > The Combined Gas Law Weather balloons carry data-gathering instruments high into Earth’s atmosphere. At an altitude of about 27, 000 meters, the balloon bursts. Slide 40 of 45 © Copyright Pearson Prentice Hall

14. 2 Section Quiz. Assess students’ understanding of the concepts in Section 14. 2. Continue to: -or- Launch: Section Quiz Slide 41 of 45 © Copyright Pearson Prentice Hall

14. 2 Section Quiz. 1. If the volume of a gas in a container were reduced to one fifth the original volume at constant temperature, the pressure of the gas in the new volume would be a. one and one fifth times the original pressure. b. one fifth of the original pressure. c. four fifths of the original pressure. d. five times the original pressure. © Copyright Pearson Prentice Hall Slide 42 of 45

14. 2 Section Quiz. 2. A balloon appears slightly smaller when it is moved from the mountains to the seashore at constant temperature. The best gas law to explain this observation would be a. Gay-Lussacs's Law. b. Graham's Law. c. Boyle's Law. d. Charles's Law. Slide 43 of 45 © Copyright Pearson Prentice Hall

14. 2 Section Quiz. 3. At 46°C and 89 k. Pa pressure, a gas occupies a volume of 0. 600 L. How many liters will it occupy at 0°C and 20. 8 k. Pa? a. 0. 600 L b. 2. 58 L c. 0. 140 L d. 2. 20 L Slide 44 of 45 © Copyright Pearson Prentice Hall

END OF SHOW