Gas Laws The Gas Laws Describe HOW gases

Gas Laws

The Gas Laws • Describe HOW gases behave. • Can be predicted by the Molecular Kinetic Theory states that all matter consists of tiny particles that are in constant motion Gases have the greatest amount of motion, solids the least “Temperature” is our perception of molecular movement

Matter Review • Solids: least motion, definite volume definite shape • Liquids: more motion, particles further apart than solids (can pour), definite volume but indefinite shape • Gases: most motion of three states, indefinite volume and indefinite shape

4 things • In order to completely describe a gas you need to measure 4 things 1. 2. 3. 4. Pressure Temperature Volume Number of particles

Gas Pressure • Pressure is defined as force per unit area. • Gas particles exert pressure when they collide with the walls of their container. • The SI unit of pressure is the pascal (Pa). • However, there are several units of pressure – Pascal (Pa) – Kilopascal (k. Pa) – Atmosphere (atm)
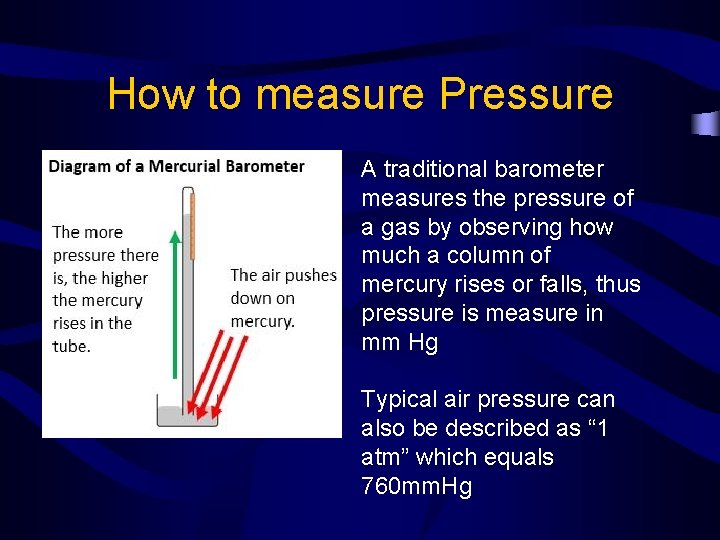
How to measure Pressure A traditional barometer measures the pressure of a gas by observing how much a column of mercury rises or falls, thus pressure is measure in mm Hg Typical air pressure can also be described as “ 1 atm” which equals 760 mm. Hg
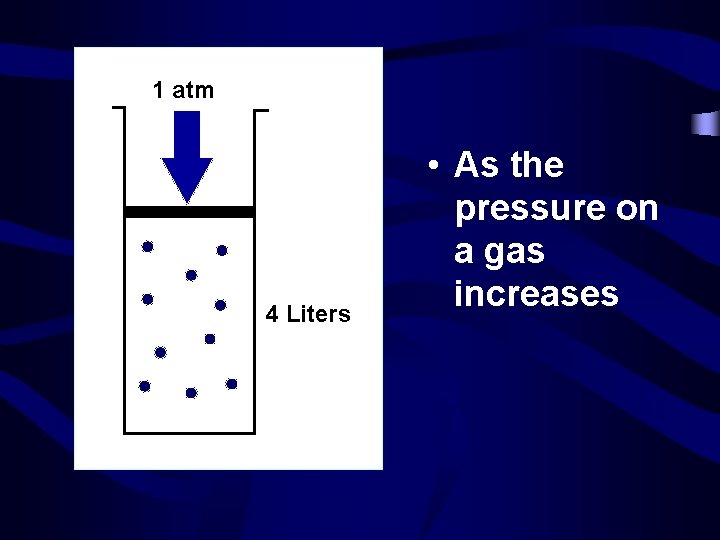
1 atm 4 Liters • As the pressure on a gas increases

2 atm 2 Liters • …the volume decreases • Pressure and volume are inversely related • P V =P V • Boyle’s Law 1 1 2 2
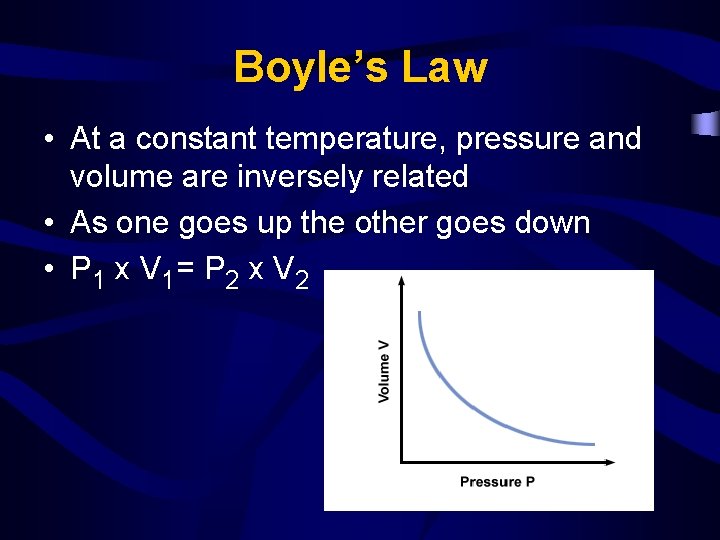
Boyle’s Law • At a constant temperature, pressure and volume are inversely related • As one goes up the other goes down • P 1 x V 1= P 2 x V 2

Temperature • Raising the temperature of a gas increases the pressure if the volume is held constant. • The molecules hit the walls harder. • The only way to increase the temperature at constant pressure is to increase the volume. If volume cannot change, such as a hard walled sealed container, the pressure must increase

Kelvin Scale Since gas laws are a result of Molecular motion, a temperature scale that reflects that must be used…no “negative” values! 0 K (zero kelvin) is called Absolute Zero…all molecular motion stops Celsius is converted to Kelvin K = Celsius temp + 273. 15 20 o. C +273. 15 = 293. 15 K Note…a degree symbol is used with Celsius but not with Kelvin

STP
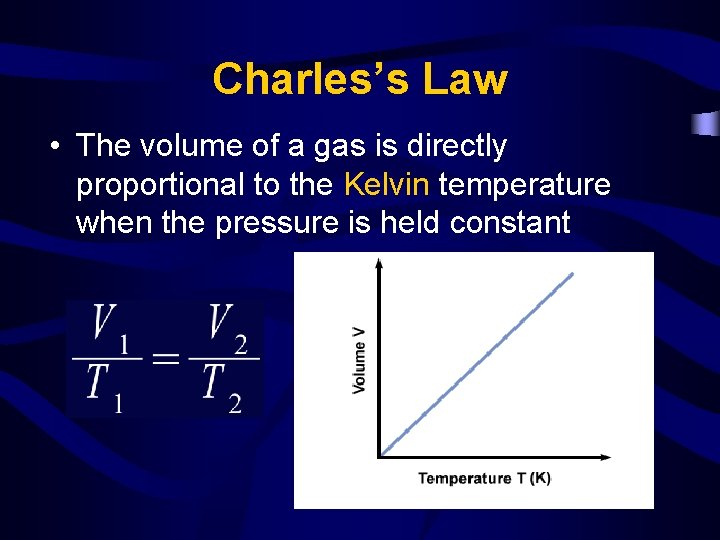
Charles’s Law • The volume of a gas is directly proportional to the Kelvin temperature when the pressure is held constant

Example • A sample of gas at 40. 0 °C occupies a volume of 2. 32 L. If the temperature is raised to 75. 0 °C what will the new volume be? V 1= 2. 32 L T 1= 40+273 V 2= ? T 2= 75+273 V 1 = V 2 T 1 T 2 2. 32 L = V 2 313 K 348 K MUST CONVERT TEMP TO K!!!!!!! 2. 58 L
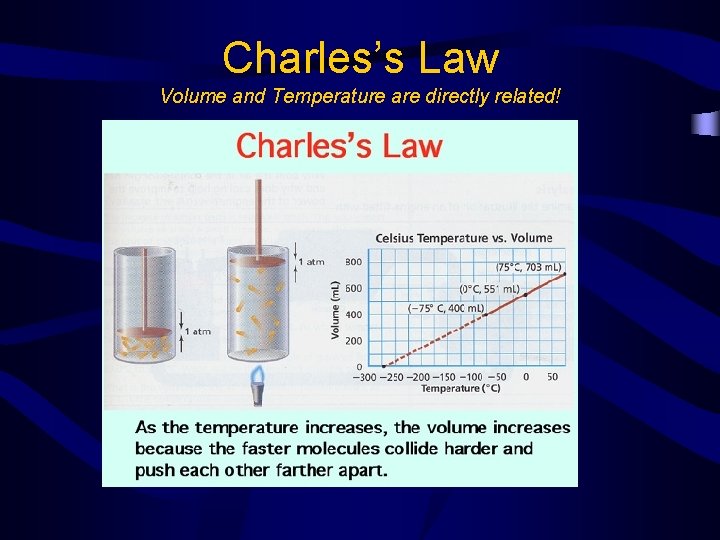
Charles’s Law Volume and Temperature are directly related!
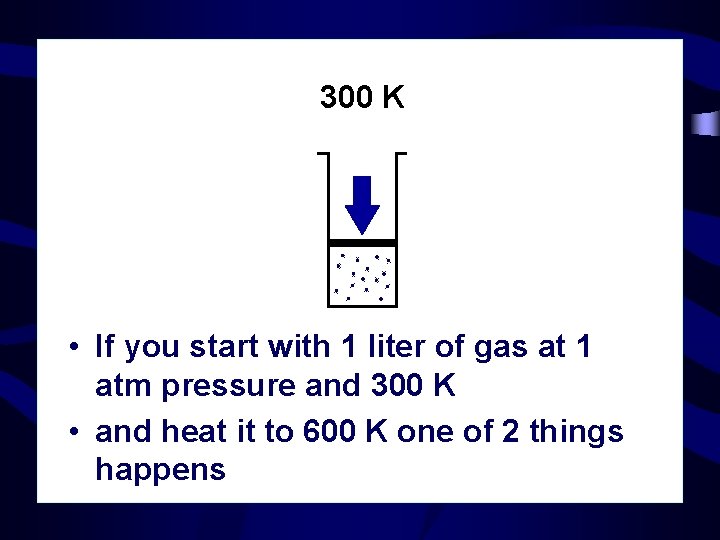
300 K • If you start with 1 liter of gas at 1 atm pressure and 300 K • and heat it to 600 K one of 2 things happens
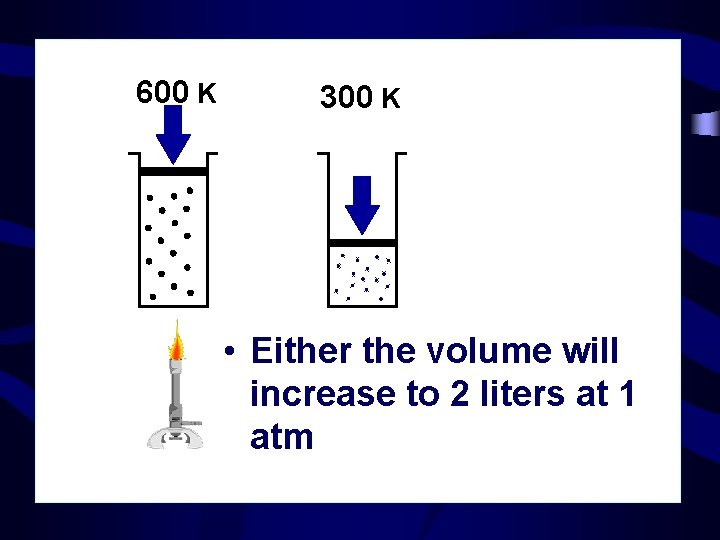
600 K 300 K • Either the volume will increase to 2 liters at 1 atm
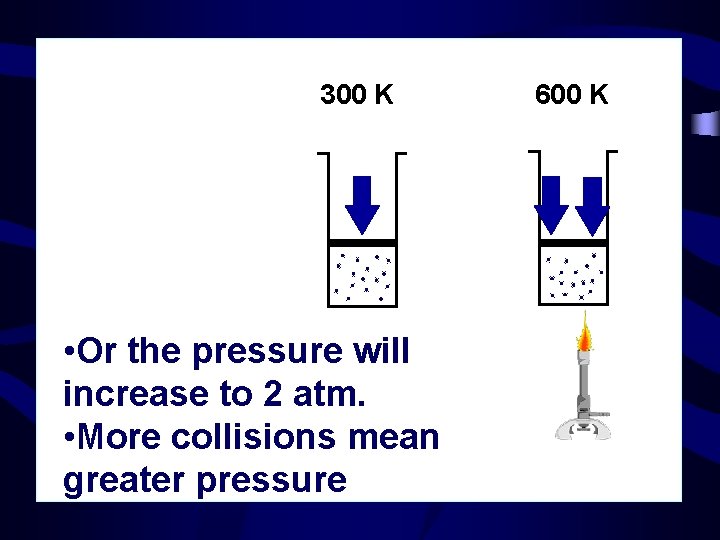
300 K • Or the pressure will increase to 2 atm. • More collisions mean greater pressure 600 K

Example • A balloon is filled with 25 L of air at 1. 0 atm pressure. If the pressure is changed to 1. 5 atm what is the new volume? P 1= 1 atm V 1= 25 L P 2= 1. 5 atm V 2= ? (1 atm)(25 L)=(1. 5 atm)(V 2) 16. 7 L

Examples • What is the new volume of a 0. 250 L balloon that starts at 25ºC if the temperature is raised to 100ºC? V 1=. 250 L T 1=25 C = 298. 15 K T 2=100 C = 373. 15 V 2=? =. 31 L

Combined Gas Law • The Combined Gas Law Deals with the situation where only the number of molecules stays constant. • P 1 x V 1 = P 2 x V 2 T 1 T 2

Example • A gas at 110. 0 k. Pa and 30. 0°C fills a flexible container to a volume of 2. 00 L. If the temperature was raised to 80. 0°C and the pressure was increased to 440. 0 k. Pa, what is the new volume?

Example • P 1 V 1 = P 2 V 2 T 1 T 2 • • • P 1 = 110. 0 k. Pa V 1 = 2. 00 L T 1 = 30. 0 °C = 303 K P 2 = 440. 0 k. Pa V 2 = ? T 2 = 80. 0 °C = 353 K

Example • P 1 V 1 = P 2 V 2 T 1 T 2 • (110. 0)(2. 00 L) = (440. 0 k. Pa)(V 2) 303 K 353 K • V 2 = 0. 583 L

The effect of adding gas • When we blow up a balloon we are adding gas molecules. • Doubling the number of gas particles doubles the pressure More molecules means more collisions Fewer molecules means fewer collisions.

• If you double the number of molecules… 1 atm
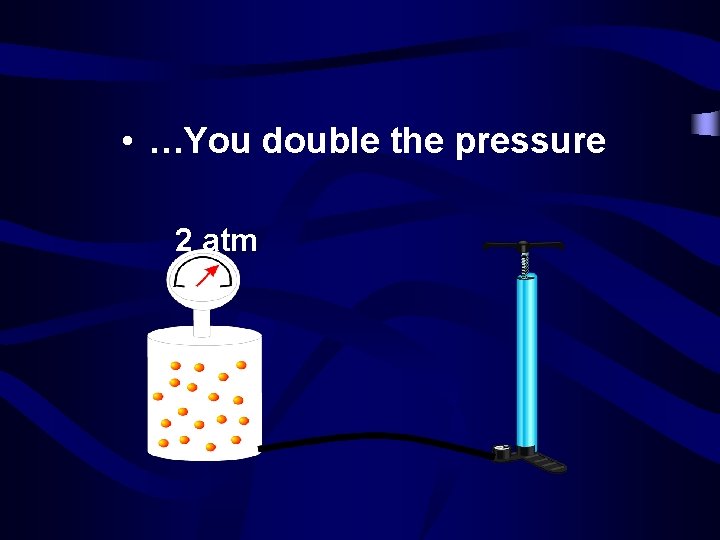
• …You double the pressure 2 atm

4 atm • As you remove molecules from a container……. .
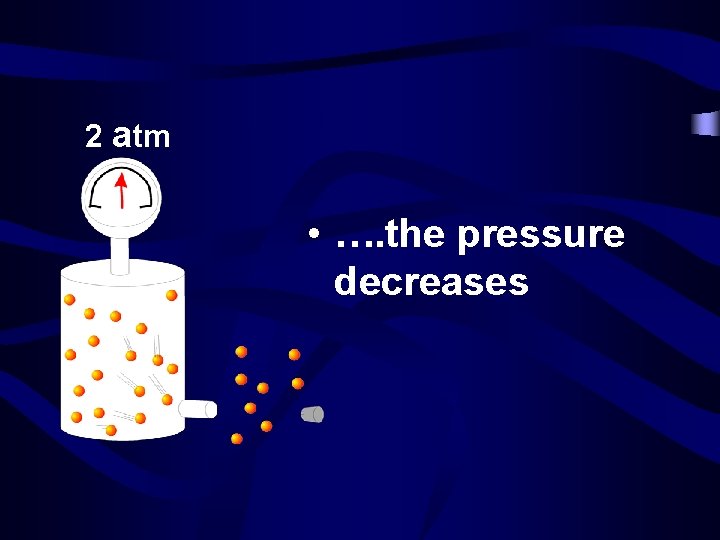
2 atm • …. the pressure decreases

Ideal Gases • We are going to assume that gases behave ideally • Does not really exist • Assume particles have no volume • Assume no attractive forces between molecules THE 2 ELEMENTS THAT BEHAVE MOST LIKE AN IDEAL GAS ARE HYDROGEN AND HELIUM

Ideal Gas Law • PV=n. RT • Where n is the number of moles and R is a constant • Use whatever R value that gives the correct pressure and/or volume units

Ideal Gases vs Real Gases deviate from the Ideal Gases: 1) Molar volume of a gas will differ, at STP not exactly 22. 4 L 2) Gas particles can condense, so they do have some forces of attraction between particles Real gases differ when at low temp and high pressure

Dalton’s Law of Partial Pressures • The total pressure inside a container is equal to the sum of the partial pressure due to each gas. • The partial pressure of a gas is the contribution by that gas hitting the wall. • PTotal = P 1 + P 2 + P 3 + …
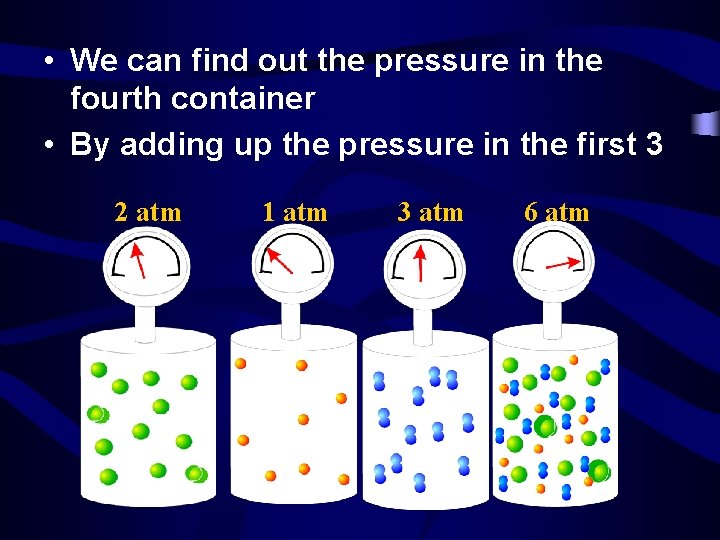
• We can find out the pressure in the fourth container • By adding up the pressure in the first 3 2 atm 1 atm 3 atm 6 atm

Dalton’s Law of Partial Pressures A gas mixture contains H 2, He, Ne, and Ar. The total pressure of the mixture is 93. 6 k. Pa. The partial pressures of He, Ne, and Ar are 15. 4 k. Pa, 25. 7 k. Pa, and 35. 6 k. Pa respectively. What is the pressure exerted by H 2? PT = PH 2 + PHe + PNe + PAr PH 2 = 16. 9 k. Pa

Dalton’s Law of Partial Pressures A person using an oxygen mask is breathing air with 33% Oxygen. What is the partial pressure of the Oxygen when the air pressure in the mask is 110 k. Pa? 33% of 110 k. Pa 36 k. Pa

Diffusion & Effusion u Molecules moving from areas of high concentration to low concentration. u Perfume molecules spreading across the room. u Effusion - Gas escaping through a tiny hole in a container. u Both depend on the speed of the molecules
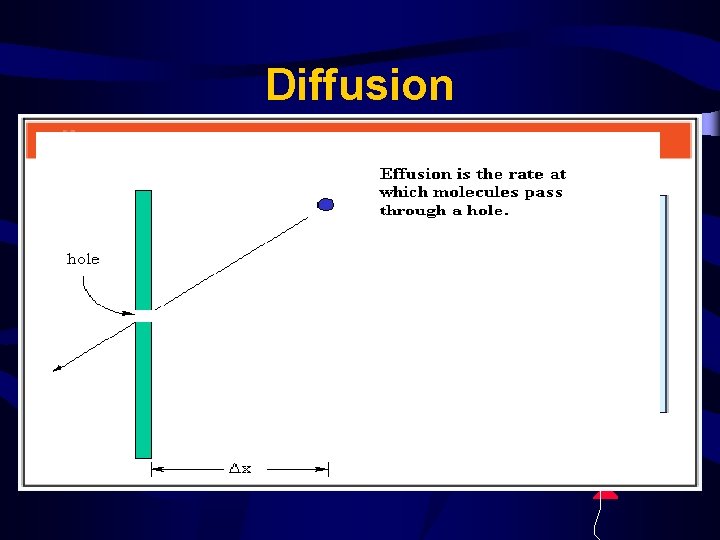
Diffusion • Bigger molecules move slower at the same temp. • Bigger molecules effuse and diffuse slower • Helium effuses and diffuses faster than air -escapes from balloon.

Henry’s Law • The solubility of a gas in a liquid is directly proportional to the pressure of the gas AT CONSTANT TEMP • However, the solubility of a gas in a liquid is decreased with increasing temperature think of leaving an open pop in a warm car vs cold car • S 1/P = S /P 1 2 2 at a constant temperature, this is Henry’s Law • S is solubility, typically given in g/L • How does it relate to Nitrogen narcosis in SCUBA divers? ? ? Remember reading!!!!!!

Kinetic Molecular Theory Three main points to the kinetic theory of gases. • Gases are made of small particles, which are spread very far apart from each other and behave independently of one another. • Gas particles constantly move, randomly, yet in a straight line until acted upon by an outside force or barrier. • All collisions are perfectly elastic which means that no energy is gained or lost during the collision.

Avogadro’s Hypothesis Nitrogen, N 2 Hydrogen, H 2 Oxygen, O 2 1 mole of each gas has the same number of molecules at STP

2
- Slides: 42