MORE GAS LAWS The Combined Gas Laws and

MORE GAS LAWS The Combined Gas Laws and Dalton’s Law of Partial Pressure

� When a weather balloon rises through the atmosphere, often the temperature, volume, and pressure of the contained gas changes. It would be difficult to keep these variables constant. � We need to combine Boyle’s, Charles’ and Gay-Lussac’s Laws.


A firefighter’s air tank contains 12. 0 L of air compressed to 1. 40 x 104 k. Pa at 22 o. C. What volume of air will this tank provide when it is used in a hot, smoke-filled building where the temperature is 42 o. C and the pressure 102 k. Pa?



�This law states that the total pressure of a mixture of non-reacting gases is equal to the sum of the partial pressure of each of the gases in the mixture. Ptotal = P 1 + P 2 + P 3 + …
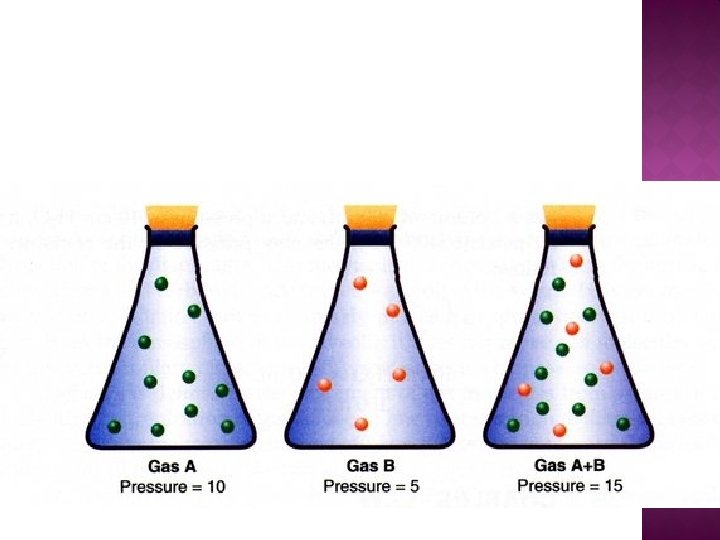

�Nitrogen gas is introduced into a 1. 0 L flask containing oxygen. The pressure in the flask increases from 35 k. Pa to 77 k. Pa. Calculate the partial pressure of each gas in the final mixture.


� Using downward displacement of water technique to collect a gas sample will always contain water vapour as a contaminant in the pure gas. � For this reason, the water vapour must be taken into account when determining the pressure of a gas collected with this method.

�What is the pressure of H 2(g) collected over water at 24. 0 OC? The atmospheric pressure is 94. 4 k. Pa and the pressure of water vapour at 24. 0 OC is given in Table 3 on page 464.

- Slides: 13