What Is Everything Made of If it has















































- Slides: 66

What Is Everything Made of? If it has mass and volume it’s…

Examples of Matter.

What Is Not Matter. • Energy (heat, light, electricity, etc). • Forces (gravity magnetism and the nuclear forces) • Space itself is a 3 D “fabric” but not matter.

Law of Conservation of Mass. • Matter cannot be created nor destroyed, except in a nuclear reaction when it becomes energy E=mc 2. • A burning log does not disappear, collect all the smoke and ashes and the mass is the same.

Mass is a Measure of the Amount of Matter • Mass is measured with a triple beam balance. • Mass is measured in grams (g), a gram is the mass of 1 cm 3 of water and is about the mass of a paperclip • 1000 g = 1 kilogram (kg)

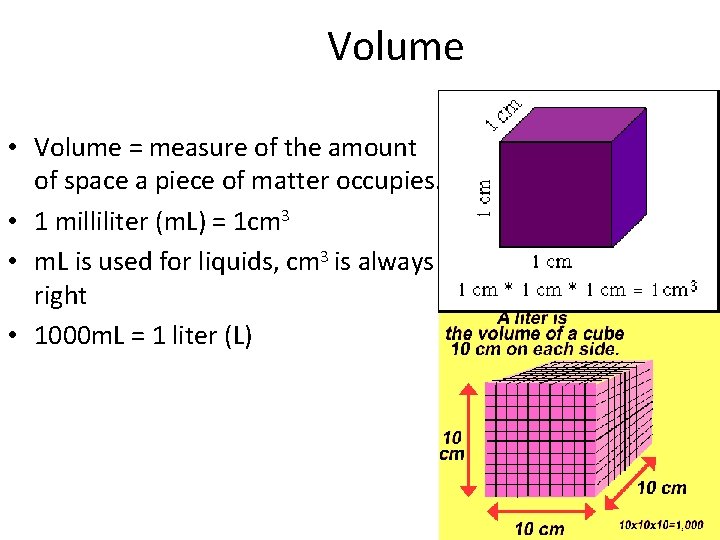
Volume • Volume = measure of the amount of space a piece of matter occupies. • 1 milliliter (m. L) = 1 cm 3 • m. L is used for liquids, cm 3 is always right • 1000 m. L = 1 liter (L)

Measuring Volume • Use a graduated cylinder for liquids. • Measure at the bottom of the meniscus • Use an overflow can for oddly shaped objects

Measuring • Measure to 2 decimal places with a ruler or triple beam balance • Measure to 1 decimal place with a graduated cylinder. • Last number is always a guess. • When calculating, your answer can never have more decimals than your data.

Estimate the volume of each example • 82. 7 m. L • 58. 0 m. L • 52. 2 m. L

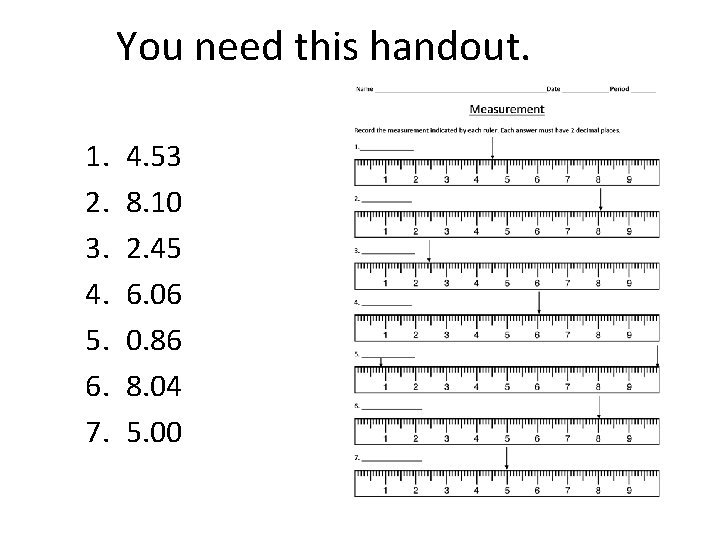
You need this handout. 1. 2. 3. 4. 5. 6. 7. 4. 53 8. 10 2. 45 6. 06 0. 86 8. 04 5. 00

Dihydrogen Monoxide • Tare a container then measure mass every 10 m. L up to 60 m. L. Graph your data • If you are accurate it will form a line, the more accurate you are, the closer your data will be to a line. • When real scientists do an experiment, their hope is their data will form a line or curve. • Draw the line of best fit. Be sure to go through (x=0, y=0)

Mass vs Weight

All Matter has Gravity • The elephant has more matter so it has more mass and more gravity. • Even dust is matter so it has gravity.

The cause of gravity remains mysterious. • It just never gives up. • Einstein described gravity as matter warping the fabric of space. • Quantum physics predicts a particle (graviton) could exist, but one has never been found.

Gravity Decreases With Distance • As a square • So it drops off quickly to next to nothing

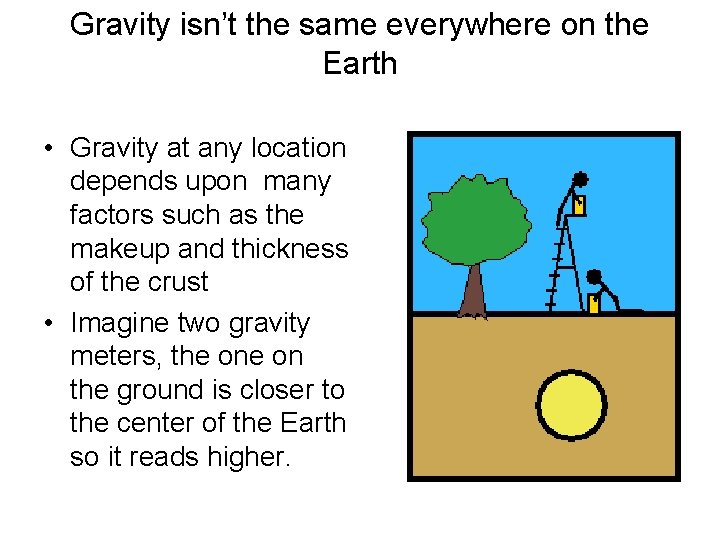
Gravity isn’t the same everywhere on the Earth • Gravity at any location depends upon many factors such as the makeup and thickness of the crust • Imagine two gravity meters, the on the ground is closer to the center of the Earth so it reads higher.

Weight is the Pull of Gravity • Weight is a measure of the pull of gravity. • Weight is measured with a spring that is compressed or stretched.

How can something be nothing? • Far enough from the pull of a planet, an astronaut could become weightless. • The astronaut and the space suit are made of something… • but, according to weight they are nothing, which is impossible!

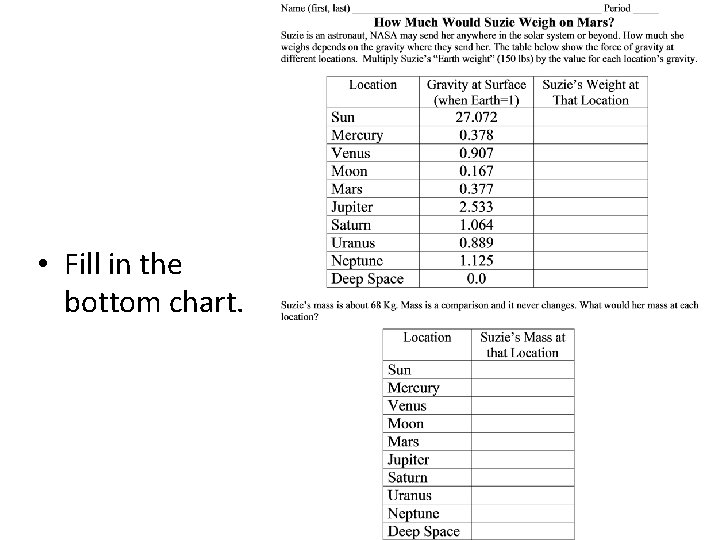
Another Problem • Weight changes when you’re standing on a different planet or on the moon • The Earth’s mass is six times the moons. • On Earth the astronaut weighs 185 lbs. • Moon has 1/6 th the gravity so the astronaut weighs only 31 lbs. • Weight is not about the astronaut, it’s about what object they’re standing on.

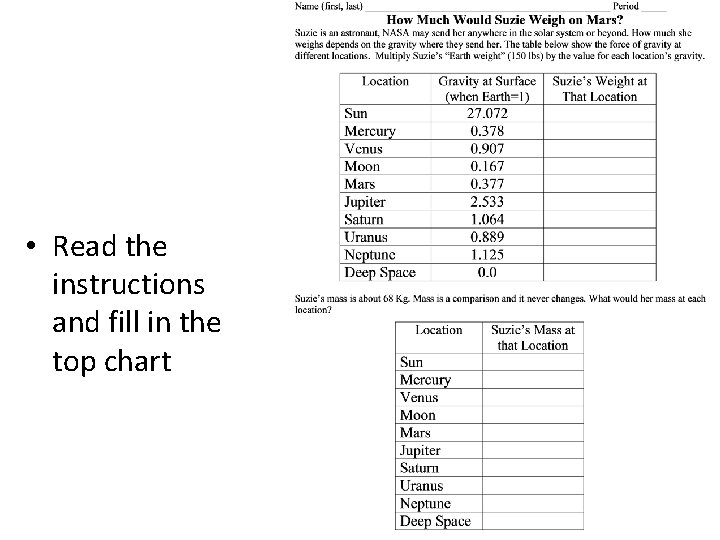
• Read the instructions and fill in the top chart

Weight changes in many situations. When an elevator drops downward. At the top of the roller coaster’s hill. When an airplane flies a parabolic arc. Skiers “unload” (flex downward at the knees) to reduce the effects of weight.

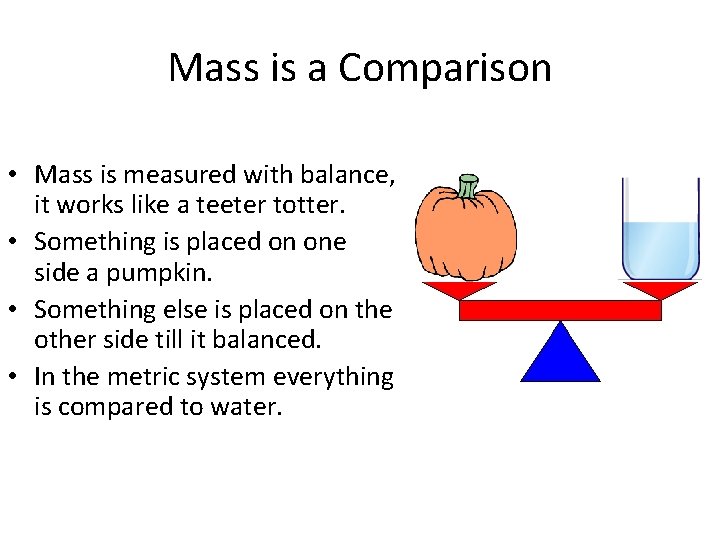
Mass is a Comparison • Mass is measured with balance, it works like a teeter totter. • Something is placed on one side a pumpkin. • Something else is placed on the other side till it balanced. • In the metric system everything is compared to water.

Mass Does Not Change • While on the Earth I will place an object on one side of a balance and an equal amount of water on the other side. • Now I will travel to the moon. • Once on the moon I will repeat the experiment. • Although the moon pulls 1/6 th as hard on the object, it also pulls 1/6 th as hard on the water, so it still balances.

• Fill in the bottom chart.

Measuring Mass in Space • All objects that have mass resist a change in motion (inertia) • The more mass the more it resists moving. • The space station uses a chair, that the astronaut must rock back and forth

Volume of Rectangular Objects • Measure length, width and height in centimeters (cm) • Multiply length x width x height • Example: A block is 10 cm long, 2 cm wide and 3 cm tall. • Its volume is 10 cm x 2 cm x 3 cm = 60 cm 3

D = m/v • Denser things sink. • The density of pure water is 1. 0 g/cm 3 by definition. • Anything denser sinks; anything less dense floats. • Saltwater is denser than fresh. • Density can be used to identify substances.

Example: An object has a mass of 200. 00 g and a volume of 340. 00 cm 3. What is its density and will it sink or will it float? • • D=m/v D= 200. 00 g/340. 0 cm 3 D=0. 6 g/cm 3 It will float because it’s density is less than 1. 0 g/cm 3

Significant digits • When you do operations (add, subtract, multiply, divide) your answer can only have as many significant digits as your weakest data • Numbers 1 -9 are always significant • Zeros between numbers are always significant, but for zeros before

Examples

Example 2: A graduated cylinder starts with 45. 12 m. L of water. An object with a mass of 12. 30 g is lowered into the water. The water in the cylinder goes up to 56. 78 m. L. What is the density of the object? • • • Volume = 56. 78 m. L - 45. 12 m. L Volume = 11. 66 ml D= m/v D= 12. 30 g/11. 65 cm 3 D=1. 16 g/cm 3 (we only get to keep 1 decimal)

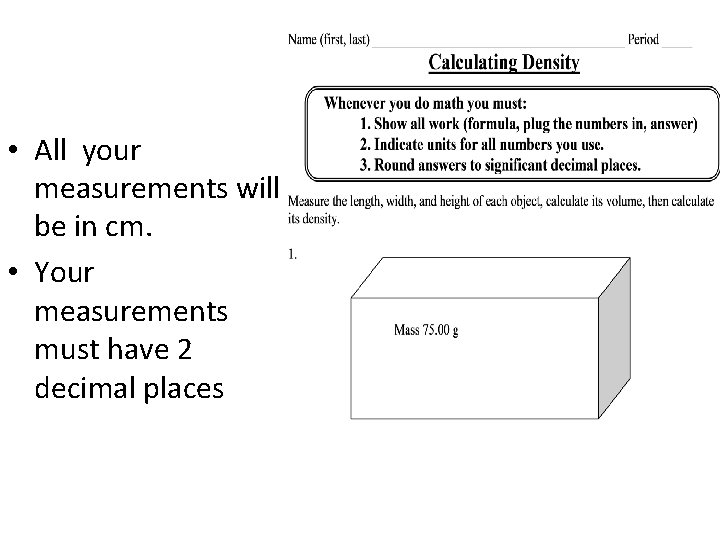
• All your measurements will be in cm. • Your measurements must have 2 decimal places

What is density? • Density is a comparison of how much matter there is in a certain amount of space. • How big something is compared to how heavy it is.

Which one is more dense?

Which one is more dense?

Density explains why: • hot air balloons take off before dawn. • very light sand grains sink, while very heavy aircraft carriers float. • the Earth has layers, and why land rises and sinks. • we can separate things like crude oil into its parts (gasoline, diesel, etc. ).

Liquid Layers • If you pour together liquids that don’t mix and have different densities, they will form liquid layers. • The liquid with the highest density will be on the bottom. • The liquid with the lowest density will be on the top.

Liquid Layers • Imagine that the liquids have the following densities: – 10 g/cm 3. 3 g/cm 3. – 6 g/cm 3. 5 g/cm 3. • Which number would go with which layer?

Liquid Layers • Imagine that the liquids on the right have the following densities: – 15 g/cm 3 – 3 g/cm 3 – 7 g/cm 3 10 g/cm 3 9 g/cm 3 12 g/cm 3 • Match the colors to the correct densities. 3 g/cm 3 7 g/cm 3 9 g/cm 3 10 g/cm 3 12 g/cm 3 15 g/cm 3

Review • What is the formula for density? • What happens if you pour together liquids that have different densities? • Will the liquid on the top have the highest or lowest density? • Will the liquid on the bottom have the highest or lowest density?

Air Pressure/Density

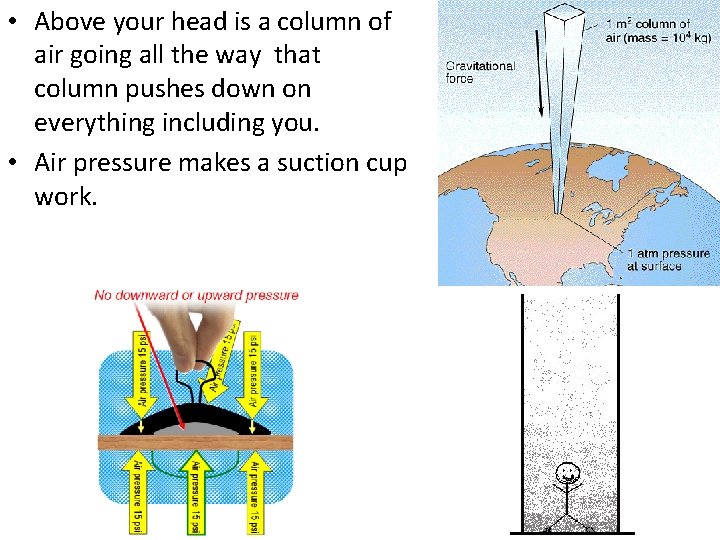
• Above your head is a column of air going all the way that column pushes down on everything including you. • Air pressure makes a suction cup work.

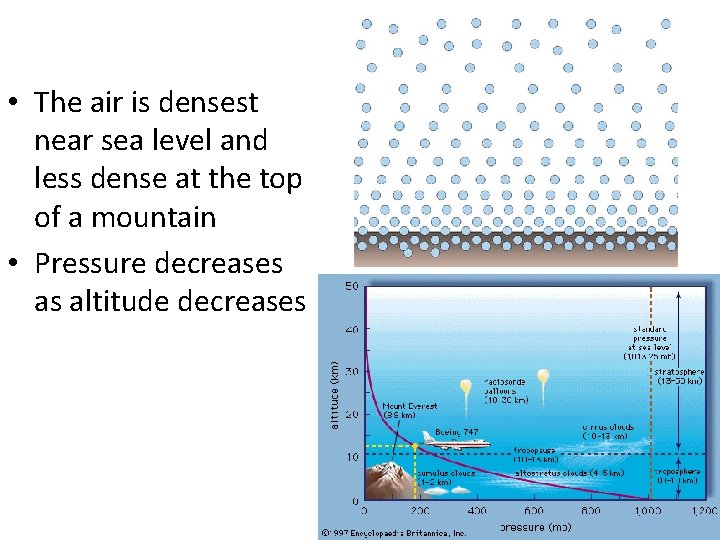
• The air is densest near sea level and less dense at the top of a mountain • Pressure decreases as altitude decreases

Air pressure affects the boiling of water • Air pressure keeps water in a container, the movement of the molecules tries to. • At sea level, the denser air pushes harder on the water so it has to reach a higher temperature to boil.

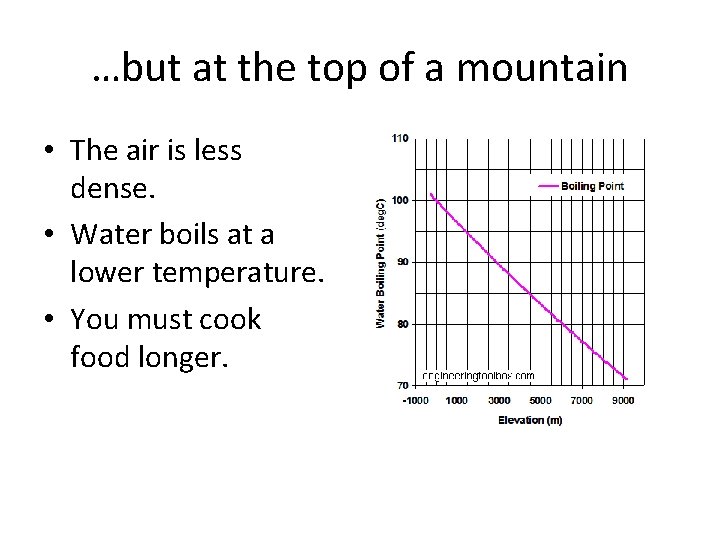
…but at the top of a mountain • The air is less dense. • Water boils at a lower temperature. • You must cook food longer.

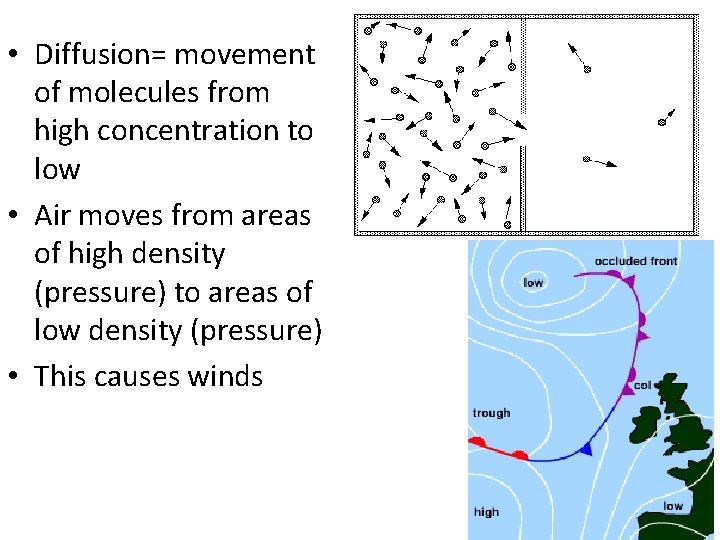
• Diffusion= movement of molecules from high concentration to low • Air moves from areas of high density (pressure) to areas of low density (pressure) • This causes winds

Matter Flashcards • m. L = unit of liquid volume • 1. 0 g/cm 3 = density of fresh water • > 1. 0 g/cm 3 = sinks in fresh water • < 1. 0 g/cm 3 = floats in fresh water • 1 g = mass of 1 cm 3 of fresh water • L x W X H = formula for volume of a rectangular solid • cm 3 = unit of solid volume

Solid, Liquid, Gas (a) Particles in solid (b) Particles in liquid (c) Particles in gas

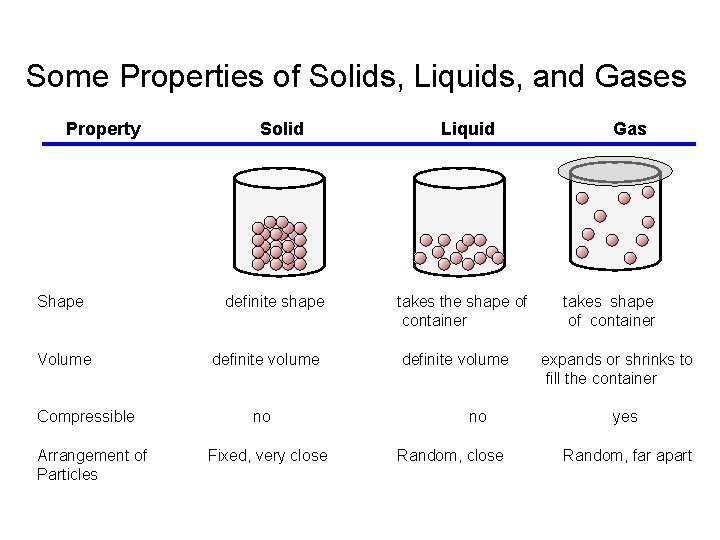
Some Properties of Solids, Liquids, and Gases Property Shape Volume Compressible Arrangement of Particles Solid definite shape definite volume no Fixed, very close Liquid takes the shape of container definite volume no Random, close Gas takes shape of container expands or shrinks to fill the container yes Random, far apart

Plasma, the 4 th State of Matter • Energy is so high, atoms vibrate themselves into + and – charged pieces (ions) • Fluorescent lights • Plasma TV’s • The sun • Plasma torches and cutters

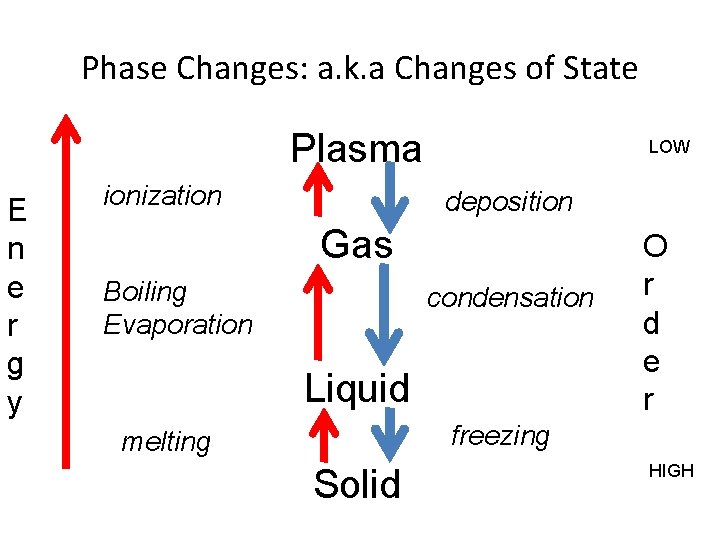
Phase Changes: a. k. a Changes of State Plasma E n e r g y ionization LOW deposition Gas Boiling Evaporation condensation Liquid O r d e r freezing melting Solid HIGH

Sublimation • When a solid goes directly to a gas without ever becoming a liquid. • Dry ice (solid CO 2) • Food in the freezer (freezer burn)

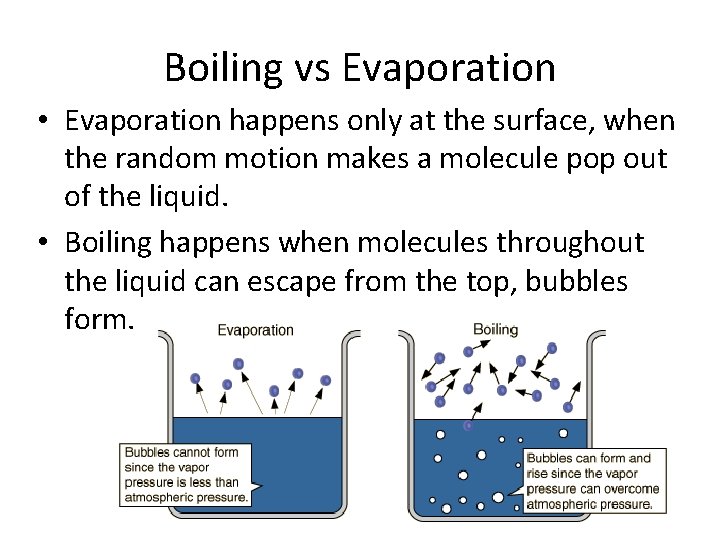
Boiling vs Evaporation • Evaporation happens only at the surface, when the random motion makes a molecule pop out of the liquid. • Boiling happens when molecules throughout the liquid can escape from the top, bubbles form.

Crystalline Solids • Crystals = solids with a regular repeating lattice pattern • Diamonds and graphite are made of the same stuff, the difference is their patterns

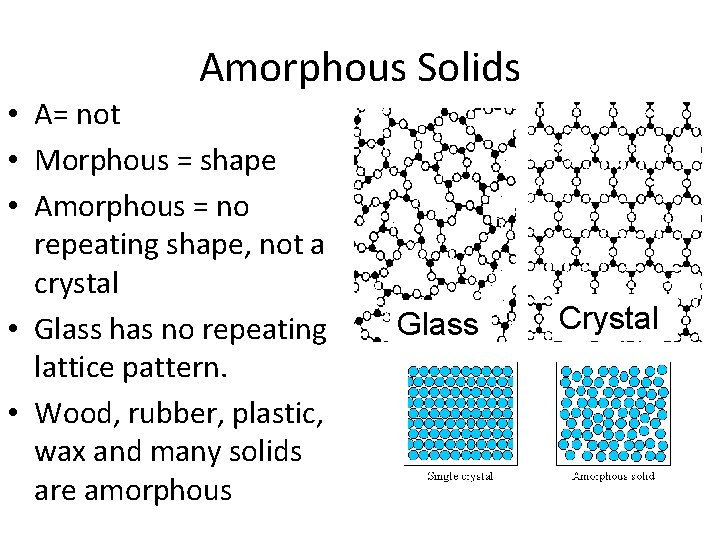
Amorphous Solids • A= not • Morphous = shape • Amorphous = no repeating shape, not a crystal • Glass has no repeating lattice pattern. • Wood, rubber, plastic, wax and many solids are amorphous Glass Crystal

Melting and Boiling Points • Every substance has a melting and boiling point that can be used to identify it. • Being frozen does not mean cold, tungsten stays frozen until it 5, 555 o. C • Boiling does not mean hot, helium boils at -268 o. C.

Example: Nitrogen • Element # 7 on the Periodic Chart • Pure nitrogen atoms travel as twins that are almost impossible to separate. • Only microbes (bacteria) can eat nitrogen from the air • Your body needs nitrogen to make DNA, and muscles (among other things)

The air we breathe is mostly nitrogen. • 78%N 2 • 21% O 2 • 1% other elements

Uses for Nitrogen • • Fertilizer Explosives Car tires, nitrous Freezing things

Properties of Nitrogen • • Colorless Odorless Will not burn MELTING POINT: − 210°C BOILING POINT: − 196°C DENSITY : 0. 0012506 g/cm 3

Lauric Acid • Main acid in coconut oil and palm kernel oil. • Antimicrobial • White, powdery solid with faint odor of bay oil or soap. • Used in many soaps and shampoos.

You will need this handout. • Record the temperature every 30 seconds. • Write an observation every thirty seconds

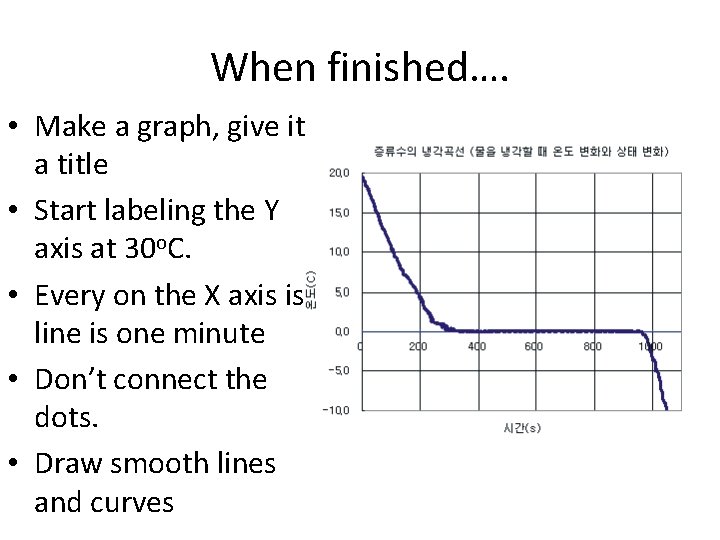
When finished…. • Make a graph, give it a title • Start labeling the Y axis at 30 o. C. • Every on the X axis is line is one minute • Don’t connect the dots. • Draw smooth lines and curves

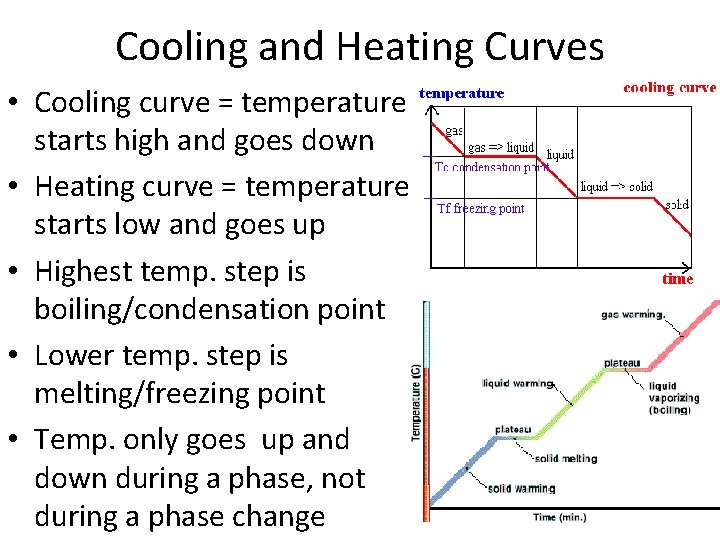
Cooling and Heating Curves • Cooling curve = temperature starts high and goes down • Heating curve = temperature starts low and goes up • Highest temp. step is boiling/condensation point • Lower temp. step is melting/freezing point • Temp. only goes up and down during a phase, not during a phase change

• What is the freezing point? • What is the melting point? • What is the boiling point? • Is the order increasing or decreasing? • Could this substance be water? • When is it a solid? • A liquid? • A gas? Example

Russia