The Empirical Gas Laws The Empirical Gas Laws












- Slides: 17

The Empirical Gas Laws

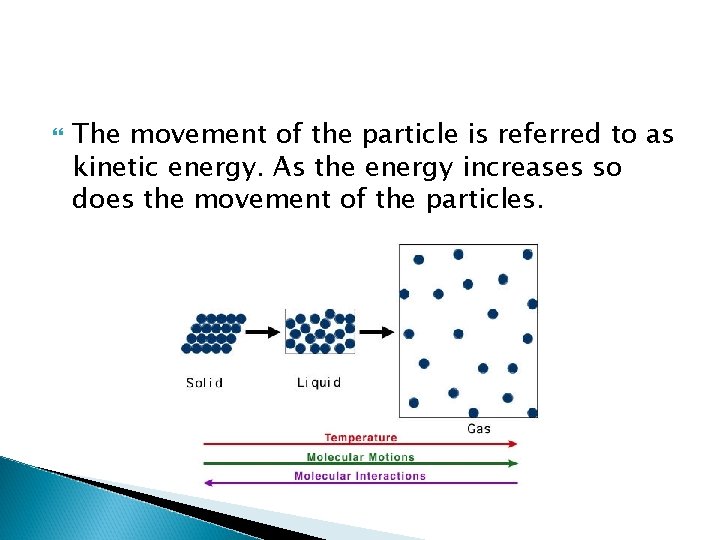
The Empirical Gas Laws: Pressure Kinetic Molecular Theory states that all substances are made of tiny particles that are in constant motion. Gases have the most amount of movement in the three states of matter. The movement of the particles is independent of each other as each particle travels in a straight line until it bumps into another particle or wall of its container.

The movement of the particle is referred to as kinetic energy. As the energy increases so does the movement of the particles.

Quantitative measurements on gases were first made by the English chemist Robert Boyle (1627 - 1691). Boyle used two instruments to measure pressure: the manometer, which measures differences in pressure, and the barometer, which measures the total pressure of the atmosphere

Units of Pressure Units of pressure were originally all based on the length of the column of liquid, usually mercury, supported in a manometer or barometer. By far the most common of these units was the mm Hg.

Units of Pressure However, the modern SI unit of pressure is derived from the fundamental units of the SI. Pressure is force per unit area, and force is the product of mass times acceleration, so the SI unit of pressure is the kg m s 2/m 2 or newton/m 2, which is called the pascal (Pa).

Units of Pressure The standard temperature and pressure that gas experiments are done at is referred to as SATP. Standard Ambient Temperature and Pressure is 25° C and 100 k. Pa.

Units of Pressure All of the older units of pressure have now been redefined in terms of the pascal. One standard atmosphere, the pressure of the atmosphere at sea level, is by definition exactly 101, 325 Pa. The torr, named in honour of Torricelli, is defined as 1/760 of a standard atmosphere or as 101, 325/760 Pa. The mm. Hg can be considered identical to the torr. The term bar is used for 100, 000 Pa, which is slightly below one standard atmosphere.

Boyle’s Law

Boyle’s Law Boyle used the manometer and barometer to study the pressures and volumes of different samples of different gases. The results of his studies can be summarized in a simple statement which has come to be known as Boyle's law: At any constant temperature, the product of the pressure and the volume of any size sample of any gas is a constant.

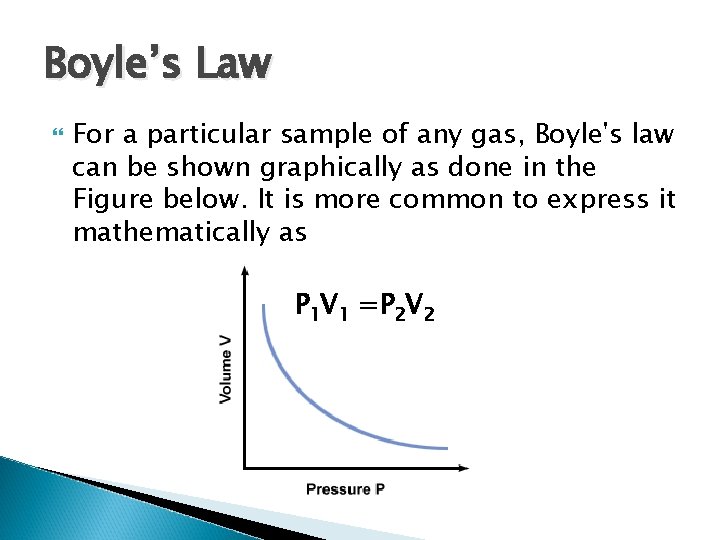
Boyle’s Law For a particular sample of any gas, Boyle's law can be shown graphically as done in the Figure below. It is more common to express it mathematically as P 1 V 1 =P 2 V 2

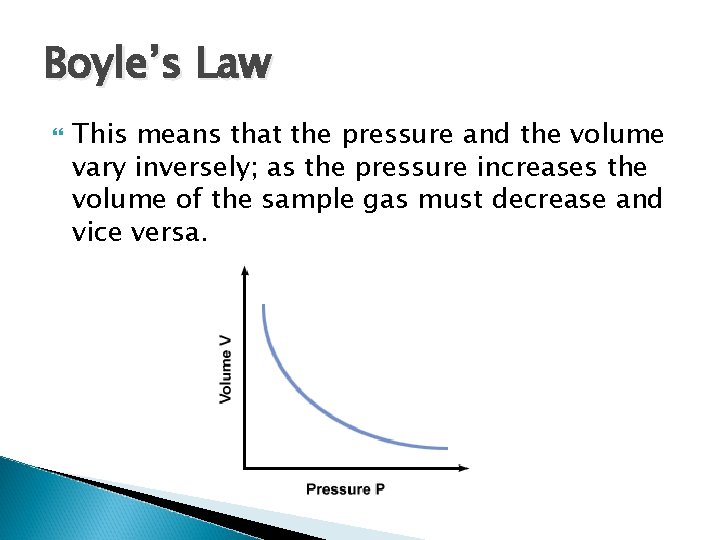
Boyle’s Law This means that the pressure and the volume vary inversely; as the pressure increases the volume of the sample gas must decrease and vice versa.

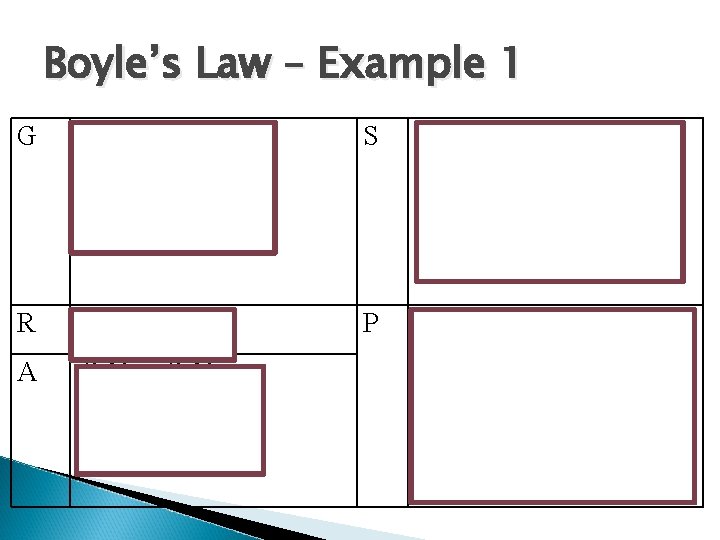
Boyle’s Law – Example 1 A sample of gas occupies a volume of 47. 3 cm 3 at 25°C with a pressure of 30 mm of mercury. If the pressure is increased to 75 mm of mercury, what will the volume be at the new pressure?

Boyle’s Law – Example 1 G P 1 = 30 mm Hg V 1 = 47. 3 cm 3 P 2 = 75 mm Hg S V 2 = (30 cm x 47. 3 cm ) 75 cm Hg 3 V 2 = 18. 9 cm 3 R V 2 = ? A P 1 V 1 = P 2 V 2 = P 1 V 1 / P 2 P Therefore, When the pressure increased from 47. 3 mm Hg to 75 mm Hg the volume decreased from 47. 5 cm 3 to 18. 9 cm 3

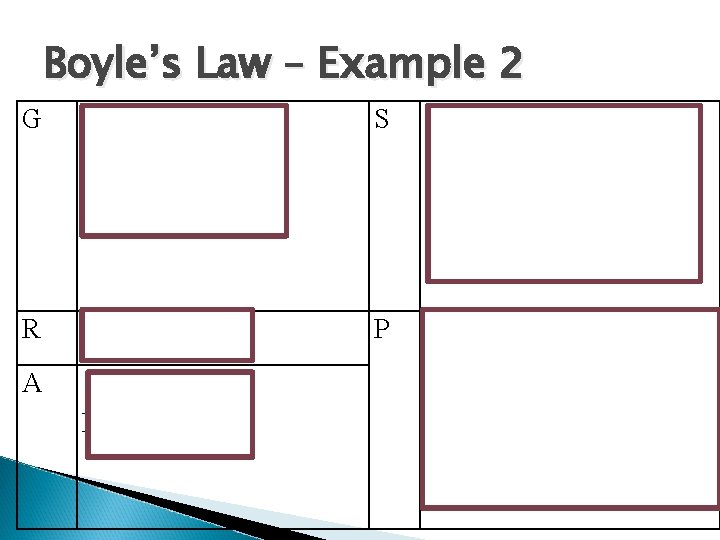
Boyle’s Law – Example 2 A student is trying to compress the gas in a cylinder that has an initial volume of 1000 cm 3 and a pressure of 100 k. Pa. The student decreases the volume by 500 cm 3. What is the pressure of the gas in the cylinder?

Boyle’s Law – Example 2 G P 1 = 100 k. Pa V 1 = 1000 cm 3 V 2 = 500 cm 3 S P 2 = (100 k. Pa x 1, 000 cm 3) 500 cm 3 P 2 = 200 k. Pa R P 2 = ? P A P 1 V 1 = P 2 V 2 P 2= P 1 V 1 / V 2 Therefore, When the student compressed the cylinder to half of its original volume the pressure doubled to 200 k. Pa.

Questions: page 425 # 1 – 4, page 428# 5 -9.