Chapter 18 The Combined Gas Law Grahams Law

Chapter 18 The Combined Gas Law & Graham’s Law

Combined Gas Law • 10. 0 cm 3 volume of a gas is collected at 75. 6 k. Pa and 60. 0 C. What would its volume be at STP? 6. 12 cm 3

Combined Gas Law Problem A sample of helium gas has a volume of 0. 180 L, a pressure of 0. 800 atm and a temperature of 29°C. What is the new temperature(°C) of the gas at a volume of 90. 0 m. L and a pressure of 3. 20 atm?

Combined Gas Law Problem A sample of helium gas has a volume of 0. 180 L, a pressure of 0. 800 atm and a temperature of 29°C. What is the new temperature(°C) of the gas at a volume of 90. 0 m. L and a pressure of 3. 20 atm? 302 K x 3. 20 atm 0. 800 atm x 90. 0 m. L = 604 K 180. 0 m. L 604 K - 273 = 331 °C

Weather Balloons

Weather Balloons • A weather balloon is an instrument which measures temperature, humidity, and atmospheric pressure. The weather balloon is generally a balloon mounted with weather measuring equipment and a radio transmitter. Some weather balloons are equipped with GPS or global positioning systems for the weather station to track it more accurately. • In North America prior to release the balloon is usually filled with hydrogen gas due to lower cost, though helium can be used as a substitute. Weather balloons may reach altitudes of 40 km (25 miles) or more where the diminishing pressure eventually causes the balloon to expand to such a degree that it pops. • Weather balloons are sometimes cited as the cause for unidentified flying object sightings.

A weather balloon with a volume of 305 L at STP is released and rises until it expands and explodes at a pressure of 7. 00 torr and a temperature of – 21°C. What was the volume of the balloon when it burst? ≈ 30, 600 L

A weather balloon with a volume of 305 L at STP is released and rises until it expands and explodes at a pressure of 7. 00 torr and a temperature of – 21°C. How many times larger is the balloon than when it was released? About 100 times larger

Diffusion • The spreading of a gas from an area of higher concentration to an area of lower concentration.
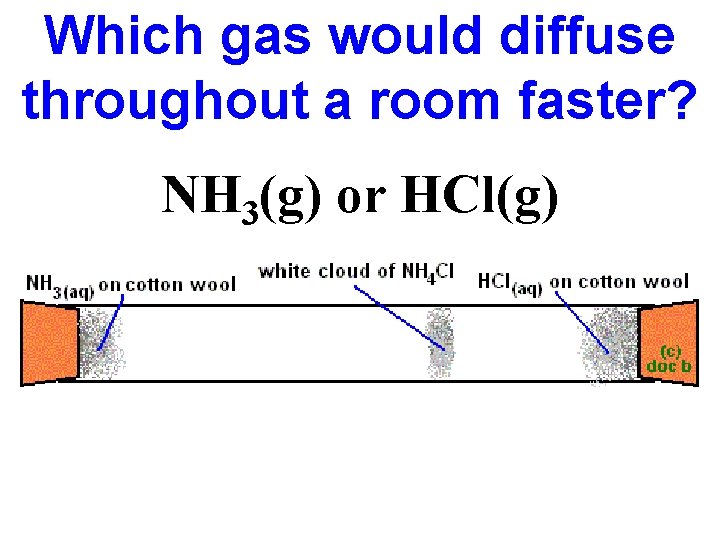
Which gas would diffuse throughout a room faster? NH 3(g) or HCl(g)


Gas Diffusion and Effusion • Graham's Law: • Allows for the calculation of the rate of diffusion of gas molecules.

Gas Diffusion and Effusion Graham's Law: governs the rate of diffusion of gas molecules.

Calculate the rate of diffusion of NH 3(g) compared to HCl(g) NH 3 diffuses 1. 47 times faster than HCl

Use measurement to confirm the rate of diffusion of NH 3(g) compared to HCl(g) HCl NH 3

Calculate the rate of diffusion of helium compared to argon. Helium diffuses 3. 2 times faster than argon

Calculate the rate of diffusion of hydrogen compared to neon. hydrogen diffuses 3. 2 times faster than neon

Homework (All due in 2 days) • Summarize the (I) Purpose and (II) Procedure of the “Gas Laws” Lab. • Study Guide Chapter 18. • Worksheet: Combined Gas Law & Graham’s Law.
- Slides: 18