The Ideal Gas Law PV n RT Adds
























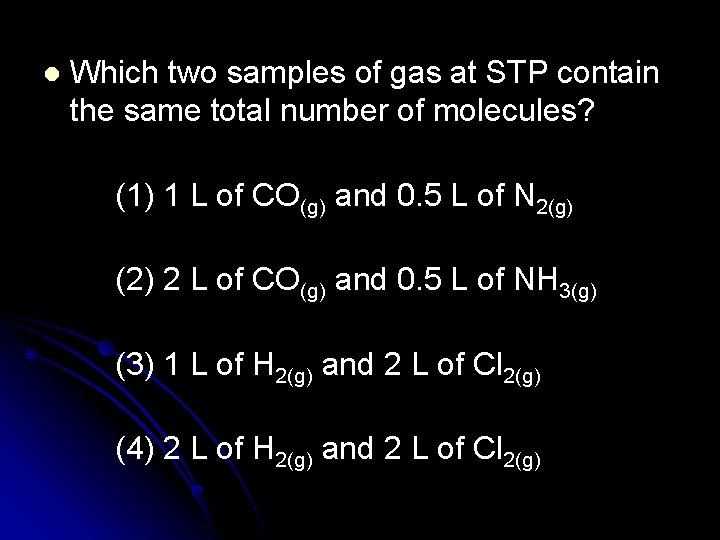







- Slides: 37

The Ideal Gas Law PV = n. RT Adds in the factor of number of moles of gas “n”. P = Pressure (atm) V = Volume (Liters) T = Temperature (Kelvin) n = number of moles Remember when dealing with moles: # moles (n) = grams of substance Gram Formula Mass

What is “R”? R is a constant, called the “Universal Gas Constant” It is derived by plugging in the values for 1 mole of gas at STP. (22. 4 Liters, 273 K, 1 atm) L • atm R = 0. 0821 Mol • K l Instead of learning a different value for R for all the possible unit combinations, we can just memorize one value and convert the units to match R.

Ideal Gas Law (Honors) l Calculate the pressure in atmospheres of 0. 412 mol of He at 16°C & occupying 3. 25 L. IDEAL GAS LAW GIVEN: WORK: P = ? atm n = 0. 412 mol T = 16°C = 289 K V = 3. 25 L R = 0. 0821 L atm/mol K PV = n. RT P(3. 25)=(0. 412)(0. 0821)(289) L mol L atm/mol K K K P = 3. 01 atm

Example l # 1 Honors Packet l A 9. 81 L cylinder contains 23. 5 moles of nitrogen at 23° C. What pressure is exerted by the gas?

Example l # 2 Honors Packet l A pressure of 850 mm. Hg is exerted by 28. 6 grams of sulfur dioxide at a temp of 40 °C. Calculate the volume of the vessel holding the gas.

Density is Hidden in this Formula l Density = Mass (g) Volume # moles = Mass (g) Gram formula mass Can you rework PV = n. RT to solve for Density?

Example l # 7 in Honors Packet l What is the density of neon at 40 °C and 1. 23 atmospheres?

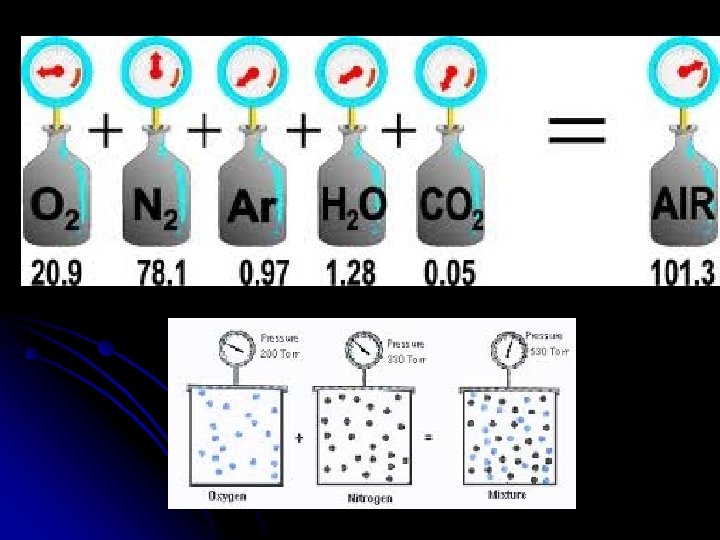
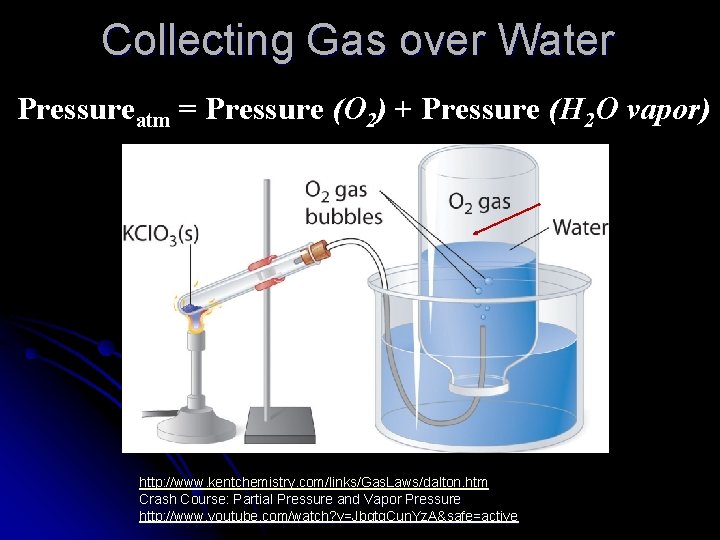
Dalton’s Law of Partial Pressures Ptotal = P 1+P 2+…. l Total pressure of a mixture of gases in a container is the sum of the individual pressures (partial pressures) of each gas, as if each took up the total space alone. l This is often useful when gases are collected “over water”


Collecting Gas over Water Pressureatm = Pressure (O 2) + Pressure (H 2 O vapor) http: //www. kentchemistry. com/links/Gas. Laws/dalton. htm Crash Course: Partial Pressure and Vapor Pressure http: //www. youtube. com/watch? v=Jbqtq. Cun. Yz. A&safe=active

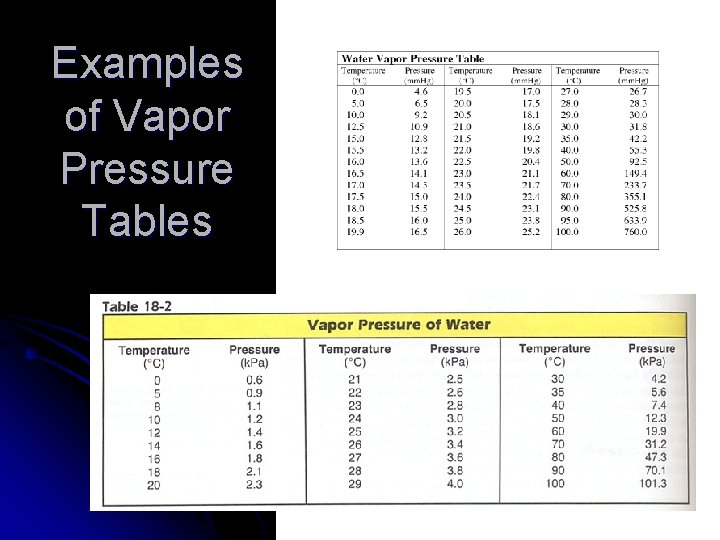
Dalton’s Law l GIVEN: PH 2 = ? Ptotal = 94. 4 k. Pa PH 2 O = 2. 72 k. Pa Look up water-vapor pressure on for 22. 5°C. H 2 gas is collected over water at 22. 5°C. Find the pressure of the dry gas if the atmospheric pressure is 94. 4 k. Pa. WORK: Ptotal = PH 2 + PH 2 O 94. 4 k. Pa = PH 2 + 2. 72 k. Pa PH 2 = 91. 7 k. Pa

Dalton’s Law l A gas is collected over water at a temp of 35. 0°C when the barometric pressure is 742. 0 torr. What is the partial pressure of the dry gas? DALTON’S LAW GIVEN: Pgas = ? Ptotal = 742. 0 torr PH 2 O = 42. 2 torr Look up water-vapor pressure for 35. 0°C. WORK: Ptotal = Pgas + PH 2 O 742. 0 torr = PH 2 + 42. 2 torr Pgas = 699. 8 torr

Examples of Vapor Pressure Tables

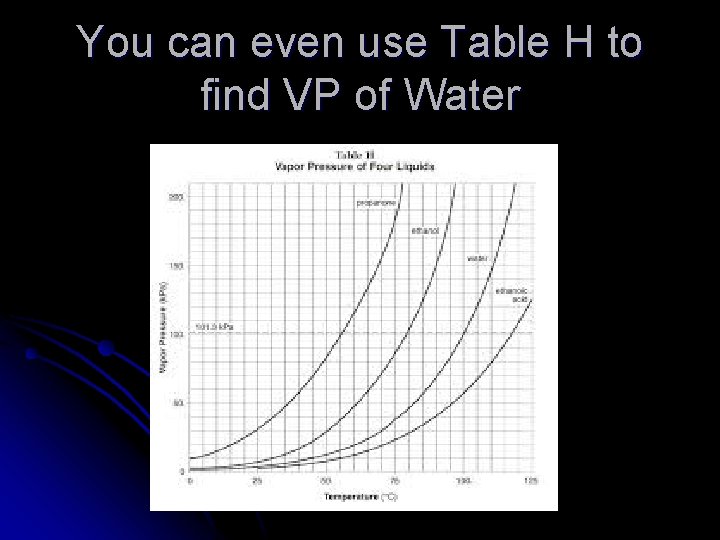
You can even use Table H to find VP of Water

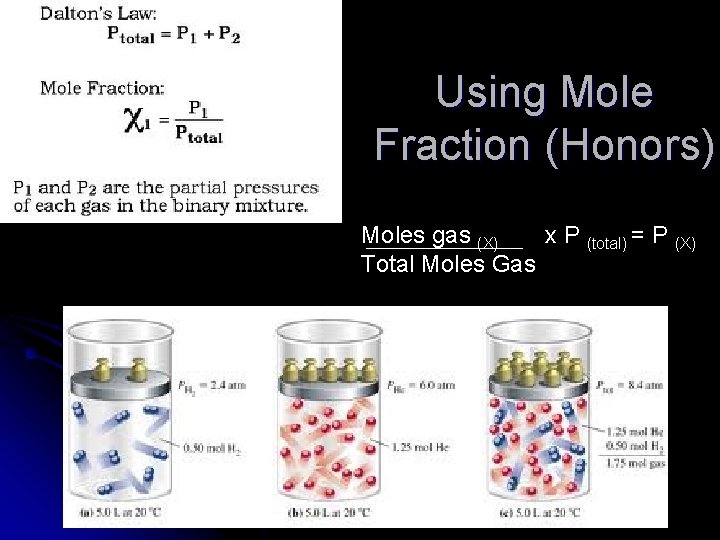
Using Mole Fraction (Honors) Moles gas (X) x P (total) = P (X) Total Moles Gas

Example l # 4 Honors Packet l A mixture of 2. 00 moles H 2, 3. 00 moles NH 3, 4. 00 moles CO 2, and 5. 00 moles N 2 exert a total pressure of 800 mm. Hg. What is the partial pressure of each gas?

Graham’s Law l Diffusion l Spreading of gas molecules throughout a container until evenly distributed. l Effusion l Passing of gas molecules through a tiny opening in a container Smaller and lighter gas particles do this faster! https: //www. youtube. com/watch? feature=player_embedded&v=H 7 Qs. Ds 8 ZRMI https: //www. youtube. com/watch? feature=player_embedded&v=L 41 Kh. BPBym. A l Crash Course: Grahams Law l http: //www. youtube. com/watch? v=TLRZAFU_9 Kg&safe=active

Graham’s Law l Speed of diffusion/effusion l At the same temp & KE, heavier molecules move more slowly. l Ex: Which of the following gases will diffuse most rapidly? A. ) N 2 B. ) CO 2 C. ) CH 4

Graham’s Law Formula l Graham’s Law l Rate of diffusion of a gas is inversely related to the square root of its molar mass. Ratio of gas A’s speed to gas B’s speed

Graham’s Law l Determine the relative rate of diffusion for krypton and bromine. The first gas is “Gas A” and the second gas is “Gas B”. Relative rate mean find the ratio “v. A/v. B”. Kr diffuses 1. 381 times faster than Br 2.

Graham’s Law l A molecule of oxygen gas has an average speed of 12. 3 m/s at a given temp and pressure. What is the average speed of hydrogen molecules at the same conditions? Put the gas with the unknown speed as “Gas A”.

Graham’s Law l An unknown gas diffuses 4. 0 times faster than O 2. Find its molar mass. The first gas is “Gas A” and the second gas is “Gas B”. The ratio “v. A/v. B” is 4. 0. Square both sides to get rid of the square root sign.

Example l # 1 Honors Packet l Under the same conditions of temp and pressure, how many times faster will hydrogen effuse compared to carbon dioxide?

Example l #7 Honors Packet l If CO 2 diffuses from a flask in 30 seconds and an unknown gas diffuses from the same flask in 5 minutes, calculate the molecular weight of this unknown gas.

Avogadro’s Principle l Volume of a gas is directly proportional to the number of moles of gas particles present. V n

l Equal volumes of gases contain equal numbers of moles of particles at constant temp & pressure l true for any gas l

Avogadros Law: https: //www. youtube. com/watch? feature=player_embedded&v=fex. Evn 0 ZOpo Molar Volume of a Gas: https: //www. youtube. com/watch? feature=player_embedded&v=4 b 852 VIEk. HQ

Practice Questions l A. At the same temperature and pressure, 1. 0 liter of CO(g) and 1. 0 liter of CO 2(g) have equal masses and the same number of molecules B. different masses and a different number of molecules C. equal volumes and the same number of molecules D. different volumes and a different number of molecules

l Which rigid cylinder contains the same number of gas molecules at STP as a 2. 0 liter rigid cylinder containing H 2(g) at STP? (1) 1. 0 -L cylinder of O 2(g) (2) 2. 0 -L cylinder of CH 4(g) (3) 1. 5 -L cylinder of NH 3(g) (4) 4. 0 -L cylinder of He(g
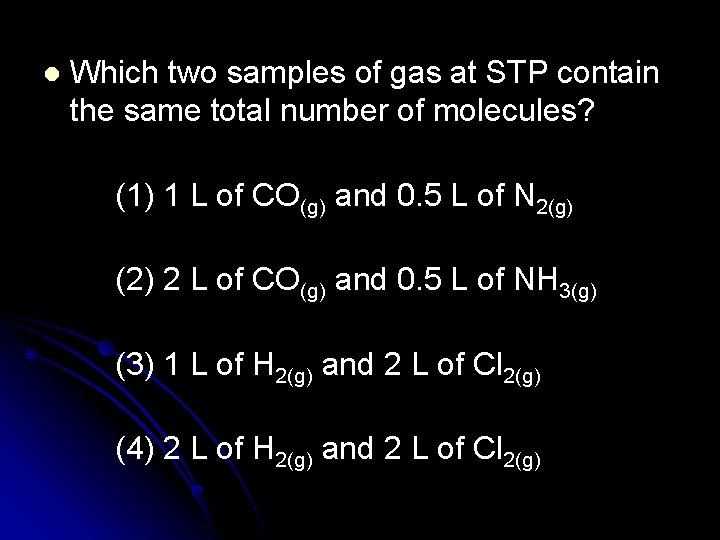
l Which two samples of gas at STP contain the same total number of molecules? (1) 1 L of CO(g) and 0. 5 L of N 2(g) (2) 2 L of CO(g) and 0. 5 L of NH 3(g) (3) 1 L of H 2(g) and 2 L of Cl 2(g) (4) 2 L of H 2(g) and 2 L of Cl 2(g)

Gas Stoichiometry l Moles Liters of a Gas STP - use 22. 4 L/mol l Non-STP - use ideal gas law l l Non-STP Problems l Given liters of gas? l l start with ideal gas law Looking for liters of gas? l start with stoichiometry conv.

Gas Stoichiometry Problem l What volume of CO 2 forms from 5. 25 g of Ca. CO 3 at 1. 017 atm & 25ºC? Ca. CO 3 Ca. O + CO 2 5. 25 g ? L Looking for liters: Start with stoich nonand calculate moles of CO. STP 5. 25 g 1 mol Ca. CO 3 CO 2 = 1. 26 mol 100. 09 1 mol Plug this into the Ideal CO 2 g Ca. C Gas Law to find liters. 2

Gas Stoichiometry Problem l What volume of CO 2 forms from 5. 25 g of Ca. CO 3 at 1. 017 atm & 25ºC? GIVEN: WORK: P = 1. 017 atm n = 1. 26 mol T = 25°C = 298 K R =. 0821 L atm/mol K PV = n. RT (1. 017 atm)V =(1 mol)(. 0821 L atm/mol K)(298 K) V = 1. 26 L CO 2

Gas Stoichiometry Problem l How many grams of Al 2 O 3 are formed from 15. 0 L of O 2 at 0. 96 atm & 21°C? 4 Al + 3 O 2 2 Al 2 O 3 15. 0 L ? g non. GIVEN: P = 0. 96 atm STP WORK: Given liters: Start with Ideal Gas Law and calculate moles of O 2. PV = n. RT V = 15. 0 L (0. 96 atm ) (15. 0 L) = n (. 0821 L atm/mol K) n = ? NEXT T = 21°C = 294 K (294 K) R =. 0821 L atm/mol K

Gas Stoichiometry Problem l How many grams of Al 2 O 3 are formed from 15. 0 L of O 2 at 0. 96 atm & 21°C? 4 Al + 3 O 2 2 Al 2 O 3 ? g Use stoich to convert moles 15. 0 L of O to grams Al O. non 0. 597 2 mol STP 101. 96 mol O 2 Al 2 O 3 g Al 2 O 3 = 40. 6 g 3 mol O 2 1 mol Al 2 O 3 2 2 3

Example l # 10 Honors Multiple Choice Ni(CO)4 (l) → Ni (s) + 4 CO (g) l What volume of CO is formed from the complete decomposition of 444 g of Ni(CO)4 at 752 torr and 22. 0 °C l Do stoich for 444 g to find moles of CO. l Plug the moles into Ideal Gas Law to get V at non standard conditions.

Some Cool Videos l l l Crash Course: Ideal Gas Laws http: //www. youtube. com/watch? v=Bx. US 1 K 7 xu 30&safe=active Crash Course: Ideal Gas Law Problems http: //www. youtube. com/watch? v=8 SRAk. XMu 3 d 0 Crash Course: Real Gases http: //www. youtube. com/watch? v=GIPrs. Wu. Sk. Qc&safe=active