NOTES 14 2 14 3 Combined and Ideal
NOTES: 14. 2 - 14. 3 (Combined and Ideal Gas Laws)

Pressure-Volume-Temperature Relationship: Combined Gas Law • Pressure and volume are inversely proportional • Both pressure and volume are directly proportional to temperature

COMBINED GAS LAW: Combining Boyle’s law (pressure-volume) with Charles’ Law (volume-temp): This is for a gas undergoing changing conditions of temp, pressure, and volume.

Combined Gas Law Example #1: The volume of a gas filled balloon is 30. 0 L at 40. 0 o. C and 1148 mm Hg of pressure. What volume will the balloon have at STP? § § § P 1 = 1148 mm Hg V 1 = 30. 0 L T 1 = 40. 0°C = 313 K P 1 V 1 T 1 § § § = P 2 = 1 atm = 760 mm Hg V 2 = ? T 2 = 0°C = 273 K P 2 V 2 T 2

Combined Gas Law Example #1: The volume of a gas filled balloon is 30. 0 L at 40. 0 o. C and 1148 mm Hg of pressure. What volume will the balloon have at STP? § § § P 1 = 1148 mm Hg V 1 = 30. 0 L T 1 = 40. 0°C = 313 K § § § P 2 = 1 atm = 760 mm Hg V 2 = ? T 2 = 0°C = 273 K P 1 V 1 T 2 = P 2 V 2 T 1 (1148 mm Hg)(30. 0 L)(273 K) = (760 mm Hg)(V 2)(313 K) V 2= 39. 5 L

Example #2: A sample of neon gas occupies 105 L at 27. 0°C under a pressure of 985 torr. What volume would it occupy at standard conditions? § § § P 1 = 985 torr V 1 = 105 L T 1 = 27. 0°C = 300 K § § § P 2 = 1 atm = 760 torr V 2 = ? T 2 = 0°C = 273 K P 1 V 1 T 2 = P 2 V 2 T 1 (985 torr)(105 L)(273 K) = (760 torr)(V 2)(300 K) V 2= 124 L

Example #3: A sample of gas occupies 10. 0 L at 240. °C under a pressure of 80. 0 k. Pa. At what temperature would the gas occupy 20. 0 L if we increased the pressure to 107 k. Pa? § § § P 1 = 80. 0 k. Pa V 1 = 10. 0 L T 1 = 240. °C = 513 K § § § P 2 = 107 k. Pa V 2 = 20. 0 L T 2 = ? P 1 V 1 T 2 = P 2 V 2 T 1 (80. 0 k. Pa)(10. 0 L)(T 2) = (107 k. Pa)(20. 0 L)(513 K) T 2= 1372 K
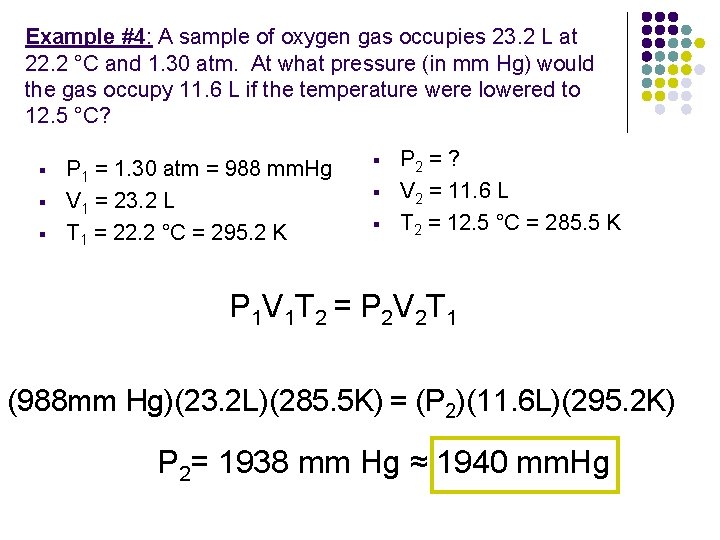
Example #4: A sample of oxygen gas occupies 23. 2 L at 22. 2 °C and 1. 30 atm. At what pressure (in mm Hg) would the gas occupy 11. 6 L if the temperature were lowered to 12. 5 °C? § § § P 1 = 1. 30 atm = 988 mm. Hg V 1 = 23. 2 L T 1 = 22. 2 °C = 295. 2 K § § § P 2 = ? V 2 = 11. 6 L T 2 = 12. 5 °C = 285. 5 K P 1 V 1 T 2 = P 2 V 2 T 1 (988 mm Hg)(23. 2 L)(285. 5 K) = (P 2)(11. 6 L)(295. 2 K) P 2= 1938 mm Hg ≈ 1940 mm. Hg

IDEAL GASES: • If a gas follows the following assumptions, it is considered to have ideal behavior: Particles take up no volume Random, straight line trajectories Elastic collisions No attractive or repulsive forces

Ideal Gases continued… • An ideal gas can be described using the following variables: Pressure (P) Volume (V) Temperature (T) Number of Moles (n) • If any three of these variables can be measured, the fourth can be determined using the ideal gas law.

Ideal Gas Law • PV = n. RT P = pressure (in atm or k. Pa) V = volume (Liters) n = moles T = temperature (Kelvin) R = ideal gas constant R = 0. 0821 (L • atm)/(mol • K) or R= 8. 314 (L • k. Pa)/(mol • K) **use whichever value matches your pressure units!

Ideal Gas Law Example #1: You fill a rigid metal container with a volume of 20. 0 L with nitrogen gas to a pressure of 197 atm at 28. 0 o. C. How many moles of gas are present in the container? § § P = 197 atm V = 20. 0 L n=? T = 28. 0°C + 273 = 301 K PV = n. RT (197 atm)(20. 0 L) = (n)(0. 0821 L·atm/mol·K)(301 K) n = 159 mol

Example #2: What volume would 50. 0 g of ethane, C 2 H 6, occupy at 140. ºC under a pressure of 1820 torr? § § P = (1820 torr)(1 atm/760 torr) = 2. 39 atm V=? n = (50. 0 g)(1 mol / 30. 0 g) = 1. 67 mol T = 140. °C + 273 = 413 K PV = n. RT (2. 39 atm)(V) = (1. 67 mol)(0. 0821 L·atm/mol·K)(413 K) V = 23. 7 L

Example #3: Calculate (a) the # moles in, and (b) the mass of an 8. 96 L sample of methane, CH 4, measured at standard conditions. (a) § § P = 1. 00 atm V = 8. 96 L n=? T = 273 K PV = n. RT (1 atm)(8. 96 L) = (n)(0. 0821 L·atm/mol·K)(273 K) n = 0. 400 mol

Example #3: Calculate (a) the # moles in, and (b) the mass of an 8. 96 L sample of methane, CH 4, measured at standard conditions. (b) § Convert moles to grams…

Example #4: Calculate the pressure exerted by 50. 0 g ethane, C 2 H 6, in a 25. 0 L container at 25. 0ºC? § § P=? V = 25. 0 L n = (50. 0 g)(1 mol / 30. 0 g) = 1. 67 mol T = 25 °C + 273 = 298 K PV = n. RT (P)(25. 0 L) = (1. 67 mol)(0. 0821 L·atm/mol·K)(298 K) P = 1. 63 atm
- Slides: 16