Ideal Gas Law Gas Mixtures Ideal Gas Law



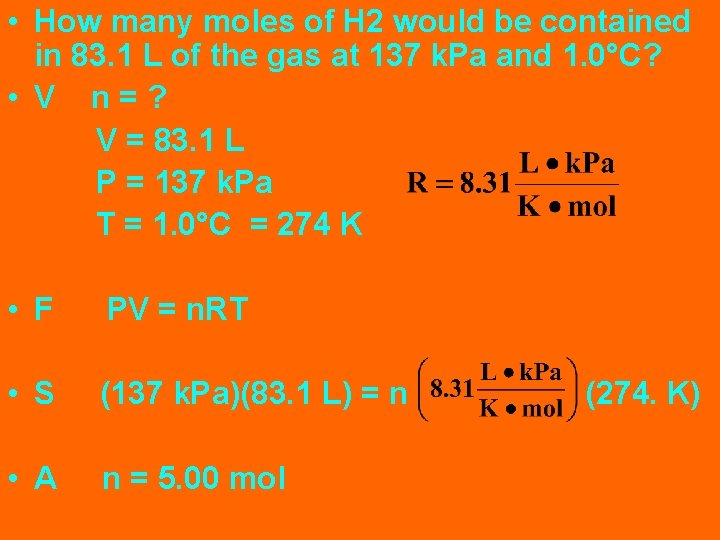



- Slides: 10

Ideal Gas Law & Gas Mixtures

• Ideal Gas Law: PV = n. RT • Where n = the number of moles • R is the Ideal Gas Constant • The ideal gas law can be used to calculate the number of moles of a contained gas.

• What is the volume (in L) that would be occupied by 1. 00 mol of O 2 at STP? • V V=? n = 1. 00 mol T = 273 K P = 101. 3 k. Pa • F PV = n. RT • S (101. 3 k. Pa) V = (1. 00 mol) • A V = 22. 4 L (273 K)
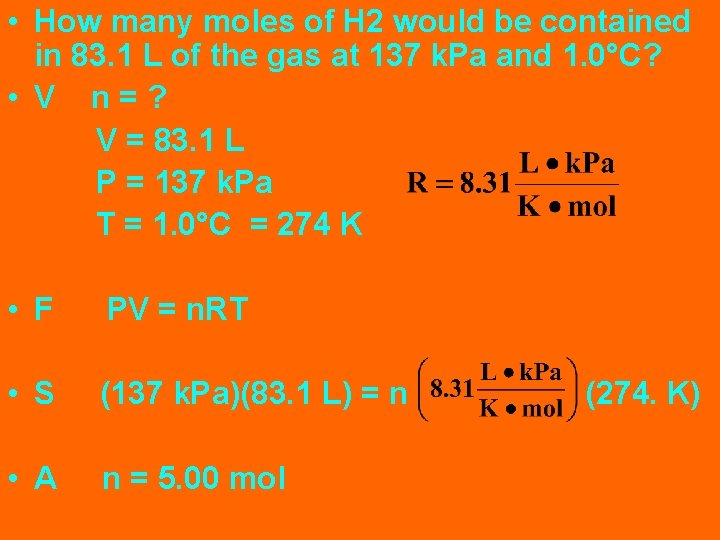
• How many moles of H 2 would be contained in 83. 1 L of the gas at 137 k. Pa and 1. 0°C? • V n=? V = 83. 1 L P = 137 k. Pa T = 1. 0°C = 274 K • F PV = n. RT • S (137 k. Pa)(83. 1 L) = n • A n = 5. 00 mol (274. K)

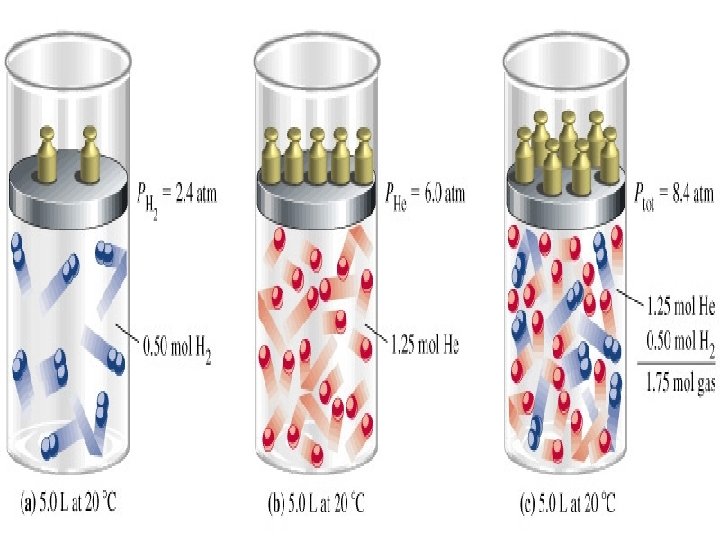
Gas Mixtures • Dalton’s Law - In a mixture of gases, the total pressure is the sum of the partial pressures of the gases. • PTotal = P 1 + P 2 + P 3 + …

• A sample of H 2 is collected over water such that the combined hydrogen-water vapor sample is held at a pressure of 1 standard atmosphere. What is the partial pressure of the H 2 if that of the water vapor is 2. 5 k. Pa? • 1 atm = 101. 3 k. Pa • 101. 3 k. Pa – 2. 5 k. Pa = 98. 8 k. Pa

• The pressure of a mixture of nitrogen, carbon dioxide, and oxygen is 150 k. Pa. What is the partial pressure of oxygen if the partial pressures of the nitrogen and carbon dioxide are 100 k. PA and 24 k. Pa, respectively? • P = Pnitrogen + Pcarbon dioxide + Poxygen • 150 k. Pa = 100 k. Pa + 24 k. Pa + Poxygen • Poxygen = 150 k. Pa - 100 k. Pa - 24 k. Pa • Poxygen = 26 k. Pa


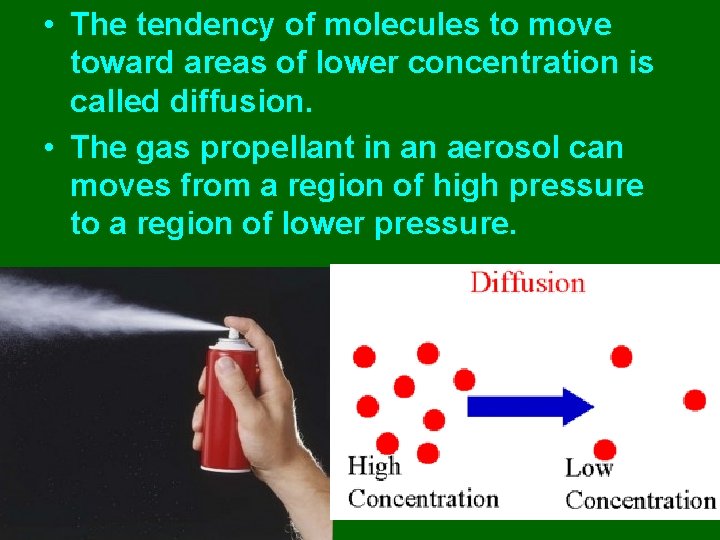
• The tendency of molecules to move toward areas of lower concentration is called diffusion. • The gas propellant in an aerosol can moves from a region of high pressure to a region of lower pressure.

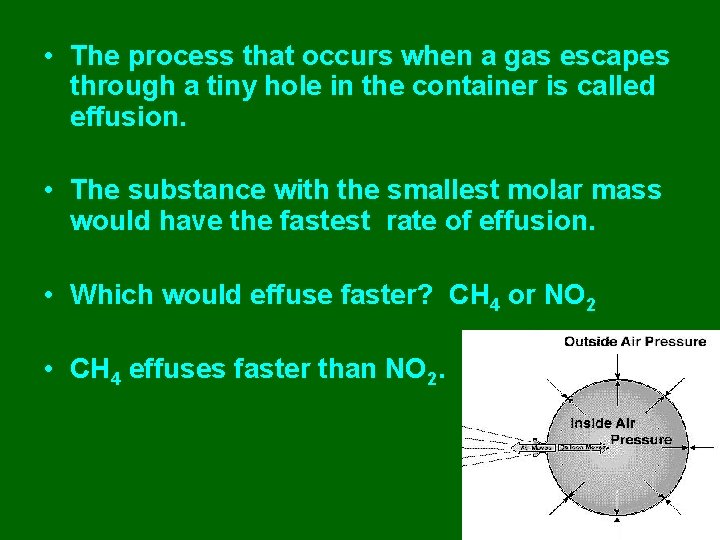
• The process that occurs when a gas escapes through a tiny hole in the container is called effusion. • The substance with the smallest molar mass would have the fastest rate of effusion. • Which would effuse faster? CH 4 or NO 2 • CH 4 effuses faster than NO 2.