Real Gases Deviation from ideal gas Law Gas














- Slides: 27

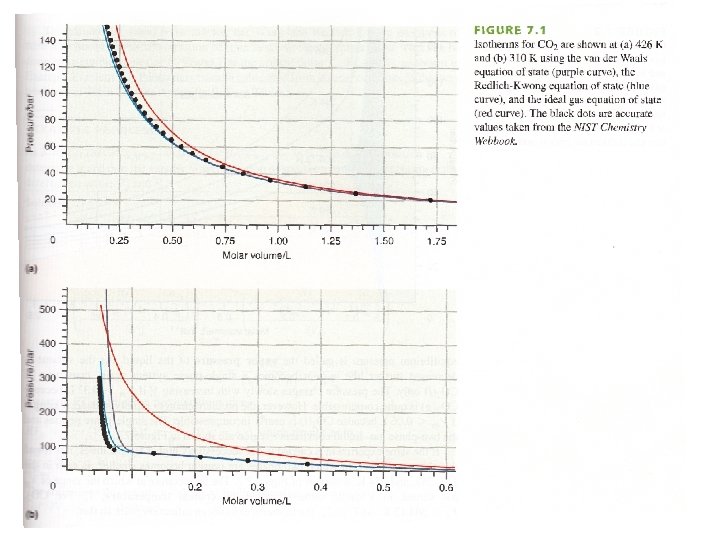
Real Gases Deviation from ideal gas Law: -Gas particles have volume - Attraction exists between gas particles (liquefication) Ideal gas law applies only at low gas density: high T, low p

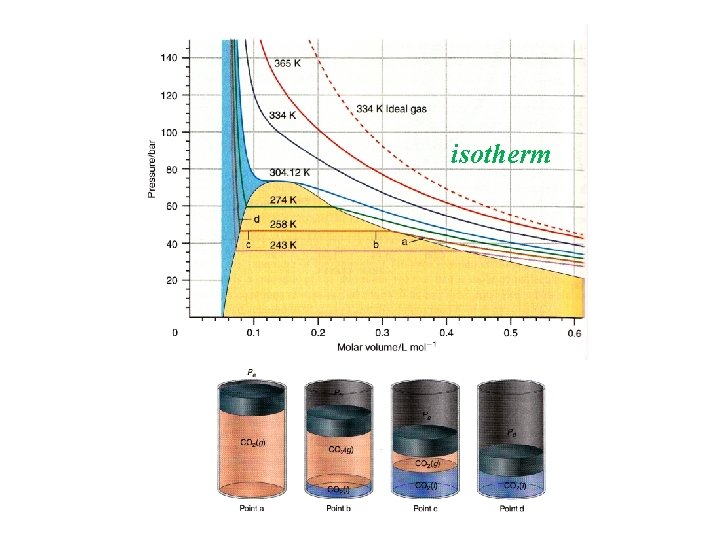
isotherm

Van der Waal’s Equation Ideal gas law needs modification: - Let’s consider the molecular volume: b: size of 1 mole of gas molecules For 1 mole of gas: Vm, total = Vm, space + b

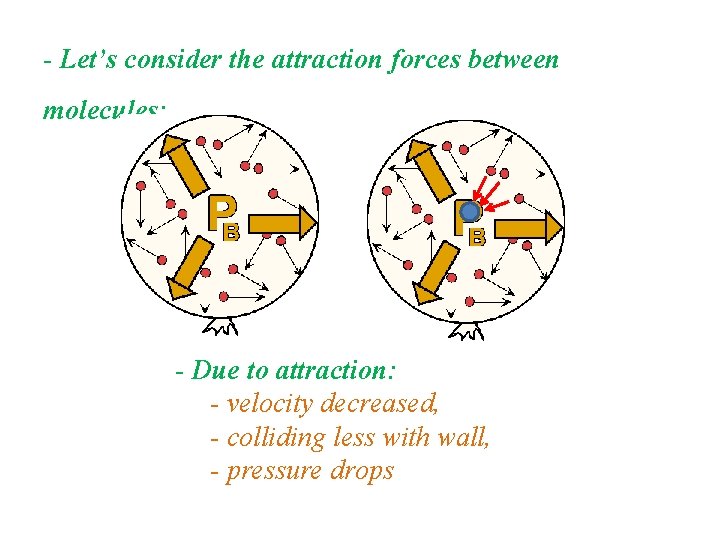
- Let’s consider the attraction forces between molecules: - Due to attraction: - velocity decreased, - colliding less with wall, - pressure drops

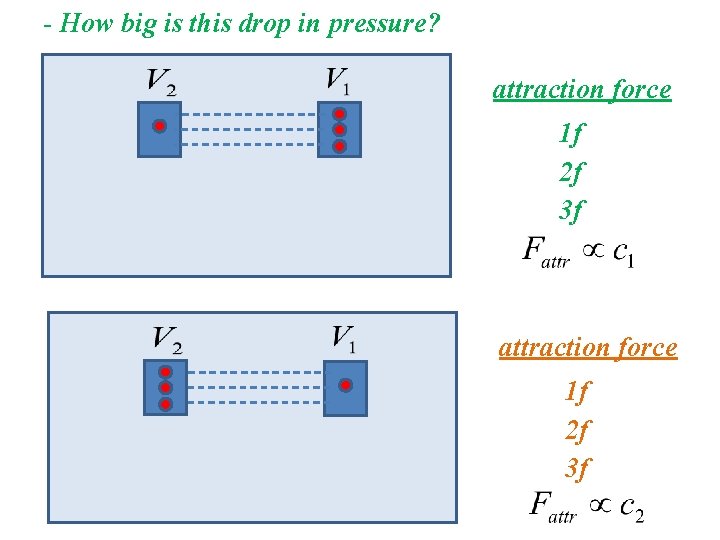
- How big is this drop in pressure? attraction force 1 f 2 f 3 f

- How big is this drop in pressure?

- How big is this drop in pressure? drop in pressure proportional to attraction force drop in pressure indirectly proportional to square of Vm

Vd. W Equation of state

b: volume of 1 mole of gas molecules. a: represents strength of attraction.


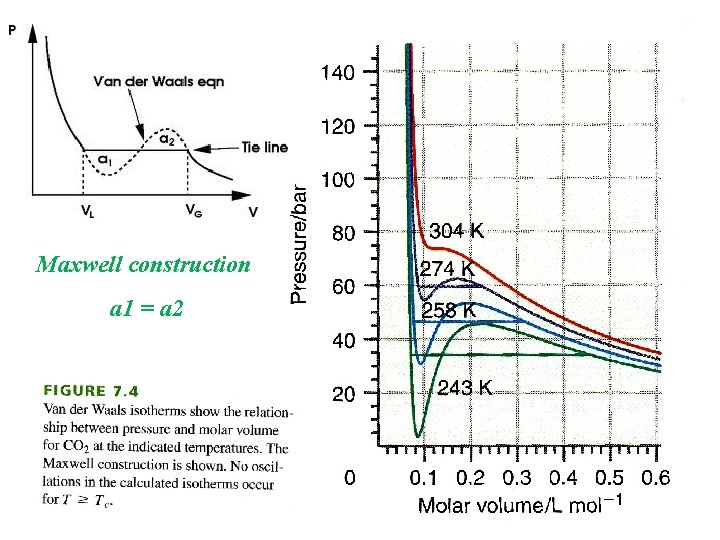
Maxwell construction a 1 = a 2

Many other equations of state Mostly empirical Virial Equation to correct, develop in a series (additional correction terms: B, C, …: second, third, … virial coefficients: exp. determined

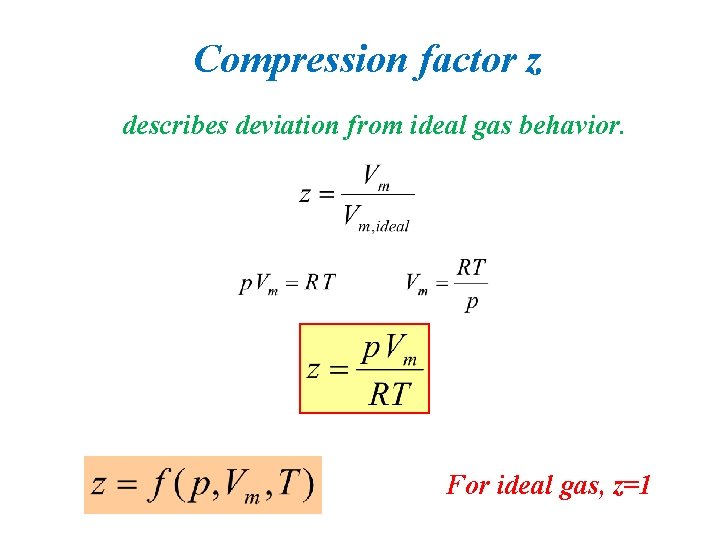
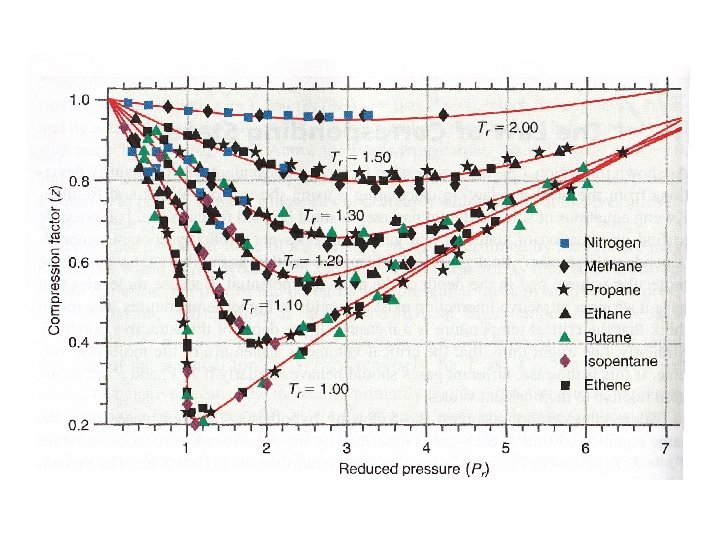
Compression factor z describes deviation from ideal gas behavior. For ideal gas, z=1

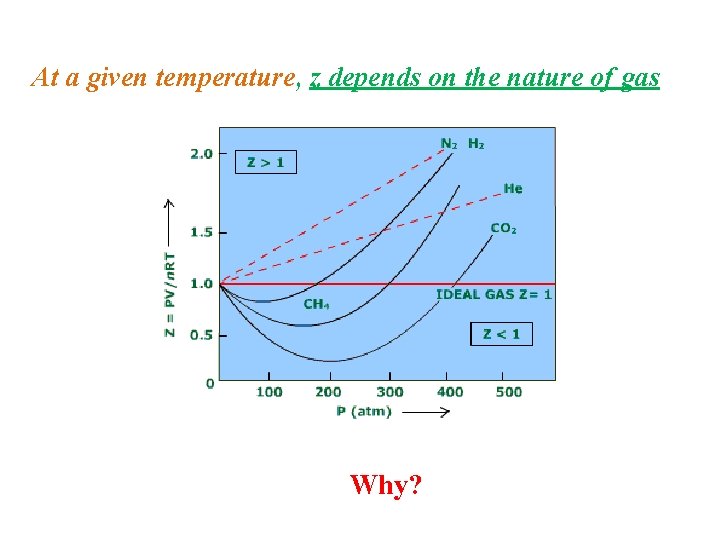
At a given temperature, z depends on the nature of gas Why?

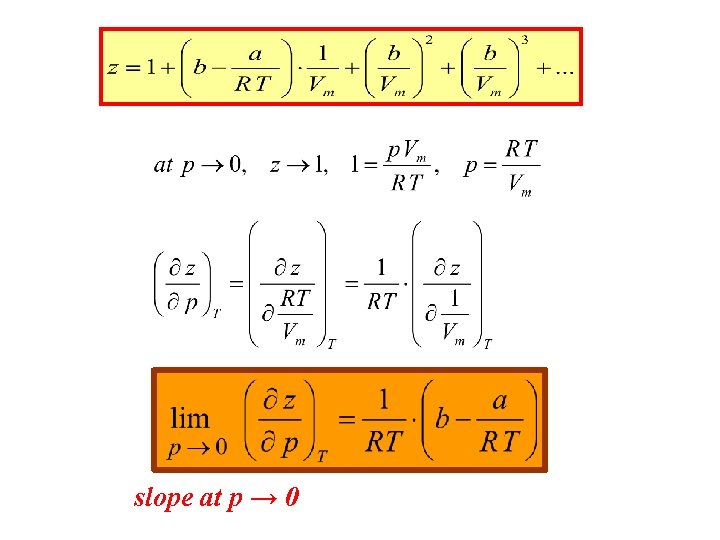
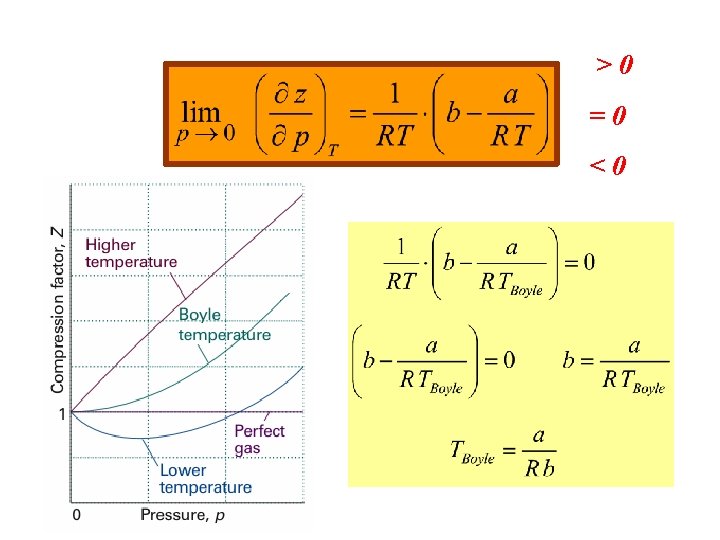
Let’s find for a Vd. W gas at p → 0 The slope of the curve z=f(p) in the previous diagram.


slope at p → 0

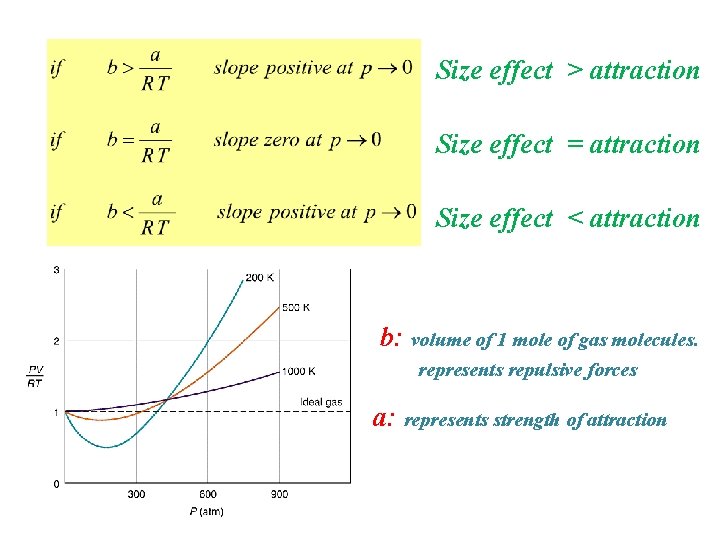
>0 =0 <0

Size effect > attraction Size effect = attraction Size effect < attraction b: volume of 1 mole of gas molecules. represents repulsive forces a: represents strength of attraction

Explain the trend!

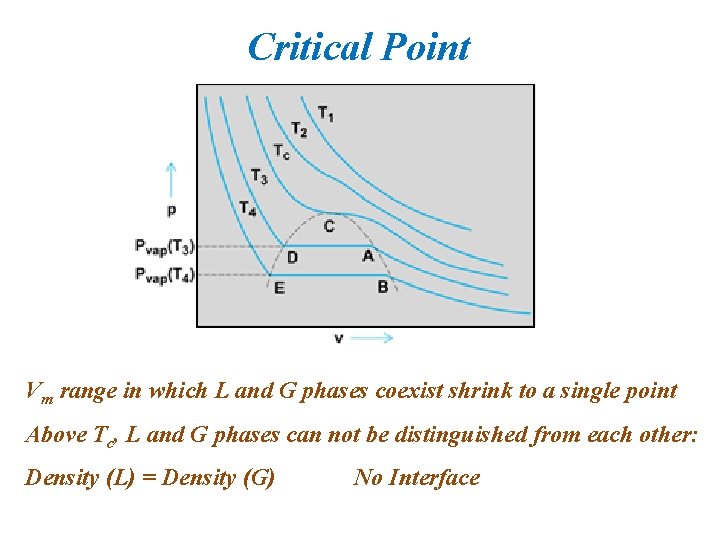
Critical Point Vm range in which L and G phases coexist shrink to a single point Above Tc, L and G phases can not be distinguished from each other: Density (L) = Density (G) No Interface


Experimental measurements of pc and Tc are more accurate than that of Vc, a and b are calculated from the experimental values of pc and Tc.

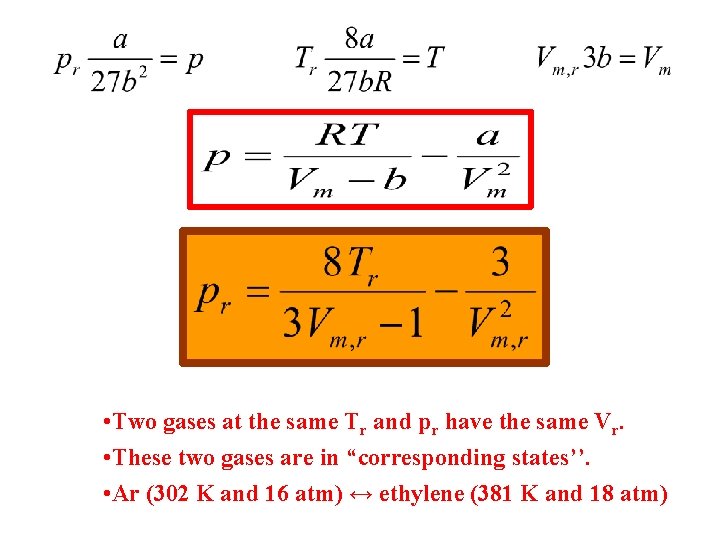
The law of corresponding states Is it possible to find an equation of state that doesn’t contain material specific constants? Reduced properties:

• Two gases at the same Tr and pr have the same Vr. • These two gases are in “corresponding states’’. • Ar (302 K and 16 atm) ↔ ethylene (381 K and 18 atm)

