Pressure 1 Gas Laws Next Slide Introduction of



















- Slides: 31

Pressure 1 Gas Laws Next Slide Introduction of pressure • Pressure is defined as : Pressure = force perpendicular to an area • Gas pressure comes from the collisions between the gas molecules and the wall of container Diagram • Molecules in air also exert pressure on all objects (even on our body) : atmospheric pressure Diagram

Pressure 2 Gas Laws Next Slide Pressure • Pressure can be measured by an instrument called Bourdon gauge Photo

Gas Laws 1 Gas Laws Next Slide Gas Laws • Relationships between P, V and T for a fixed amount of gas • Boyle’s law : P and V’s relation at a fixed T Diagram • Pressure’s law : P and T ’s relation at a fixed V Diagram • Charle’s law : V and T’s relation at a fixed P Diagram • General gas equation and examples Example

Kinetic Theory 1 Gas Laws Next Slide Real gas and Ideal gas • Real gas : a gas which does not obey the general gas equation for all values of P, V and T. • Ideal gas : a gas which obeys the general gas equation for all values of P, V and T. • A real gas behaves almost exactly the same as an ideal gas at low pressures and temperatures well above its boiling point.

Kinetic Theory 2 Gas Laws Next Slide Kinetic Theory 1. all matter is made up of very tiny constantly moving particles, namely atoms, ions and molecules. 2. the particles are in constant random motion. 3. the particles move more quickly when the matter is heated. 4. the particles exert practically no force on each other when they are widely separated. 5. the particles attract each other when they are slightly apart, but if they are forced too close together, they will repel each other strongly.

Kinetic Theory 3 Gas Laws Next Slide States of matter • Structure of a solid • Structure of a liquid • Structure of a gas Diagram • Brownian motion in gas Diagram • Three-Dimension Kinetic Theory Model: Explanation of Gas Laws Diagram

END of Gas Laws

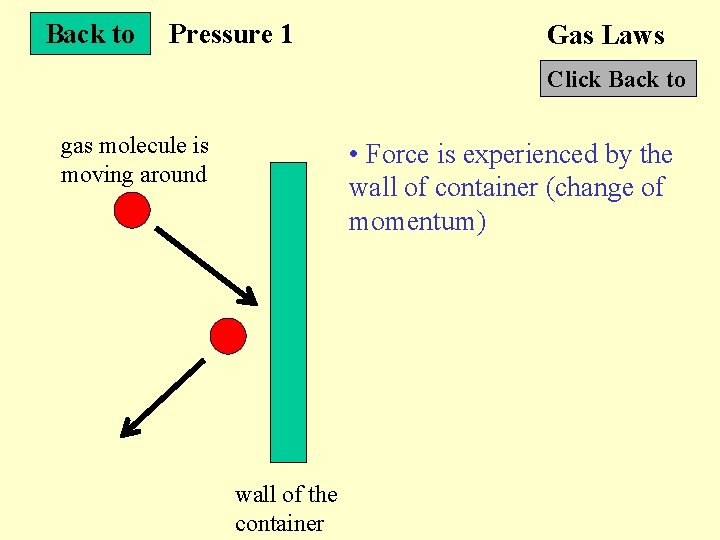
Back to Pressure 1 Gas Laws Click Back to gas molecule is moving around • Force is experienced by the wall of container (change of momentum) wall of the container

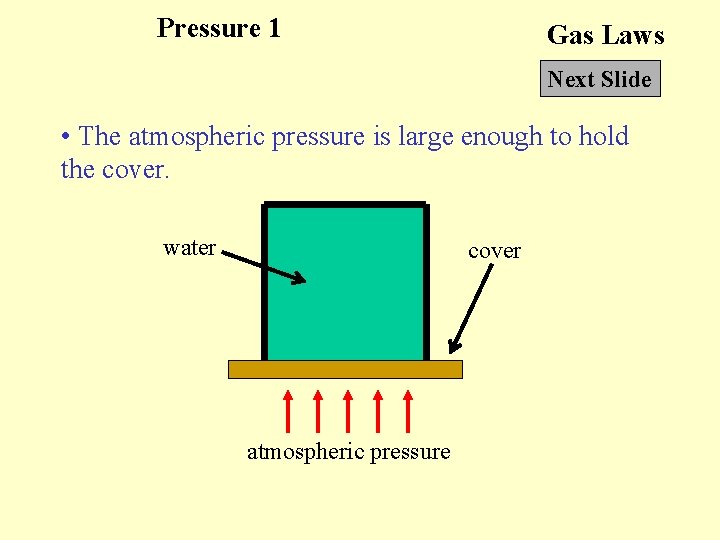
Pressure 1 Gas Laws Next Slide • The atmospheric pressure is large enough to hold the cover. water cover atmospheric pressure

Back to Pressure 1 Gas Laws Click Back to • Atmospheric pressure presses the water through the sucking tube into your month. atmospheric pressure water

Back to Pressure 2 Gas Laws Click Back to • Bourdon gauge:

Gas Laws 1 Gas Laws Next Slide • An air column is trapped in the glass tube as shown. The pressure and the volume of the gas in the glass tube can be read from the Bourdon gauge and the ruler respectively.

Gas Laws 1 Gas Laws Next Slide • The volume of the rubber tube will cause an error. The volume is unknown and should be made as small as possible. • The volume of the gas should be changed slowly to avoid error in measuring the pressure and the volume. • Boyle’s law : the pressure of a fixed mass of gas at constant temperature is inversely proportional to its volume

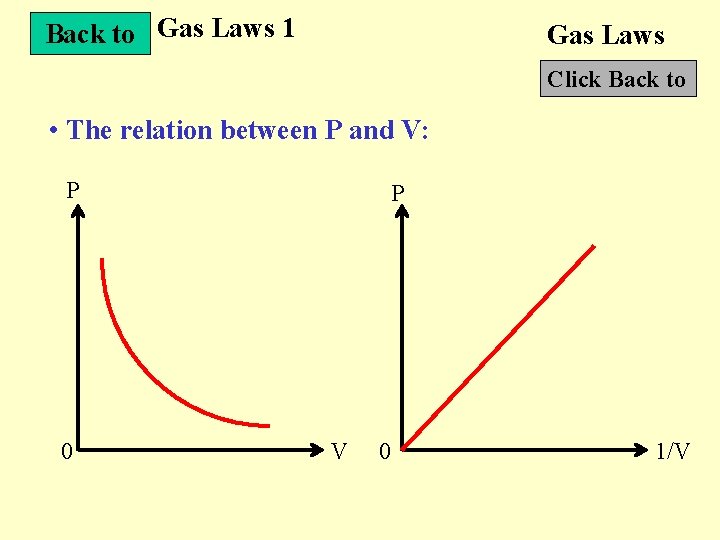
Back to Gas Laws 1 Gas Laws Click Back to • The relation between P and V: P 0 P V 0 1/V

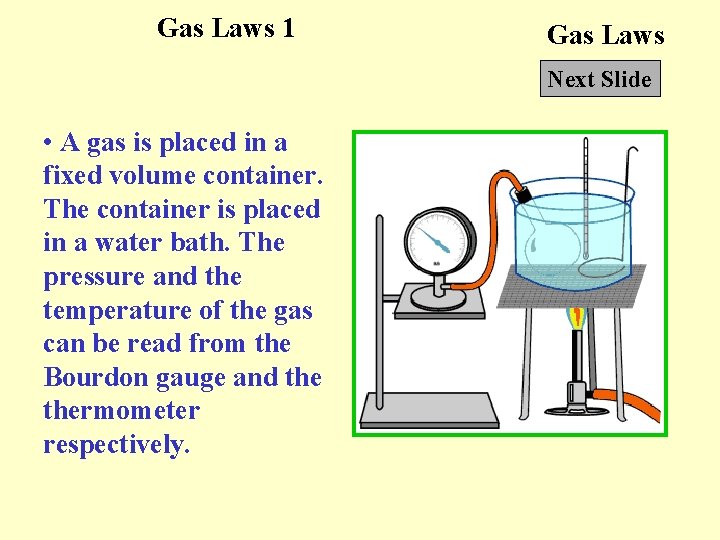
Gas Laws 1 Gas Laws Next Slide • A gas is placed in a fixed volume container. The container is placed in a water bath. The pressure and the temperature of the gas can be read from the Bourdon gauge and thermometer respectively.

Gas Laws 1 Gas Laws Next Slide • The length of the rubber tube should be made as short as possible since we cannot control the temperature of the gas in the tube. • The flask should be completely immersed in water to ensure a homogeneous temperature in the gas. • Pressure law : the pressure of a fixed mass of gas at constant volume is directly proportional to its Kelvin temperature

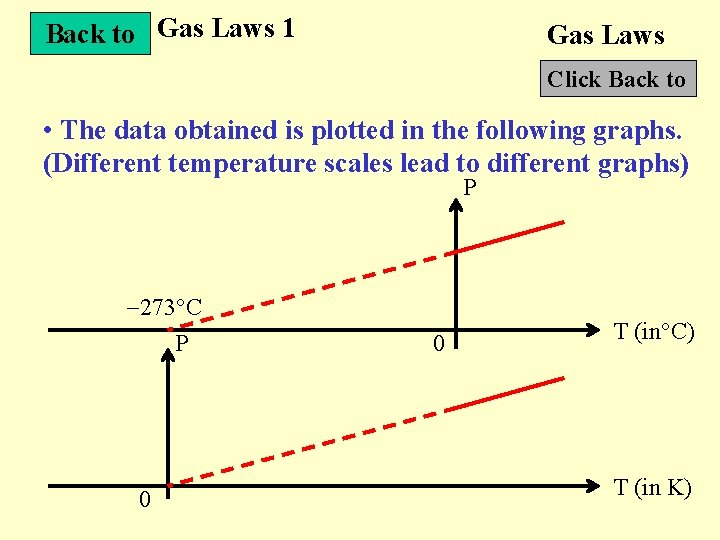
Back to Gas Laws 1 Gas Laws Click Back to • The data obtained is plotted in the following graphs. (Different temperature scales lead to different graphs) P 273 C P 0 0 T (in C) T (in K)

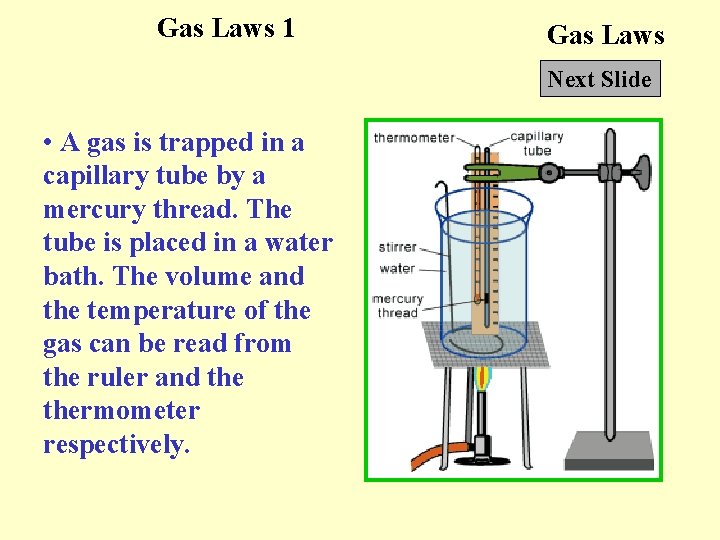
Gas Laws 1 Gas Laws Next Slide • A gas is trapped in a capillary tube by a mercury thread. The tube is placed in a water bath. The volume and the temperature of the gas can be read from the ruler and thermometer respectively.

Gas Laws 1 Gas Laws Next Slide • Since the mercury thread can freely move up and down and is in contact with atmosphere, the pressure of the gas is equal to the atmospheric pressure. • The friction between the mercury thread and the container should be small. • Charle’s law : the volume of a fixed mass of gas at constant pressure is directly proportional to its Kelvin temperature

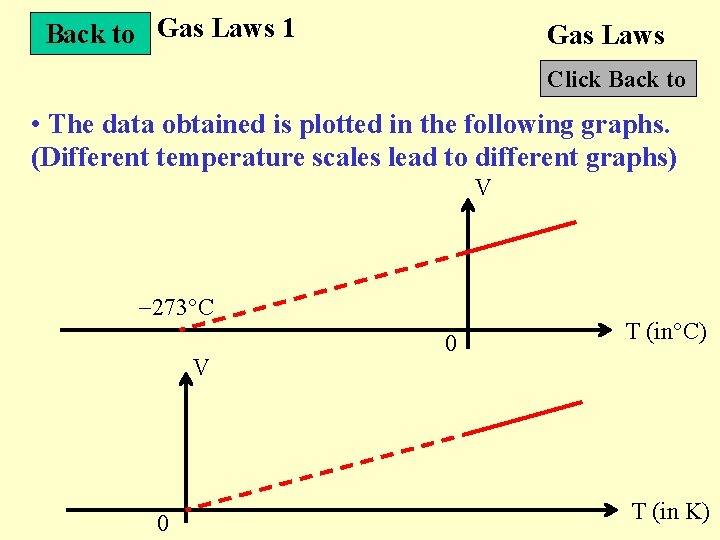
Back to Gas Laws 1 Gas Laws Click Back to • The data obtained is plotted in the following graphs. (Different temperature scales lead to different graphs) V 273 C V 0 0 T (in C) T (in K)

Gas Laws 1 Gas Laws Next Slide • All the gas laws can be summarized by general equation : Example : A weather balloon contains 5 m 3 of helium at the normal atmospheric pressure of 100 k. Pa and a temperature of 27 C. What will be its volume when it rises to an altitude where the pressure is 80 k. Pa and the temperature is 7 C?

Back to Gas Laws 1 Gas Laws Click Back to

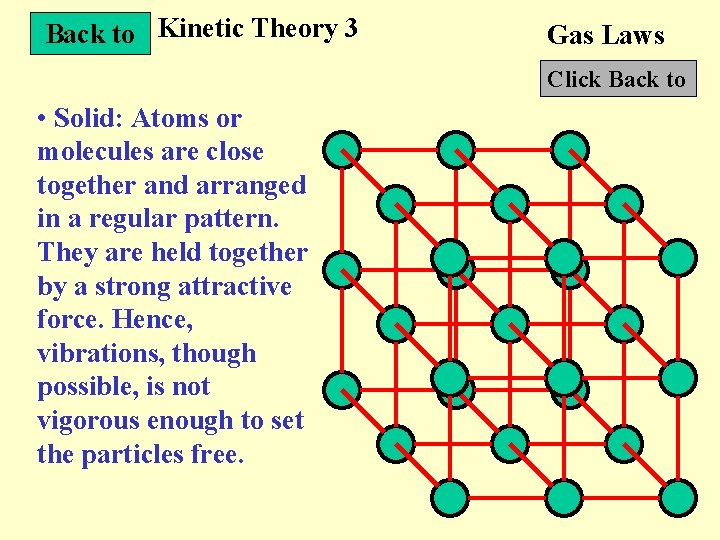
Back to Kinetic Theory 3 Gas Laws Click Back to • Solid: Atoms or molecules are close together and arranged in a regular pattern. They are held together by a strong attractive force. Hence, vibrations, though possible, is not vigorous enough to set the particles free.

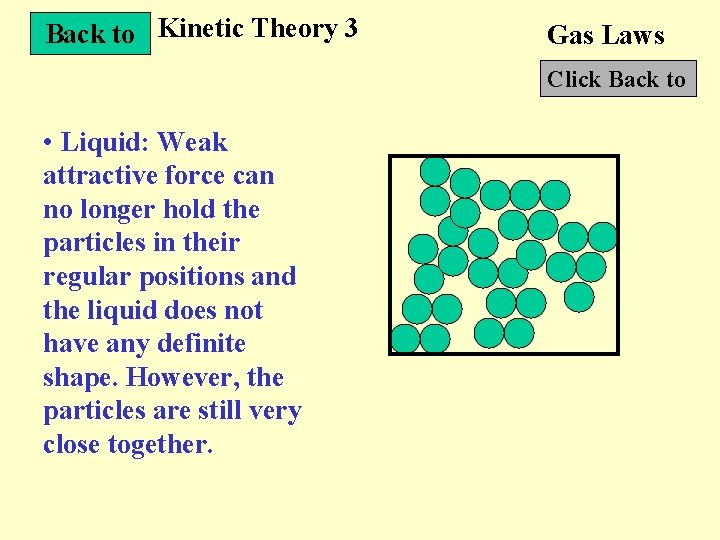
Back to Kinetic Theory 3 Gas Laws Click Back to • Liquid: Weak attractive force can no longer hold the particles in their regular positions and the liquid does not have any definite shape. However, the particles are still very close together.

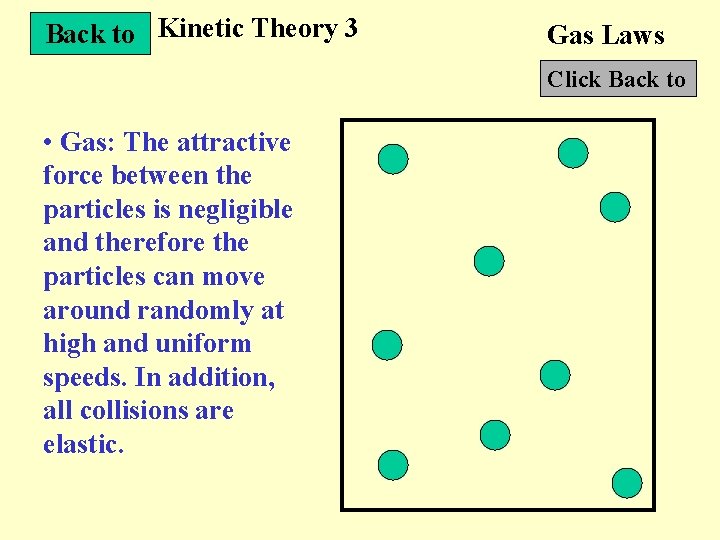
Back to Kinetic Theory 3 Gas Laws Click Back to • Gas: The attractive force between the particles is negligible and therefore the particles can move around randomly at high and uniform speeds. In addition, all collisions are elastic.

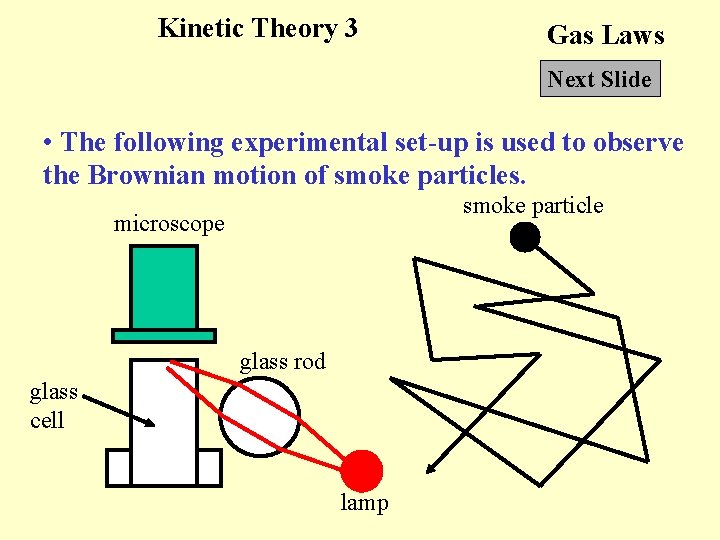
Kinetic Theory 3 Gas Laws Next Slide • The following experimental set-up is used to observe the Brownian motion of smoke particles. smoke particle microscope glass rod glass cell lamp

Back to Kinetic Theory 3 Gas Laws Click Back to • When we observe the smoke particles in a smoke cell under microscope, the smoke particles move jerkily about. The motion is called Brownian motion. The collisions between the air molecules and the smoke particles cause the random motions of the smoke particles.

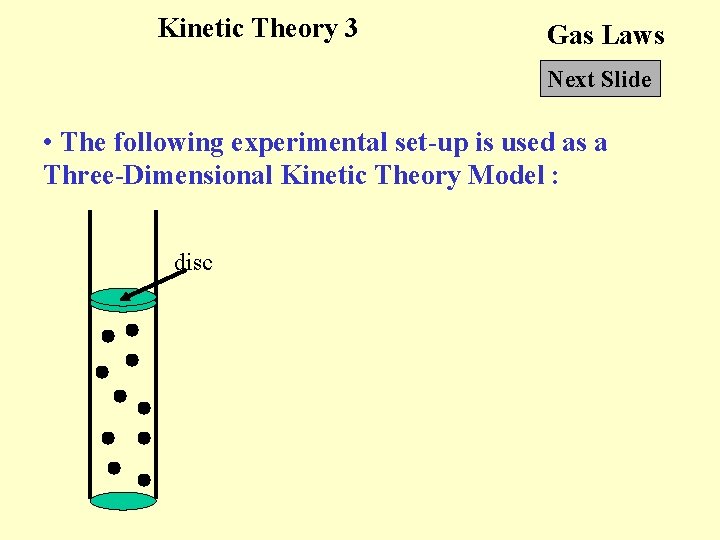
Kinetic Theory 3 Gas Laws Next Slide • The following experimental set-up is used as a Three-Dimensional Kinetic Theory Model : disc

Kinetic Theory 3 Gas Laws Next Slide • The motor under the cylinder set the bearings in motion. Due to the collisions between the disc and the bearings, the disc rises to a certain level. Pressure of the ‘gas’ : weight of the disc Volume of the ‘gas’ : volume occupied by the bearings Temperature of the ‘gas’ : voltage of the power supply which drives the motor

Kinetic Theory 3 Gas Laws Next Slide • Boyle’s law’s demonstration : • We keep the voltage of the motor constant (T constant), and add discs one after another (P increases). We can observe a decrease in volume. • Pressure law’s demonstration : • We add discs one after another (P increases). In order to maintain a constant volume (V constant), we must increase the voltage gradually (T increases).

Back to Kinetic Theory 3 Gas Laws Click Back to • Charles’ law’s demonstration: • We keep the no. of discs constant (P constant) and increase the voltage (T increases). We can observe an increase in volume.