Gases The behavior of gases can be explained

Gases The behavior of gases can be explained by the KINETIC MOLECULAR THEORY. This theory has three assumptions about what matter is made up of. Link to webpage describing Kinetic Mol. Theory

Three Assumptions of K. M. Theory 1. Matter is made up of molecules. 2. Molecules are in constant random motion 3. Molecules undergo perfectly elastic collisions (they do not lose energy when they collide).

Pressure How is pressure defined? Pressure is defined as the force applied to a given area. How is pressure commonly measured (think measuring the pressure in your bike tire)? Pounds per square inch (PSI). Other units of pressure include atmospheres, mm of Hg, Torr, and Pascals.

How does a gas exert pressure? Let’s take a look at a simulation to see if we can answer this question.

Pressure depends on The molecules of a gas colliding with the walls of the container. Anything that increases the number of collisions per unit area, or the force of the collisions will increase the pressure of the gas.

Using a mercury barometer to measure pressure • A mercury barometer works by measuring the height of the column of mercury supported by the air pressure. • Standard Pressure is when the column of mercury is 760 millimeters high.
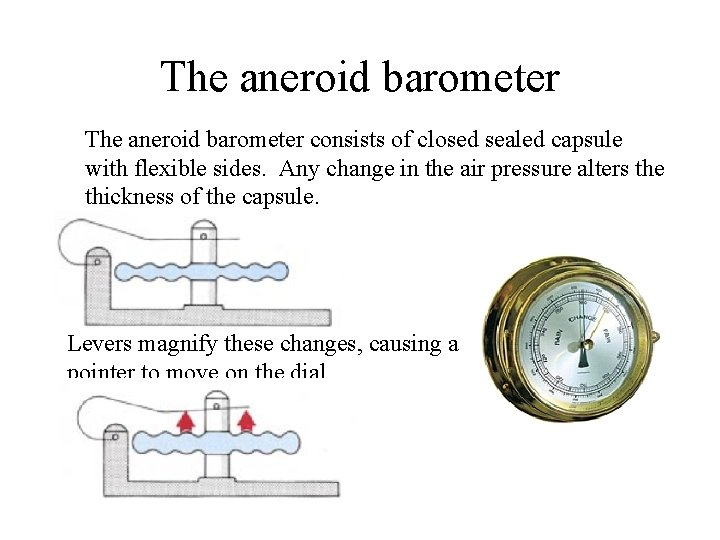
The aneroid barometer consists of closed sealed capsule with flexible sides. Any change in the air pressure alters the thickness of the capsule. Levers magnify these changes, causing a pointer to move on the dial.

The air pressure can be measured using a barometer • The pressure of the atmosphere in any location changes as a result of the uneven heating of the atmosphere. • Normal atmospheric pressure at sea level is called Standard Pressure.

Values for Standard Pressure is expressed by any of the following units: • 1. 0 atmospheres (atm) • 760 millimeters of mercury (mm of Hg) • 760 Torr (in honor of Torricelli – inventor of the Hg barometer • 101 kilo. Pascals (k. Pa)

Temperature The temperature of a gas is a measure of the average kinetic energy of the molecules making up the gas.

Average Molecular Motion • Not all molecules in a sample of gas move at the same speed. • Study this graph, how are the molecules in this sample like the runners in a big cross country race. In a xc race some runners are fast, some are slow, but most run at about the same speed. The same is true for molecules in a sample of a gas.

Temperature & Molecular Motion • Increasing the temperature of a sample of gas will cause the average speed of the molecules of the gas to increase. • Click here to view a simulation showing the relationship of molecular motion to temperature.

The Kelvin Temperature Scale • The Kelvin Temperature Scale is referred to as the Absolute Temperature scale. • Zero Kelvin is the temperature at which all molecular motion stops. • Temperatures in the lab are usually measure in Celsius and converted to Kelvin using: K = o. C + 273

Standard Temperature is equal to 273 K (or 0 o. C).

STP This is the abbreviation for Standard Temperature & Pressure It indicates that the pressure of the gas is 1 atm and the temperature is 273 K

Volume • The volume of a gas refers to the space taken up by the gas (as opposed to the volume of the gas molecules). • The volume of a gas is always equal to the volume of its container.

Boyle’s Law • Boyle’s Law states that for a sample of gas containing a definite number of molecules at constant temperature, as the pressure of the gas increases, the volume will decrease.
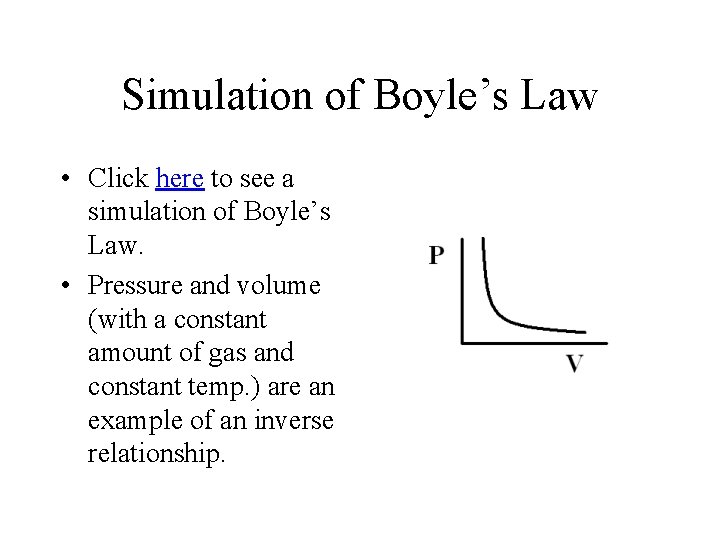
Simulation of Boyle’s Law • Click here to see a simulation of Boyle’s Law. • Pressure and volume (with a constant amount of gas and constant temp. ) are an example of an inverse relationship.

Mathematical Statement of Boyle’s Law • The equation which describes the relationship shown in the graph is P 1 x V 1 = P 2 x V 2 Where P 1 and V 1 are the initial pressure and volume of a sample of gas, and P 2 and V 2 are the pressure of the gas after it has been expanded or contracted (Temp. remains constant).

Units and Boyle’s Law • Make sure that the units you use for pressure are the same for both P 1 and P 2. (Never use one unit (example atm) for P 1 and a different unit (example Torr for P 2. ) • Units for volume must also agree. (1 milliliter (m. L) is the same as 1 cubic centimeter (cm 3 or cc)

Sample Boyle’s Law Problem • Suppose you have a cylinder & piston containing 8 liters of hydrogen at 1 atmosphere of pressure. (P 1 = 1 atm and V 1 = 8 liters) • What will happen if the piston is pushed in till the volume of the gas is equal to 4 liters?

Boyle’s Law Problem -2 • The molecules will be pushed closer together thus increasing the pressure. • The volume of the gas will decrease. (V 2 = 4 liters)

Boyle’s Law Calculation • Using the equation P 1 x V 1 = P 2 x V 2 and plugging in we get 1 atm x 8 L = P 2 x 4 L Solving for P 2 gives us the value of the new pressure: P 2 = 2 atm

Practice Problems Solve the following problems (assume the temperature & amount of gas is constant): 1. The initial pressure of a sample of gas is 75 k. Pa and the initial volume is 15 ml. If the pressure is increased to 101 k. Pa, what will be the new volume of the gas? 11. 1 m. L 2. The pressure of a sample of gas is 760 Torr and the volume of the gas is 2. 5 L. What will be the pressure of the gas is compressed to 1. 75 L? 1086 Torr

Charles’ Law • Charles’ Law states that for a sample of gas containing a definite number of molecules at constant pressure, as the temperature of the gas increases, the volume will increase.
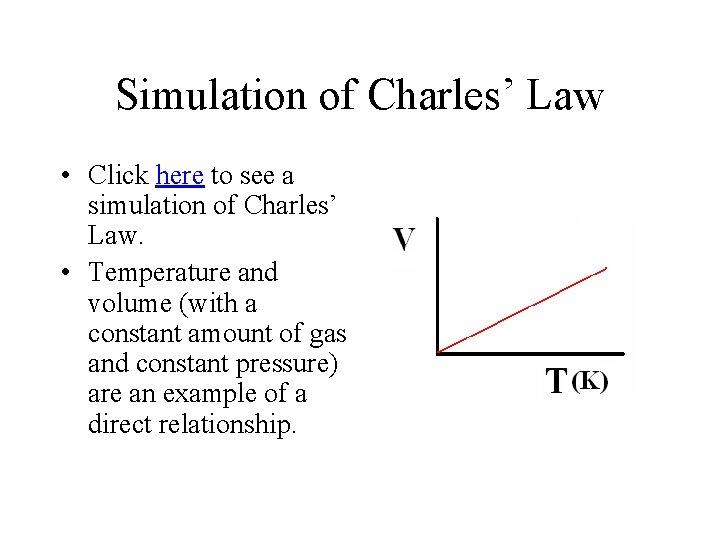
Simulation of Charles’ Law • Click here to see a simulation of Charles’ Law. • Temperature and volume (with a constant amount of gas and constant pressure) are an example of a direct relationship.
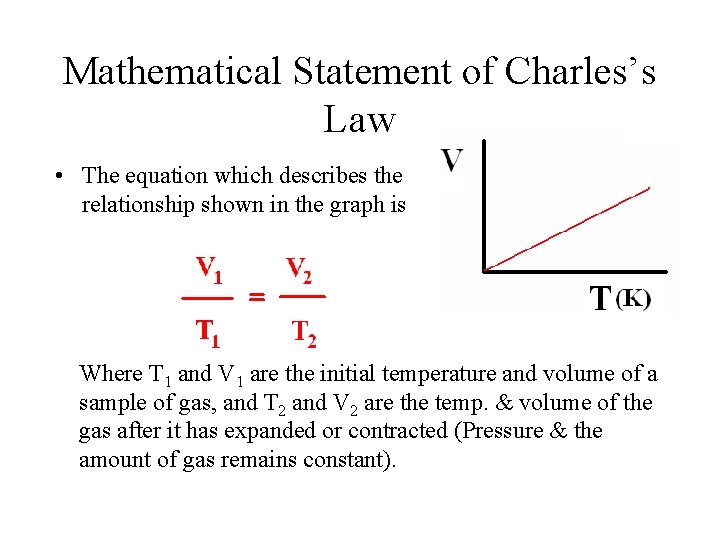
Mathematical Statement of Charles’s Law • The equation which describes the relationship shown in the graph is Where T 1 and V 1 are the initial temperature and volume of a sample of gas, and T 2 and V 2 are the temp. & volume of the gas after it has expanded or contracted (Pressure & the amount of gas remains constant).

Charles’s Law Sample Problem • A 12 m. L sample of a gas is enclosed in a syringe at 25 o. C. If the temperature is raised to 30 o. C, what will the new volume of the gas be if the pressure remains constant? V 1 = 12 ml T 1 = 298 K (temperature MUST be converted to Kelvin) V 2 = ? m. L T 2 = 303 K (temperature MUST be converted to Kelvin)

Charles’s Law Solution

Charles’s Law -Practice Problems 1. A 4. 5 L sample of gas at 20 o. C is heated to a temperature of 80 o. C. If the amount of gas and the pressure of the gas remain constant, what will be the new volume of the gas? 5. 4 L 2. A sample of gas at 60 o. C occupies a volume of 35 m. L. If the gas is allowed to expand until it occupies a volume of 52 m. L, what will be the new temperature of the gas (assuming the quantity and pressure of the gas are constant)? 495 K ( 222 o. C )

Combined Gas Law Boyle’s Law is valid when you have a constant amount of gas at constant temperature. Charles’s Law is valid when you have a constant amount of gas at constant pressure. But what happens when the volume, the pressure, and the temperature all change at the same time? Use the Combined Gas Law

The equation of the Combined Gas Law Boyle’s Law P 1 x V 1 Charles’s Law = P 2 x V 2 THE COMBINED GAS LAW

Sample Combined Gas Law A 25 m. L sample of a gas has a pressure of 81 k. Pa and a temperature of 25 o. C, what would the volume of the gas be at STP? Identify and label the various variables: V 1 = 25 m. L V 2 = ? P 1 = 81 k. Pa P 2 = 101 k. Pa T 1= 298 K (Must convert to Kelvin) T 1 = 273 K (Must convert to Kelvin)
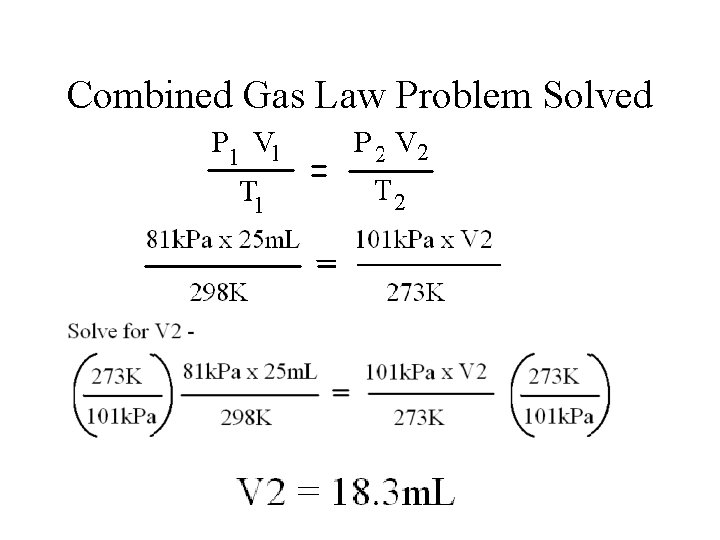
Combined Gas Law Problem Solved

Combined Gas Law Problems 1. A 35 cm 3 sample of gas at 25 o. C and a pressure of 95. 6 k. Pa is compressed and cooled until it has a pressure of 250 k. Pa and a temperature of -15 o. C. What would be the new volume of the gas? 11. 6 cm 3 2. 1. 83 L of gas with a temperature of 53 o. C and a pressure of 843 Torr is converted to STP. What will be the new volume of the gas? 1. 70 L

Dalton’s Law of Partial Pressures Suppose a sample of gas contains a mixture of gases, how do the pressure of the individual gases affect the total pressure of the gases? To answer the question, let’s look at a diagram of the container holding the gases:
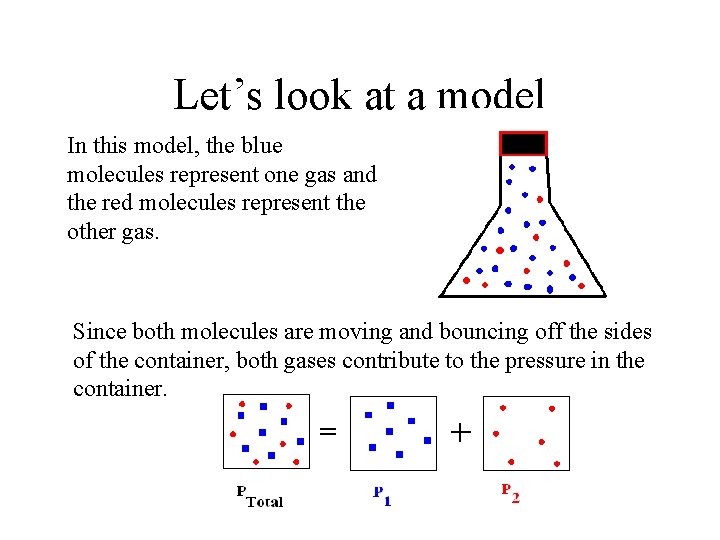
Let’s look at a model In this model, the blue molecules represent one gas and the red molecules represent the other gas. Since both molecules are moving and bouncing off the sides of the container, both gases contribute to the pressure in the container.

Statement of Dalton’s Law Since both gases contribute to the pressure: The total pressure in a mixture of gases is equal to the sum of the pressures of the individual gases in the mixture. This law can be expressed by the following equation: Ptotal = Pgas a + Pgas b

Sample Dalton’s Law Problem What is the pressure of a mixture of He and Ne gas when the He has a pressure of 480 Torr and the Ne has a pressure of 290 Torr? The formula states: Ptotal = PHe + P Ne Plugging in we get: Ptotal = 480 Torr + 290 Torr Final answer: Ptotal = 770 Torr

Dalton’s Law Practice 1. 2. A container contains three gases. The pressure of O 2 is 320 Torr, the pressure of He is 220 Torr, and the pressure of Ne is 56 Torr. What is the total pressure in the container? In this case there are three gases, so Ptotal = 320 Torr + 220 Torr + 56 Torr =596 Torr The pressure in a container is measure to be 150 k. Pa. If the container contains N 2 and O 2 gases and the pressure of the N 2 gas is 110 k. Pa, what is the pressure of the O 2 gas? Ptotal = 150 k. Pa = 110 k. Pa + PO 2 = 40 k. Pa
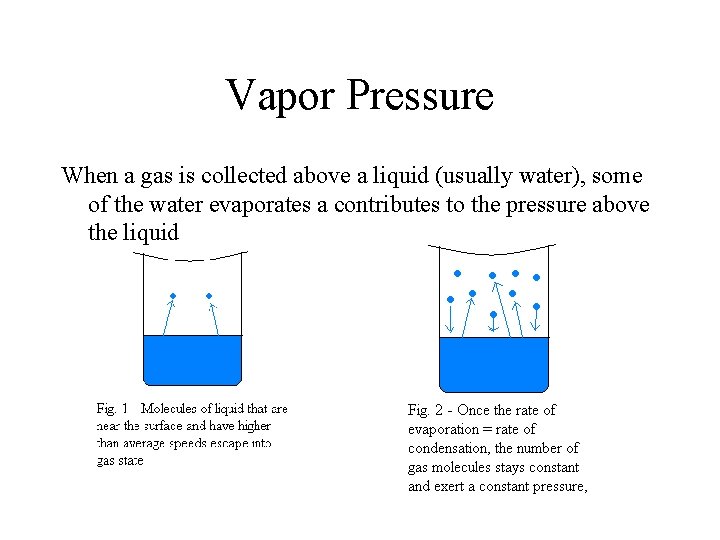
Vapor Pressure When a gas is collected above a liquid (usually water), some of the water evaporates a contributes to the pressure above the liquid

Two Factors determine vapor pressure 1. The strength of the intermolecular attractions in the liquid.

Factors Affecting Pvapor 2. The temperature of the liquid.

Vapor Pressure and the Boiling Point • The vapor pressure of a liquid is related to the boiling point of a liquid. • The higher the vapor pressure of a substance, the lower the boiling point of the substance. • Why? Both vapor pressure and boiling point relate to how easily a liquid can become a gas. The stronger the intermolecular attractions the lower the vapor pressure and the higher the boiling point.

Definition of Boiling Point • The boiling point is defined as the temperature at which the vapor pressure of a liquid becomes equal to the pressure of the gas above the liquid. • The normal boiling point is the temperature at which the vapor pressure becomes equal to standard pressure (101 k. Pa).

Determining Normal Boiling Points Use Table H to determine the normal boiling points of the following substances: 56 o. C • Propanone o. C 79 • Ethanol 100 o. C • Water • Ethanoic acid 118 o. C

Boiling Point depends on the pressure on the liquid • The bubbles formed when a liquid boils cannot form when the pressure on the surface of the liquid is greater than the pressure of the gas in the bubble.
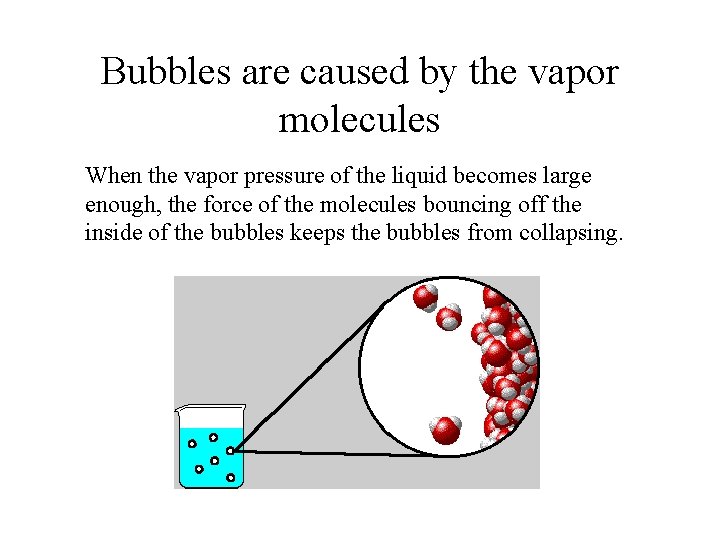
Bubbles are caused by the vapor molecules When the vapor pressure of the liquid becomes large enough, the force of the molecules bouncing off the inside of the bubbles keeps the bubbles from collapsing.
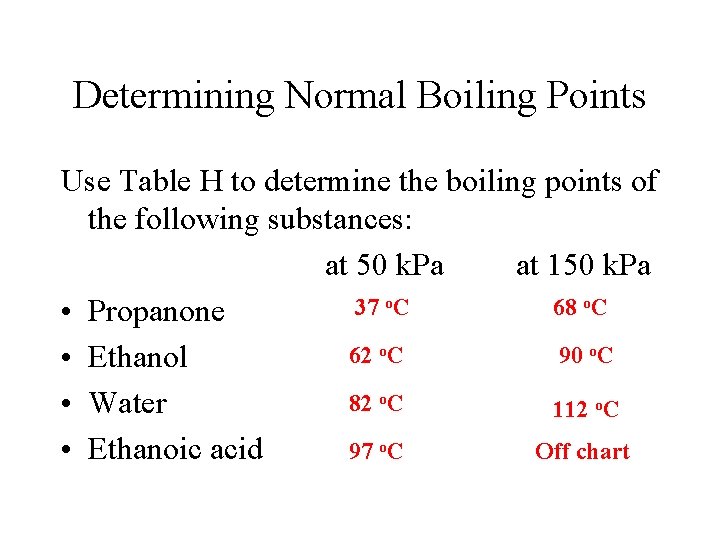
Determining Normal Boiling Points Use Table H to determine the boiling points of the following substances: at 50 k. Pa at 150 k. Pa o. C 37 68 • Propanone 62 o. C 90 o. C • Ethanol 82 o. C • Water 112 o. C 97 o. C Off chart • Ethanoic acid

The Pressure Cooker A pressure cooker cooks faster because it allows the water to be heated to a higher temperature without boiling.

Drying out a “wet” gas Any gas collected over water is a “wet” gas (a mixture of water vapor and the collected gas). According to Dalton’s Law, the total pressure of the gases is equal to the pressure of the collected gas plus the water vapor pressure. Pwet H 2 = PH 2 + Pvapor

Drying Out a Wet Gas - 2 The temperature determines the vapor pressure of the liquid (see Table H). If the total pressure is measured the pressure of the “dry” gas can be calculated by rearranging Dalton’s Law. Pwet H 2 = PH 2 + Pvapor PH 2 = P wet H 2 - P vapor

Vapor Pressure Problems 1. A sample of nitrogen gas is collected over water at 50 o. C. The pressure of the “wet” gas is found to be 98 k. Pa and the water vapor pressure is 12 k. Pa (see Table H). What is the pressure of just the nitrogen gas (the “dry” nitrogen)? 86 k. Pa 2. A sample of helium is collected over water at 75 o. C. If the helium exerts a pressure of 120 k. Pa, use Table H to determine the water vapor pressure, and the pressure of the “wet” Helium. Pvapor = 38 k. Pa P wet He = 158 k. Pa

How did this happen? Click here for more pictures and more details.

Avogadro’s Hypothesis • Under the same conditions of temperature and pressure, equal volumes of gases contain the same number of molecules.

Molar Volume As a consequence of Avogadro’s Hypothesis, the molar volume of any gas at STP is 22. 4 Liters

Ideal Gases Strictly speaking, the gas laws that we have discussed work perfectly only for IDEAL GASES Ideal gases are gases that conform perfectly to the kinetic molecular theory. These gases would have • No intermolecular attractions • No molecular volumes There are no ideal gases in nature!

Real gases are those gases that exist in nature (N 2, O 2, CO 2, etc). These gases differ from Ideal gases in that they have • Intermolecular attractions • Their molecules take up space (have volume)

Real Gases act Ideal most of the time The concept of ideal gases is useful because even though there are no ideal gases, real gases act like they’re ideal most of the time. Real gases stop acting like ideal gases under the conditions when they become liquids.
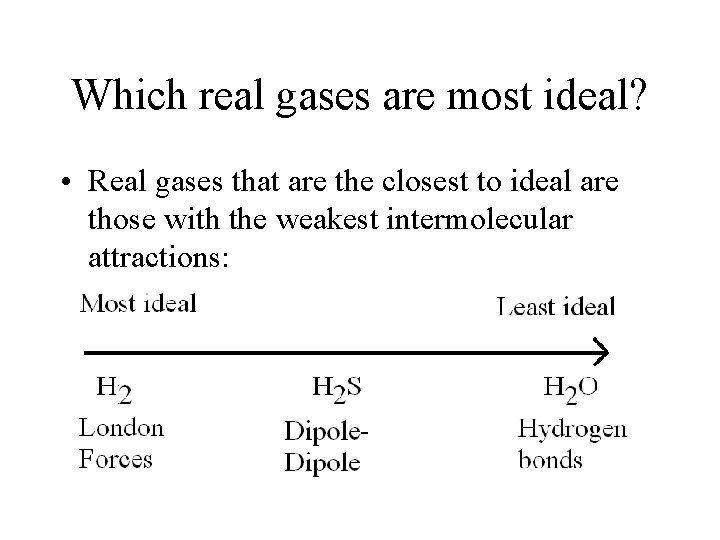
Which real gases are most ideal? • Real gases that are the closest to ideal are those with the weakest intermolecular attractions:
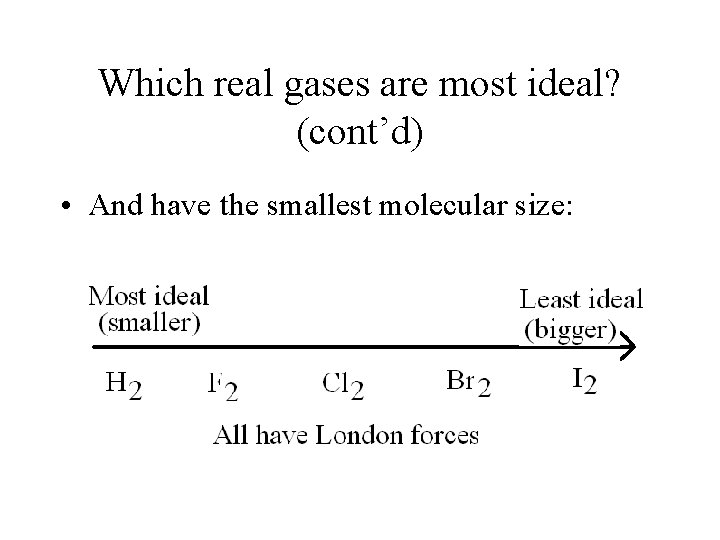
Which real gases are most ideal? (cont’d) • And have the smallest molecular size:

Practice 1. What is the most ideal noble gas? What is the least ideal noble gas? Most – He Least - Rn 2. Which of the following gases would be most ideal? (and why? ) a) N 2 or CO b) CH 4 or NH 3 c) HF or Ne a) N 2 (non-polar vs dipole) b) CH 4 (non-polar vs H bond) c) Ne (non-polar vs H bond)

When do real gases deviate from ideal behavior? Real gases act pretty much like ideal gases predict as long as they are gases. They stop acting like ideal gases when they become liquids. That is at 1. Low temperatures 2. High pressure

When are real gases most ideal? Real gases act most like ideal gases when they are at 1. High temperature 2. Low pressure Under these conditions the weak intermolecular attractions and molecular volumes become insignificant.

Practice 1. At which temperature would water vapor be most ideal? a) 0 K b) 250 K c) 0 o. C d) 250 o. C 2. At which pressure would water vapor be most ideal? a) 10 k. Pa b) 100 k. Pa c) 10 atm d) 10 k. Pa

More Practice Use the School Island assignment on Gases to get more practice at doing these problems.
- Slides: 66