The Gas Laws Mathematical relationships between volume temperature

The Gas Laws Mathematical relationships between volume, temperature, pressure & amount of gas

Variables that Affect Behavior l Number of particles (n) l Pressure l Volume (P) (V) l Temperature (T)

Standard Temperature and Pressure STP l Standard Temperature 0 o. C or 273 K [Remember: K = o. C + 273] l Standard Pressure 1 atm 760 mm. Hg 760 torr 101. 3 k. Pa STP

Boyles Law Constant Variables: l Number of particles (amount) l Temperature Formula: P 1 V 1 = P 2 V 2 Volume of gas is inversely proportional to the pressure l V= P l
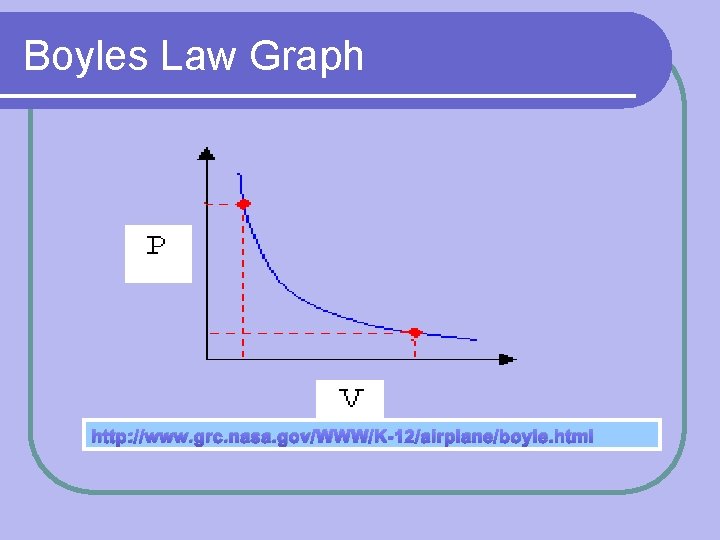
Boyles Law Graph http: //www. grc. nasa. gov/WWW/K-12/airplane/boyle. html

Boyle’s Practice P 1 V 1 = P 2 V 2 A gas occupies a volume of 259 cm 3 at 112 k. Pa. What volume will gas occupy at standard pressure? V 1 = P 1 = V 2 = P 2 =

Charles’ Law Constant Variables: l Number of particles (amount) l Pressure V 1 Formula: l Volume T 1 = V 2 T 2 of gas is proportional to temperature l T= V
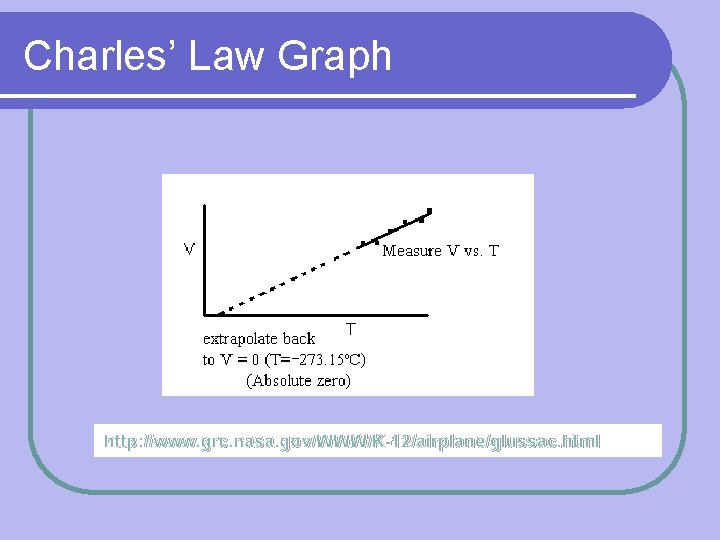
Charles’ Law Graph http: //www. grc. nasa. gov/WWW/K-12/airplane/glussac. html

*VERY IMPORTANT* Temperature MUST, MUST be in KELVIN

V 1 Charles Practice T 1 = V 2 T 2 A gas occupies 907 cm 3 at 19. 0 o. C. If the pressure remains constant what volume will the gas occupy at standard temperature? V 1 = T 1 = V 2 = T 2 =

Gay-Lussac’s Law Constant Variables: l Number of particles (amount) l Volume Formula: l Pressure P 1 T 1 = P 2 T 2 is proportional to temp (K) l P= T
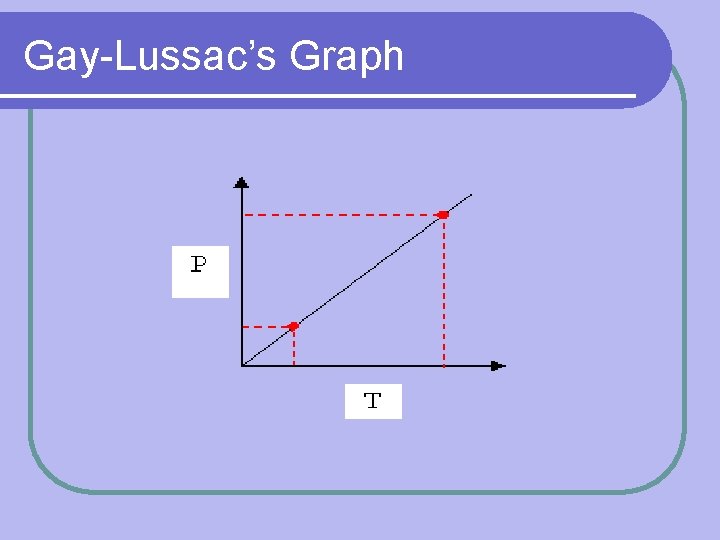
Gay-Lussac’s Graph

*VERY IMPORTANT* Temperature MUST, MUST be in KELVIN

Gay-Lussac Practice P 1 T 1 = P 2 T 2 A gas at 715 mm. Hg has a temperature of 25. 0 o. C. If the volume is held constant, what will the temperature of the gas be at standard pressure? T 1 = T 2 = P 1 = P 2 =

Combined Gas Law l Combination of all three laws l Based on fact that gases generally undergo changes in T, V and P simultaneously Formula: l Remember: Temp in Kelvin

P 1 V 1 Combined Practice T 1 = P 2 V 2 T 2 A gas occupies 51. 7 m. L at 27. 0 o. C and 90. 9 k. Pa. What volume will the gas occupy at STP? V 1 = V 2 = T 1 = P 1 = T 2 = P 2 =

Website l http: //www. blackgold. ab. ca/ict/Division 4/ Science/Div. %204/Boyles%20 Law/boyle slaw. htm http: //videos. howstuffworks. com/hsw/12267 -investigating-chemical -properties-scuba-diving-video. htm
- Slides: 17