II The Gas Laws A Boyles Law PV

II. The Gas Laws

A. Boyle’s Law PV = k P V

A. Boyle’s Law • The pressure and volume of a gas are inversely related oat constant mass & temp PV = k P V

Example Problems pg 335 # 10 &11 1. The pressure on 2. 50 L of anesthetic gas changes from 105 k. Pa to 40. 5 k. Pa. What will be the new volume if the temp remains constant? P 1 = 105 k. Pa V 1 = 2. 5 L P 2 = 40. 5 k. Pa V 2 = ? P 1 × V 1 = P 2 × V 2 (105) (2. 5) = (40. 5)(V 2) 262. 5 = 40. 5 (V 2) 6. 48 L = V 2

Example Problems pg 335 # 10 &11 2. A gas with a volume of 4. 00 L at a pressure of 205 k. Pa is allowed to expand to a volume of 12. 0 L. What is the pressure in the container if the temp remains constant? P 1 = 205 k. Pa P 2 = ? V 1 = 4. 0 L V 2 = 12. 0 L P 1 × V 1 = P 2 × V 2 (205) (4. 0) = (P 2)(12) 820 = (P 2) 12 68. 3 k. Pa = P 2

B. Charles’ Law V T

B. Charles’ Law • The volume and absolute temperature (K) of a gas are directly related o at constant mass & pressure V T

Example Problems pg. 337 # 12 & 13 3. If a sample of gas occupies 6. 80 L at 325°C, what will be its volume at 25°C if the pressure does not change? V 1= 6. 8 L V 2 = ? T 1 = 325°C = 598 K T 2 = 25°C = 298 K 6. 8 = V 2 598 298 6. 8 X 298 = V 2 X 598 2026. 4 = V 2 X 598 3. 39 L = V 2

Example Problems pg. 337 # 12 & 13 4. Exactly 5. 00 L of air at -50. 0°C is warmed to 100. 0°C. What is the new volume if the pressure remains constant? V 1= 5. 0 L T 1 = -50°C = 223 K 5 = V 2 223 373 5 X 373 = V 2 X 223 1865 = V 2 X 223 8. 36 L = V 2 = ? T 2 = 100°C = 373 K

C. Gay-Lussac’s Law P T

C. Gay-Lussac’s Law • The pressure and absolute temperature (K) of a gas are directly related o at constant mass & volume P T

Example Problems 5. The gas left in a used aerosol can is at a pressure of 103 k. Pa at 25°C. If this can is thrown onto a fire, what is the pressure of the gas when its temperature reaches 928°C? P 1= 103 k. Pa T 1 = 25°C = 298 K 103 = P 2 298 1201 298 × P 2 = 123, 703 P 2 = 415 k. Pa P 2 = ? T 2 = 928°C = 1201 K

Example Problem pg. 338 # 14 6. A gas has a pressure of 6. 58 k. Pa at 539 K. What will be the pressure at 211 K if the volume does not change? P 1= 6. 58 k. Pa T 1 = 539 K 6. 58 = P 2 539 211 6. 58 X 211 = P 2 X 539 1388. 38 = P 2 X 539 2. 58 k. Pa = P 2 = ? T 2 = 211 K

D. Avogadro’s Law • Under the same condition of temperature and pressure, equal volumes of all gases contain the same number of molecules.
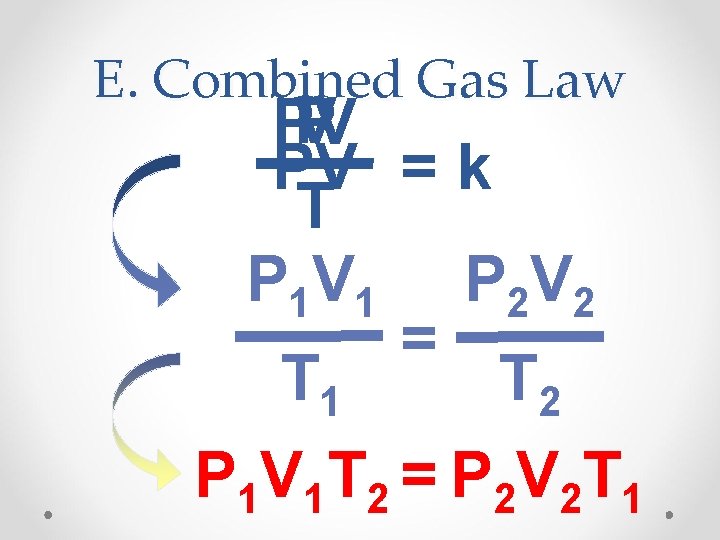
E. Combined Gas Law P V PV PV = k T P 1 V 1 P 2 V 2 = T 1 T 2 P 1 V 1 T 2 = P 2 V 2 T 1

F. Gas Law Problems • A gas occupies 473 cm 3 at 36°C. Find its volume at 94°C. CHARLES’ LAW GIVEN: T V V 1 = 473 cm 3 T 1 = 36°C = 309 K V 2 = ? T 2 = 94°C = 367 K WORK: P 1 V 1 T 2 = P 2 V 2 T 1 (473 cm 3)(367 K)=V 2(309 K) V 2 = 562 cm 3

F. Gas Law Problems • A gas occupies 100. m. L at 150. k. Pa. Find its volume at 200. k. Pa. BOYLE’S LAW GIVEN: P V V 1 = 100. m. L P 1 = 150. k. Pa V 2 = ? P 2 = 200. k. Pa WORK: P 1 V 1 T 2 = P 2 V 2 T 1 (150. k. Pa)(100. m. L)=(200. k. Pa)V 2 = 75. 0 m. L

F. Gas Law Problems • A gas occupies 7. 84 cm 3 at 71. 8 k. Pa & 25°C. Find its volume at STP. COMBINED GAS LAW GIVEN: P T V WORK: V 1 = 7. 84 cm 3 P 1 V 1 T 2 = P 2 V 2 T 1 P 1 = 71. 8 k. Pa (71. 8 k. Pa)(7. 84 cm 3)(273 K) T 1 = 25°C = 298 K =(101. 325 k. Pa) V 2 (298 K) V 2 = ? P 2 = 101. 325 k. Pa V 2 = 5. 09 cm 3 T 2 = 273 K

F. Gas Law Problems • A gas’ pressure is 765 torr at 23°C. At what temperature will the pressure be 560. torr? GAY-LUSSAC’S LAW GIVEN: P T WORK: P 1 = 765 torr P 1 V 1 T 2 = P 2 V 2 T 1 = 23°C = 296 K (765 torr)T 2 = (560. torr)(309 K) P 2 = 560. torr T 2 = 226 K = -47°C T 2 = ?
- Slides: 19