Review Boyles Law Deals with which 2 characteristics

Review

Boyle’s Law • Deals with which 2 characteristics of gases? • What’s the equation?

Boyle’s Law • The volume of a gas at 99. 0 k. Pa is 300. 0 m. L. If the pressure is increased to 188 k. Pa, what will be the new volume? 99. 0 x 300 = 29, 700 ? X 188 = 29, 700/ 188 = 157. 9787

Other Gas Laws

Charles’s Law the relationship between temperature and volume of a gas
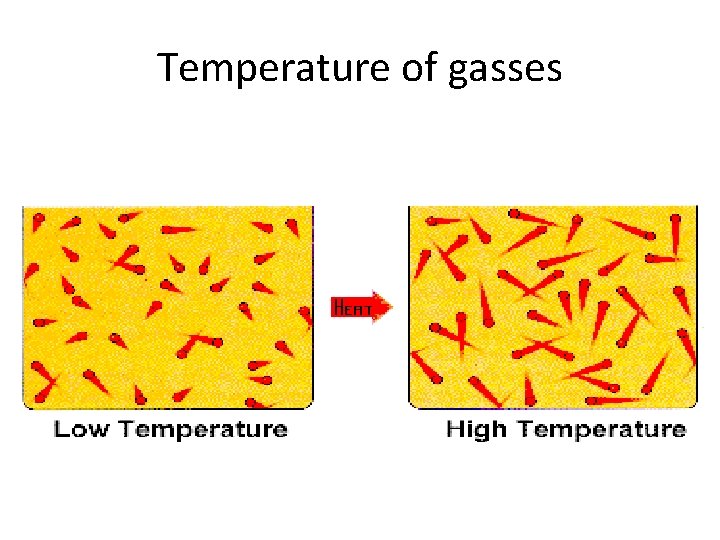
Temperature of gasses

Charles’s Law • the volume of a gas is directly proportional to its temperature French Physicist Jacques Charles (1746 -1823)
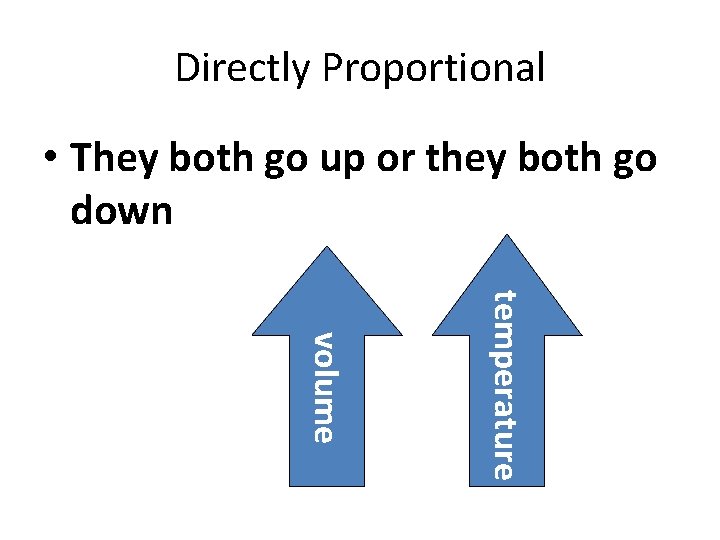
Directly Proportional • They both go up or they both go down temperature volume

Charles’s Law at work…
Constants • Amount and pressure

Examples of Charles Law at work: http: //www. youtube. com/w atch? v=s. Ebx. Lr. P_ZCU
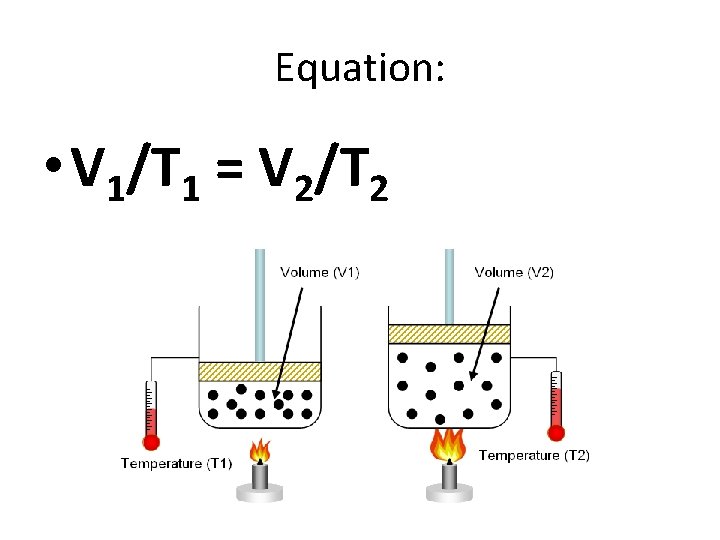
Equation: • V 1/T 1 = V 2/T 2

Charles’s Law Illustrated 0. 963/263 =. 00366 1. 000/273 =. 00366 1. 037/283 =. 00366

Charles’s Law • A sample of Carbon dioxide in a pump has volume of 20. 5 m. L and it is at 40. 0 o. C. When the amount of gas and pressure remain constant, find the new volume of Carbon dioxide in the pump if temperature is increased to 65. 0 o. C. 20. 5/313 = V₂/338

Try some more V 1/T 1 = V 2/T 2 • 150 m. L / 300 K = 237 ml / X(K) • 170 m. L/ 32 C = 193 m. L/ X(K) • 300 m. L/ 300 K = 400 m. L / X(C

Gay-Lussac’s Law the relationship between temperature and pressure of a gas
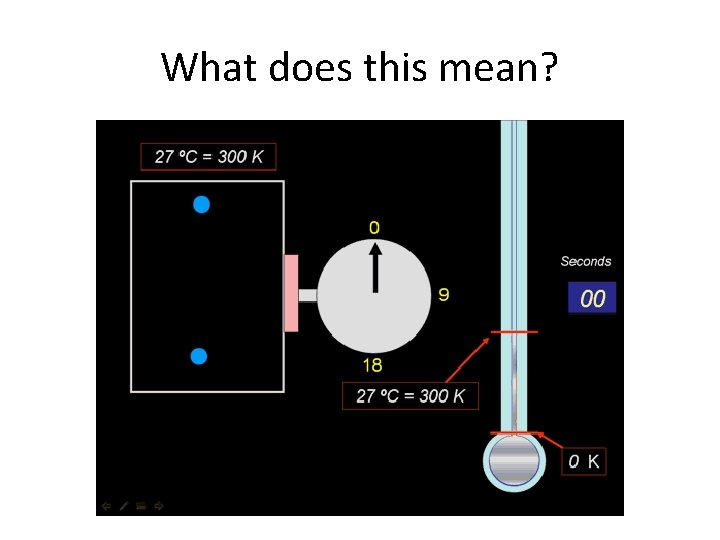
What does this mean?

Gay-Lussac’s Law • the pressure of a gas is directly proportional to its temperature French Chemist and Physicist Joseph Louis Gay-Lussac (1778– 1850)
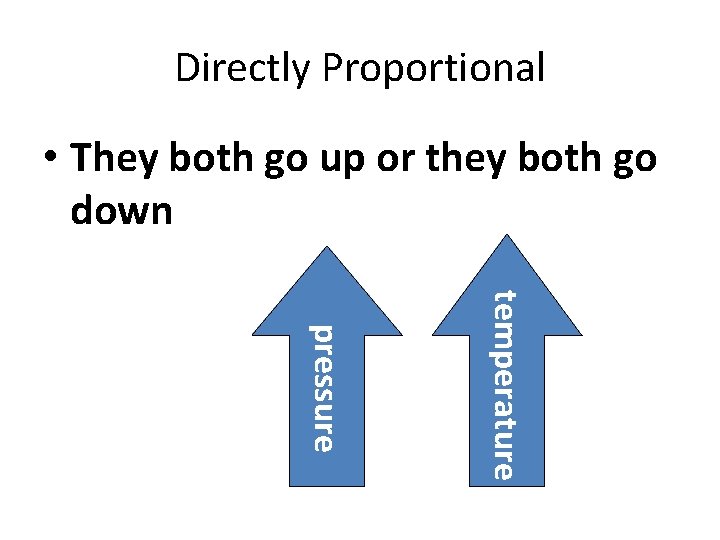
Directly Proportional • They both go up or they both go down temperature pressure

Constants • Amount and Volume

Gay-Lussac’s Law
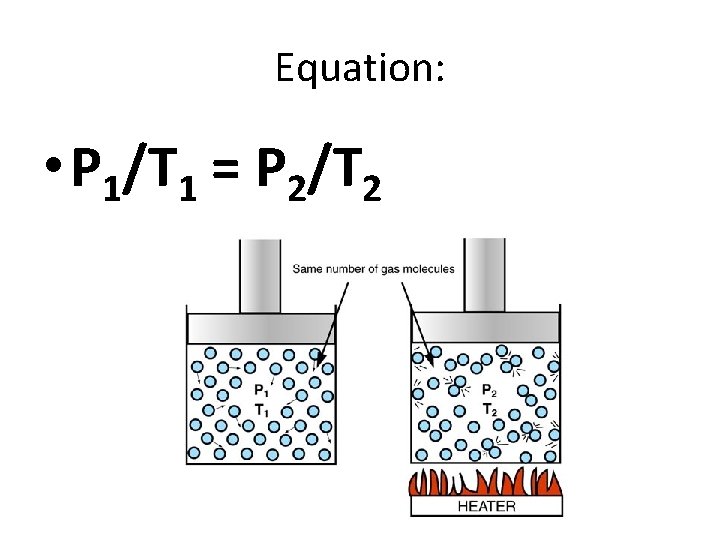
Equation: • P 1/T 1 = P 2/T 2
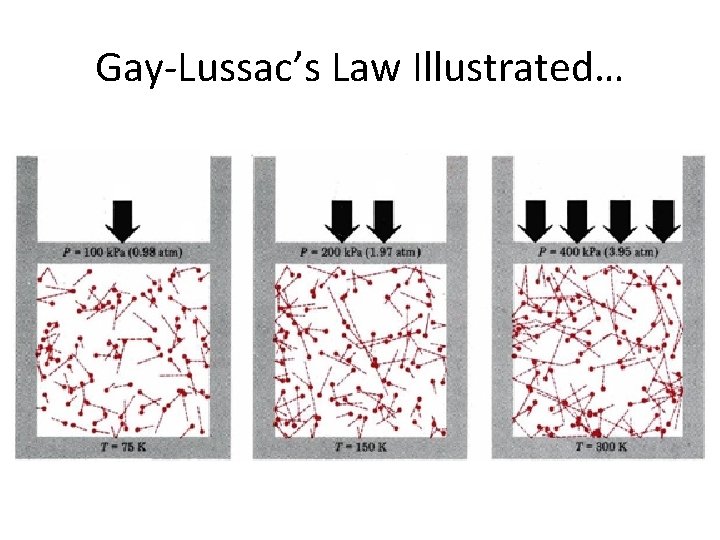
Gay-Lussac’s Law Illustrated…

Gay-Lussac’s Law • A gas has a pressure of 125 k. Pa at a temperature of 303 K. If the pressure in the container is increased to 201 k. Pa, what is the new temperature? 125/303 = 201/ T₂ 487 K

Try some more P 1/T 1 = P 2/T 2 • 2. 5 atm/ 300 K = 6. 5 atm/ X • 1. 88 atm/ 25°C = X/ 37°C • 1. 12 atm/ X = 2. 56 atm/ 36. 5°C


Question 1 Based on Boyle’s Law (p * V = constant), when the number of moles (n) and temperature (T) are held constant, pressure and volume are: a. Inversely proportional: if one goes up, the other comes down. b. Directly proportional: if one goes up, the other goes up. c. Not related

Question 2 Based on Charles’ Law (V / T = constant) when the number of moles (n) and pressure (p) are held constant, volume and temperature are: a. Inversely proportional: if one goes up, the other comes down. b. Directly proportional: if one goes up, the other goes up. c. Not related

Question 3 • Which gas law do you need to use to solve the problem: A gas at 20°C occupies 1. 00 L. Assuming pressure remains constant, what volume will it occupy at 30. 0°C? a. Boyle’s Law b. Charles’s Law c. Gay-Lussac’s Law

Question 3 Lets put the Ideal Gas Law (p*V=n*R*T) to some practical use. To inflate a tire of fixed volume, what is the most effective way to increase the pressure in the tire? a. Increase the force pressing on the outside of the tire. b. Increase the temperature of the gas (air) in the tire. c. Increase the amount (number of moles) of gas in the tire.
- Slides: 30