The Gas Laws Boyles Law Charles Law GayLussacs

The Gas Laws Boyle’s Law Charles’ Law Gay-Lussac’s Law

Behavior of Gases 16. 3 Pressure • Pressure is the amount of force exerted per unit of area, or P = F/A. • A balloon and a bicycle tire are considered to be containers. • They remain inflated because of collisions the air particles have with the walls of their container.

Behavior of Gases 16. 3 Pressure • This collection of forces, caused by the collisions of the particles, pushes the walls of the container outward. • If more air is pumped into the balloon, the number of air particles is increased. • This causes more collisions with the walls of the container, which causes it to expand.

Behavior of Gases 16. 3 Pressure • Pressure is measured in a unit called Pascal (Pa), the SI unit of pressure, but we’ll use k. Pa, or kilo. Pascals. • At sea level, atmospheric pressure is 101. 3 k. Pa. • Unit for pressure = k. Pa

Behavior of Gases 16. 3 Boyle’s Law • What happens to the gas pressure if you decrease the size (volume) of the container? • If you squeeze gas into a smaller space, its particles will strike the walls more often giving an increased pressure. The opposite is true, too. • Lower volume = higher pressure • Higher volume = lower pressure

Behavior of Gases 16. 3 Boyle’s Law • Robert Boyle (1627 -1691), a British scientist, described this property of gases. • According to Boyle’s law, if you decrease the volume of a container of gas and hold the temperature constant, the pressure of the gas will increase. • An increase in the volume of the container causes the pressure to drop, if the temperature remains constant.
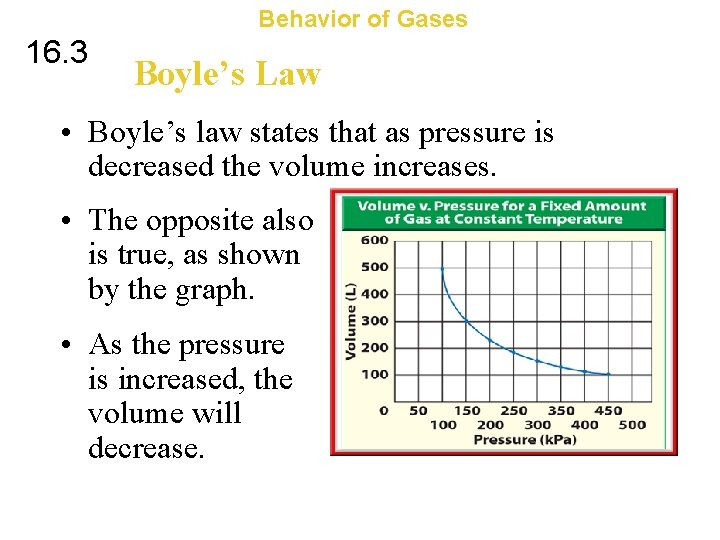
Behavior of Gases 16. 3 Boyle’s Law • Boyle’s law states that as pressure is decreased the volume increases. • The opposite also is true, as shown by the graph. • As the pressure is increased, the volume will decrease.

Behavior of Gases 16. 3 Boyle’s Law in Action • When Boyle’s law is applied to a real life situation, we can predict the new volume or new pressure of a gas mathematically.

Behavior of Gases 16. 3 Boyle’s Law in Action • You can use the equation P 1 V 1 = P 2 V 2 to express this mathematically. • P 1 = initial pressure (in k. Pa) • V 1 = initial volume (in L) • P 2 = new pressure (in k. Pa) • V 2 = new volume (in L)

Example • 15 L of gas has a pressure of 3 k. Pa. The pressure is increased to 5 k. Pa. What is the new volume of the gas?

Behavior of Gases 16. 3 Charles’s Law • Jacques Charles (1746 -1823) was a French scientist who studied gases. • According to Charles’s law, the volume of a gas increases with increasing temperature, as long as pressure does not change.
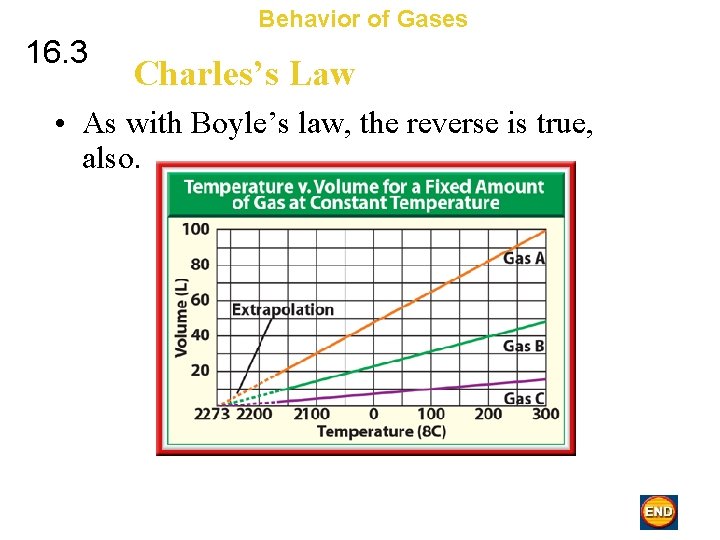
Behavior of Gases 16. 3 Charles’s Law • As with Boyle’s law, the reverse is true, also.

Behavior of Gases 16. 3 Charles’s Law • Charles’s law can be explained using the kinetic theory of matter. • As a gas is heated, its particles move faster and its temperature increases. • Because the gas particles move faster, they begin to strike the walls of their container more often and with more force.

Behavior of Gases 16. 3 Using Charles’s Law • The formula that relates the variables of temperature to volume shows a direct relationship, V 1/T 1 = V 2/T 2, when temperature is given in Kelvin (pressure remains constant). • To convert from Celsius to Kelvin, simply add 273 to the Celsius temperature. • Example: 10 C + 273 = 283 K

Behavior of Gases 16. 3 Using Charles’s Law • What would be the resulting volume of a 2. 0 -L balloon at 25. 0 C that was placed in a container of ice water at 3. 0 C?

Behavior of Gases 16. 3 Using Charles’s Law • As Charles’s law predicts, the volume decreased as the temperature of the trapped gas decreased.

Behavior of Gases 16. 3 The Pressure-Temperature Relationship • What happens if you heat an enclosed gas? The particles of gas will strike the walls of the canister more often. Why? • If the pressure becomes greater than the canister can hold, it will explode. • At a constant volume, an increase in temperature results in an increase in pressure.

Pressure and Temperature • Joseph Louis Gay-Lussac was a French professor who studied the behavior of gases with many scientists. • In the early 1800 s, he observed a relationship between the pressure and temperature of gases.

Gay-Lussac’s Law • Just like Charles’ law, Gay-Lussac’s law describes a direct relationship between two variables (as one increases, so does the other). • According to the law, as the temperature of a gas increases, so does the pressure (as long as the volume remains constant). As the temperature decreases, so does the pressure (as long as the volume remains constant).

Mathematically speaking… • We can look at this law mathematically. • P 1/T 1 = P 2/T 2 • P 1 = initial pressure (in k. Pa) • T 1 = initial temperature (in K) • P 2 = new pressure (in k. Pa) • T 2 = new temperature (in K)

Example • A gas in an aerosol can has a temperature of 30 C and a pressure of 3 k. Pa. If the temperature is increased to 45 C, what will the new pressure of the gas be? • Don’t forget to convert C to K first.

Section Check 16. 3 Question 1 What would be the resulting volume of a 3. 0 -L balloon at 25. 0º C that was placed in a container of ice water at 4. 0º C, if pressure is constant? A. 2. 8 L B. 3. 0 L C. 4. 8 L D. 5. 0 L

Section Check 16. 3 Answer The answer is A. Use the formula that relates volume to temperature given in Kelvin, V 1/T 1 = V 2/T 2. In this case, V 1 = 3. 0 L, T 1 = 25. 0º C + 273 = 298º K, T 2 = 4. 0º C + 273 = 277º K. Solving for V 2 gives 2. 8 L.

Section Check 16. 3 Question 2 The SI unit of pressure is the pascal, but we use the _____. A. B. C. D. coulomb tesla Watt kilo. Pascal

Section Check 16. 3 Answer The answer is D. The SI unit of pressure is the Pascal; we measure pressures in kilo. Pascals.
- Slides: 25