GAS LAWS DAY 1 Ideal Boyles Lussac Charles

GAS LAWS DAY 1 Ideal Boyles Lussac Charles Combined

When doing word problems…. 1. Find the question word: determine what you are looking for. 2. What #s did the problem give you 3. If only one # always start grid with that # 4. If Multiple #’s you need a formula

Variables that describe a Gas P= Pressure T = Temperature n= Amount V= Volume

Ideal Gas Law PV =n. RT R = gas constant R = 8. 31 L k. Pa mole K R= 62. 4 mm. Hg L mole K R= 0. 0821 L atm mole K all other units can be converted

PRACTICE Gas Law Worksheet

1. Calculate the pressure, in atmospheres, exerted by each of the following: a. 250 L of gas containing 1. 35 moles at 320 K b. 4. 75 L of gas containing 0. 86 moles at 300 K.

2. Calculate the volume, in liters, occupied by each of the following: a. 4 grams of H 2 at 300 K and 1. 25 atm. b. 0. 425 moles of ammonia gas (NH 3) at 10. 5 PSI and 37 C.

3. Determine the amount: a. In moles, of 1. 25 L of O 2 at 805. 6 mm. Hg and 250 K. b. In grams, of 0. 80 L of ammonia gas at 0. 925 atm and 27 C. Finish for homework: answer: 0. 51 g

GAS LAWS
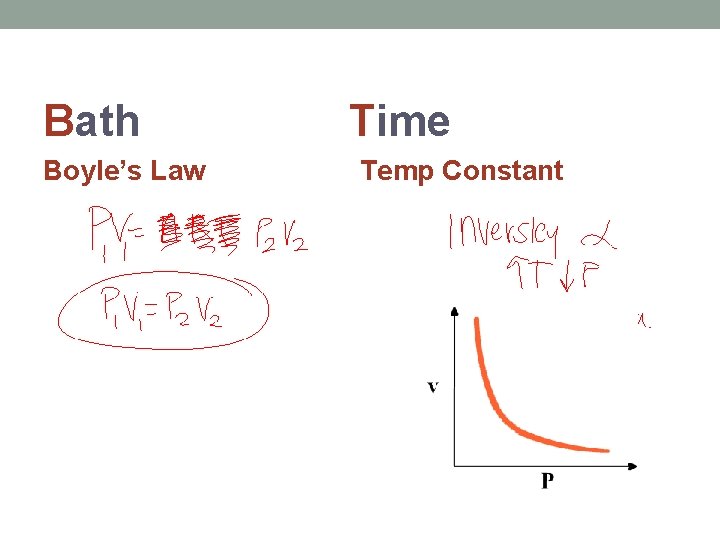
Bath Boyle’s Law Time Temp Constant
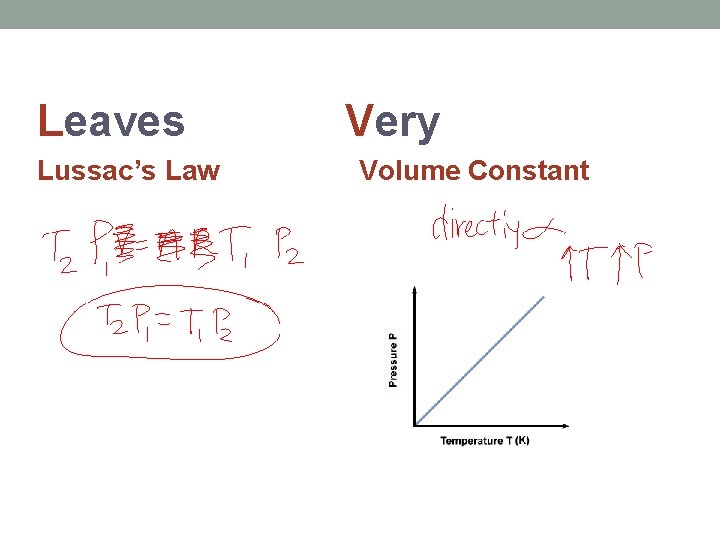
Leaves Lussac’s Law Very Volume Constant
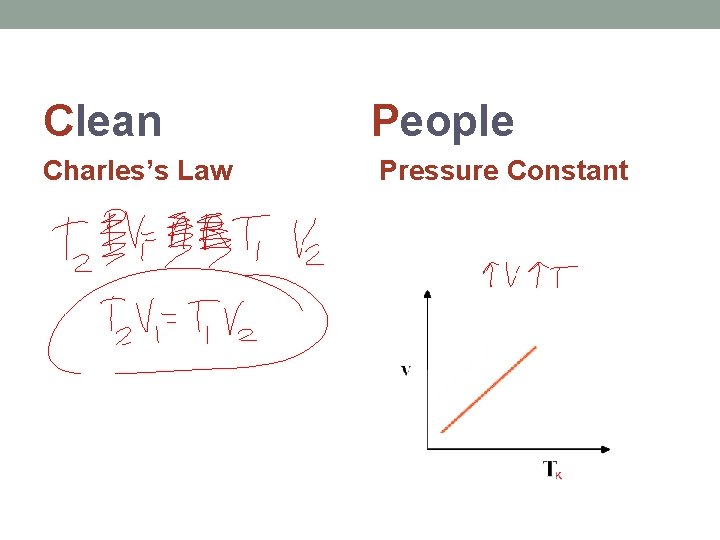
Clean People Charles’s Law Pressure Constant

Combined Gas Law

4. A 550. 0 m. L sample of nitrogen gas is warmed from 77. 0 C to 80. 0 C. Find the new volume if the pressure remains constant. What law does this situation follow?

5. Convert 338 L at 6. 38 x 103 k. Pa to its new volume at standard pressure. What law does this situation follow?
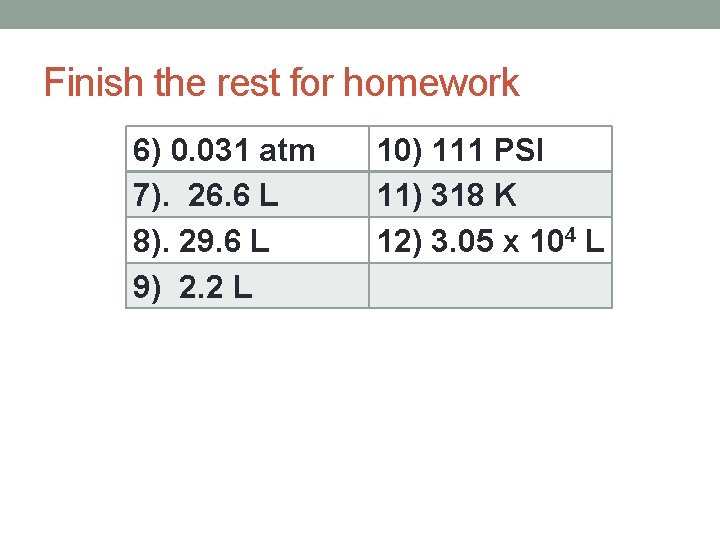
Finish the rest for homework 6) 0. 031 atm 7). 26. 6 L 8). 29. 6 L 9) 2. 2 L 10) 111 PSI 11) 318 K 12) 3. 05 x 104 L
- Slides: 16