THE GAS LAWS AVOGADROS BOYLES CHARLESS GAYLUSSACS AND

THE GAS LAWS AVOGADRO’S, BOYLE’S, CHARLES’S, GAY-LUSSAC’S AND COMBINED GAS LAWS

Kinetic Theory of Gases Particles of a gas ª are in constant random motion. ª lose no energy in their collisions, meaning the collisions are perfectly elastic. ª are very far apart with no attractive or repulsive forces.

Four Parameters Define a Gas ªMOLES ªPRESSURE ªVOLUME ªTEMPERATURE

Avogadro’s Law The volume of a gas is directly proportional to the number of moles of gas in the container.

Pressure ª FORCE OF THE PARTICLES HITTING THE CONTAINER ª MEASURED WITH BAROMETERS OR GUAGES ª Units: pounds per square inch, atmospheres, millimeters of mercury or torr, kilopascals ª 14. 7 psi = 1 atm = 760 mm. Hg = 101. 3 k. Pa

Volume ª THE SPACE OCCUPIED BY A SAMPLE OF MATTER ª GASES OCCUPY THE ENTIRE SPACE OF THE CONTAINER IN WHICH THEY ARE HELD ª UNITS: Liters, milliliters, decimeter 3, centimeter 3 ª 1. 00 L = 1000 ml = 1. 00 dm 3 = 1000 cm 3

Temperature ª A MEASURE OF THE AVERAGE KINETIC ENERGY OF A SAMPLE OF MATTER ª MEASURED WITH A THERMOMETER ª UNITS: KELVIN, FARHENHEIT AND CELSIUS ª K = C + 273 = (F - 32) + 273 1. 8

Boyle’s Law ª STATES THAT THE VOLUME OF A FIXED AMOUNT OF GAS VARIES INVERSELY WITH THE PRESSURE AT CONSTANT TEMPERATURE P V
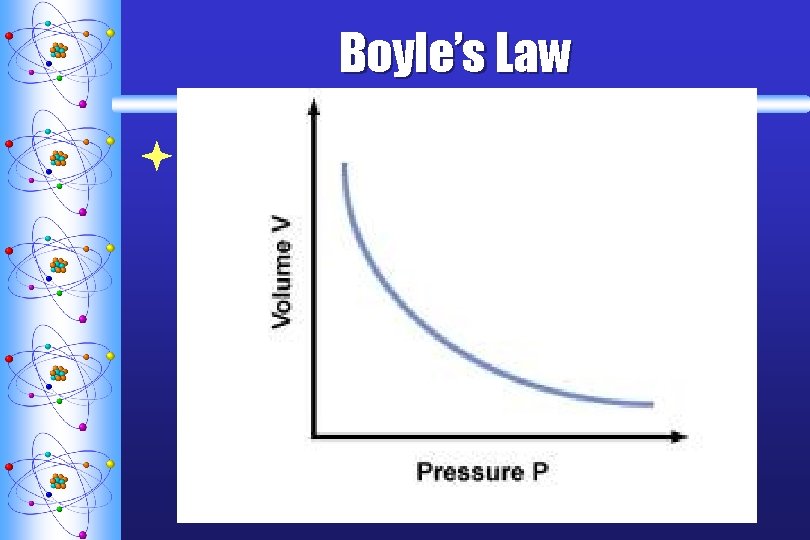
Boyle’s Law ªEquation: P 1 V 1 = P 2 V 2 when the T is constant

Sample Problem ª A 500. 0 ml can of gas with a pressure of 20. 0 k. Pa is run over by a garbage truck and flattened to a volume of 10. 0 ml. What is the new pressure of the gas inside the container? THINK AND SOLVE!!!

Charles’s Law ª STATES THAT THE VOLUME OF A GIVEN AMOUNT OF GAS IS DIRECTLY PROPORTIONAL TO THE KELVIN TEMPERATURE AT CONSTANT PRESSURE T V
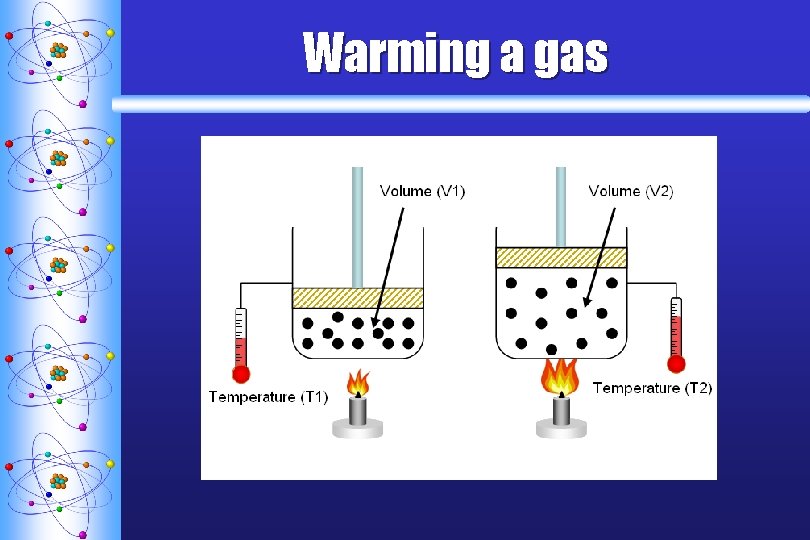
Warming a gas
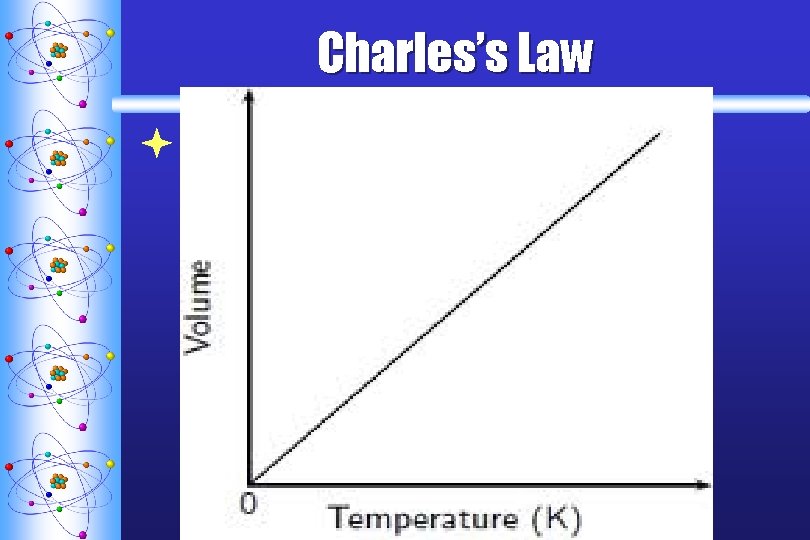
Charles’s Law ªEquation: V 1 V 2 = T 1 T 2 when the P is constant T must be in Kelvin Liquid Nitrogen Video

Sample Problem ª A helium balloon in a closed car occupies a volume of 2. 23 L at 40. 0 C. If the car is parked on a hot day and the temperature inside rises to 75. 0 C, what is the new volume of the balloon if the pressure remains constant? THINK AND SOLVE!!!

Gay-Lussac’s Law ª STATES THAT THE PRESSURE OF A FIXED AMOUNT OF GAS VARIES DIRECTLY WITH THE KELVIN TEMPERATURE WHEN THE VOLUME REMAINS CONSTANT T P
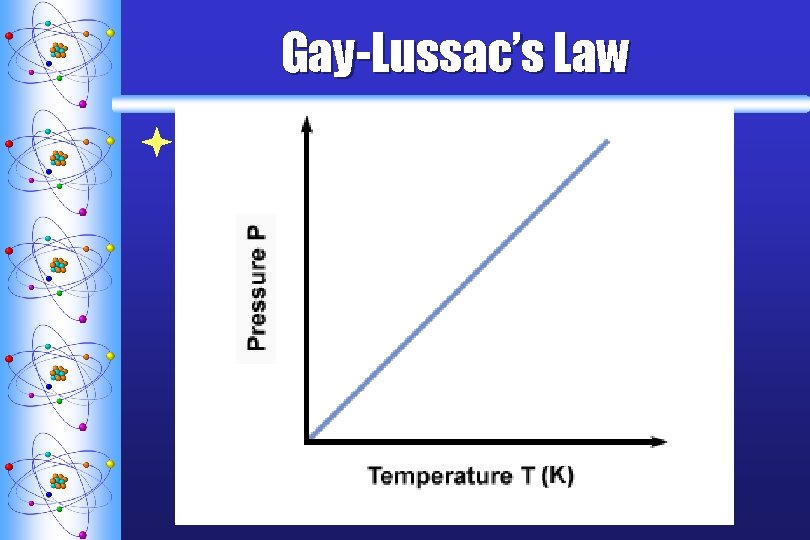
Gay-Lussac’s Law ªEquation: P 1 P 2 = T 1 T 2 when the V is constant Temperature must be in Kelvin!!!!!!

Sample Problem ª The pressure of oxygen gas inside a canister is 5. 00 atm at 25. 0 C. The canister is located at a camp high on Mount Everest. If the temperature there falls to -10. 0 C, what is the new pressure inside the canister? THINK AND SOLVE!!!

Combined Gas Law ª STATES THE RELATIONSHIP BETWEEN THE TEMPERATURE, VOLUME, AND PRESSURE OF A FIXED SAMPLE OF GAS ª THERE IS A CHANGE IN ALL THREE PARAMETERS THAT DEFINE THE GAS

Combined Gas Law ªEquation: P 1 V 1 P 2 V 2 = T 1 T 2 Temperature must be in Kelvin!!!!!!

Sample Problem ª A gas at 110 k. Pa and 30. 0 C fills a flexible container with an initial volume of 2. 00 liters. If the temperature is raised to 80. 0 C and the pressure increases to 440 k. Pa, what is the new volume? THINK AND SOLVE!!!

Avogadro’s Principle ª Equal volumes of gases at the same temperature and pressure have the same number of particles (and moles of particles)

Standard Conditions ª Standard Temperature and Pressure is known as STP ª Standard Temperature is 0. 00 C or 273 Kelvin ª Standard Pressure is 1. 00 atm or 760 mm. Hg or 101. 3 k. Pa or 14. 7 psi (all equal, but different units)

Molar Volume ª The volume occupied by 1. 00 mole of any gas at STP is 22. 4 liters ª This can be used as yet another conversion factor in chemistry 22. 4 L / 1. 00 mole or 1. 00 mole / 22. 4 L ª Keep in mind that this also represents a certain number of particles: 6. 02 x 1023 atoms or molecules / 1. 00 mole

Sample Problems ª How many kilograms of CO 2 are there in 50. 0 L at STP? ª How many L of N 2 are there in 12, 345 mg at STP?

Ideal Gas Law ª Relates moles to pressure, volume and temperature under any conditions:

What is R? ª R is the Ideal Gas Constant ª It is expressed in many different ways!!!!!

Sample Problems ª How many moles of ammonia, NH 3, are present in a 3. 0 L vessel at 300. 0 Kelvin and 1. 50 atm? ª How many grams of CO 2 are there in a 50. L container at 25 C and 105 k. Pa?

Gas Density ª Density is the ratio of mass to volume for any sample ª Equation: D = m V Find the mass and divide by the volume!!!! Gas densities are LOW!!

Sample Problem ª What is the density of a sample of N 2 that exerts a pressure of 5. 30 atm in a 3. 50 L container at 125 C?

Molar Mass of the Gas ª Molar mass (M) is the ratio of grams to moles for a substance ª Equation: M = mass moles

Sample Problem ª What is the molar mass of a gas if 2. 75 grams occupies 2. 00 L at 1. 08 atm and 15. 0 C?

Real Gases vs. Ideal Gases Real gases deviate from the ideal gas theory in that: ª The particles actually have volume. ª The particles do exhibit intermolecular forces when they are polar gas molecules or larger gas molecules. This behavior happens at very low temperatures and high pressures when the particles are close together.
- Slides: 32