Chapter 12 Gas Laws KMT Objective To describe

Chapter 12: Gas Laws

KMT Objective: To describe the kinetic molecular theory.

Properties of Gases have the following properties: Gases have mass Gases are compressible Gases fill their containers Gases diffuse (spread out) Gases exert pressure

Kinetic Molecular Theory (KMT) KMT is a theory used to explain the behaviors and characteristics of gases. The word “kinetic” refers to motion The work “molecular” refers to molecules

KMT: 3 Basic Assumptions 1. ) A gas is composed of small hard particles with an insignificant volume. 2. ) Gas particles constantly move in straight line paths unless they collide with another particle or the side of the container. 3. ) Gas particles undergo perfectly elastic collisions. This means that no energy is lost or gained. The total kinetic energy remains constant.

The Variables necessary to study the behavior of gases include: Volume Amount of Gas Temperature Pressure

Volume (V) The volume of the gas is simply the volume of the container it is contained in. The metric unit of volume is the liter (L)

Amount of Gas (n) The quantity of as in a given sample expressed in terms of moles of gas.

Temperature (T) The temperature of a gas is generally measured with a thermometer in degrees Celsius. All calculations involving gases should be made after converting the degrees Celsius into Kelvin = ⁰C + 273

Temperature Conversions Convert the following measurements into Kelvin: 1. ) 0 ⁰C 2. ) 50 ⁰ C Convert the following measurement into degrees Celsius: 3. ) 400 K

Temperature Conversions Convert the following measurements into Kelvin: 1. ) 0 ⁰C = 273 K 2. ) 50 ⁰ C = 323 K Convert the following measurement into degrees Celsius: 3. ) 400 K = 127 ⁰C

Pressure (P) The pressure of a gas is the force exerted on the wall of the container a gas is trapped in. There are several units for pressure depending on the instrument used to measure it including: atmospheres (atm) millimeters of Mercury (mm. HG) kilopascal (k. Pa) or Pascal (Pa) torr psi bar

Pressure Conversions We will use the following conversion factors: 1 atm = 101. 325 k. Pa = 101325 Pa = 760 mm. Hg = 760 torr = 14. 7 psi = 1. 013 bar

Pressure Conversions 1. ) Convert 3 atm into torr.

Pressure Conversions 1. ) Convert 3 atm into torr. 3 atm 760 torr = 2280 torr 1 atm

Pressure Conversions 2. ) Convert 38 k. Pa into bar.

Pressure Conversions 2. ) Convert 38 k. Pa into bar. 38 k. Pa 1. 013 bar = 0. 38 bar 1 -1. 325 k. Pa

Pressure Conversions 3. ) Convert 50 mm. Hg into psi.

Pressure Conversions 3. ) Convert 50 mm. Hg into psi. 50 mm. Hg 14. 7 psi = 0. 97 psi 760 mm. Hg

Standard Temperature and Pressure (STP) The behavior of a gas depends very strongly on the temperature and the pressure at which the gas is held. To make it easier to discuss the behavior of gases, it is convenient to designate standard conditions, called STP. At STP, Temperature = 273 K Pressure = 1 atm

Boyle’s Law Objective: To solve problems using Boyle’s Law.

Boyle’s Law Robert Boyle discovered the relationship between the pressure and volume of a gas. He measured the volume of air at different pressures, and observed a pattern which led to his mathematical law. During his experiments, temperature and amount of gas were held constant. http: //en. wikipedia. org/wiki/Robert_Boyle
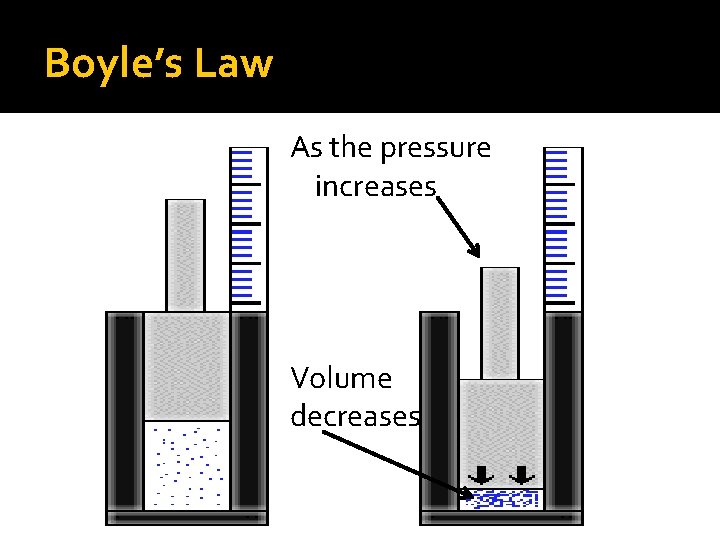
Boyle’s Law As the pressure increases Volume decreases
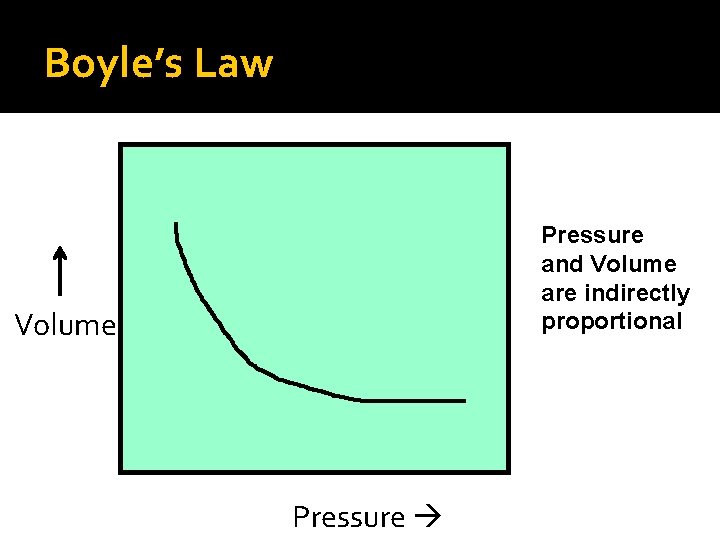
Boyle’s Law Pressure and Volume are indirectly proportional Volume Pressure

Boyle’s Law Boyle’s Mathematical Law: P 1 V 1 = P 2 V 2

Boyle’s Law: Example A gas has a volume of 3. 0 L at 2 atm. its volume at 4 atm? What is

Boyle’s Law: Example A gas has a volume of 3. 0 L at 2 atm. volume at 4 atm? What is its 1. ) Determine which variables you have P 1 = 2 atm V 1 = 3. 0 L P 2 = 4 atm V 2 = ? 2. ) Determine which law is being used P and V = Boyle’s Law 3. ) Solve for the missing variable P 1 V 1 = P 2 V 2 (2 atm)(3. 0 L) = (4 atm)V 2 = 1. 5 L

Charles’ Law Jacques Charles discovered the relationship between temperature and volume. He measured the volume of air at different temperatures and observed a pattern which led to his mathematical law. During his experiments, pressure of the system and amount of gas were held constant. http: //en. wikipedia. org/wiki/File: Jacques_Alexandre_ C%C 3%A 9 sar_Charles. jpg

Charles’ Law
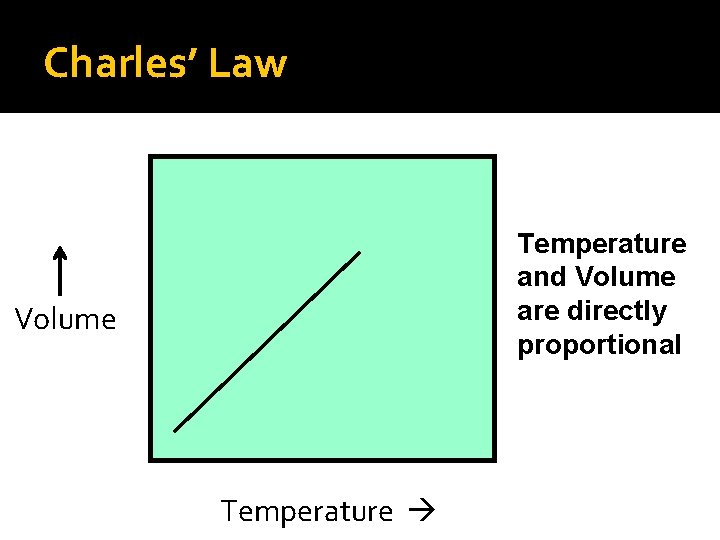
Charles’ Law Temperature and Volume are directly proportional Volume Temperature

Charles’ Law Charles’ mathematical law: V 1 = V 2 T 1 T 2

Charles’ Law: Example A gas has a volume of 3. 0 L at 127⁰C. its volume at 2727⁰C? What is

Charles’ Law: Example A gas has a volume of 3. 0 L at 127⁰C. volume at 2727⁰C? What is its 1. ) Determine which variables you have: T 1 = 127⁰C + 273 = 400 K V 1 = 3. 0 L T 2 = 227⁰C + 273 = 500 K V 2 = ? 2. ) Determine which law is being used: V 1 = V 2 T and V = Charles’ Law T 1 T 2 3. 0 L = V 2 3. ) Solve for the unknown variable 400 K 500 K V 2 = 3. 8 L

Gay-Lussac’s Law Gay Lussac discovered the relationship between temperature and pressure of a gas. He measured the temperature of air at varying pressures and observed a pattern which led to his mathematical law. During his experiments, volume of the system and amount of gas were held constant. http: //en. wikipedia. org/wiki/File: Gaylussac. jpg
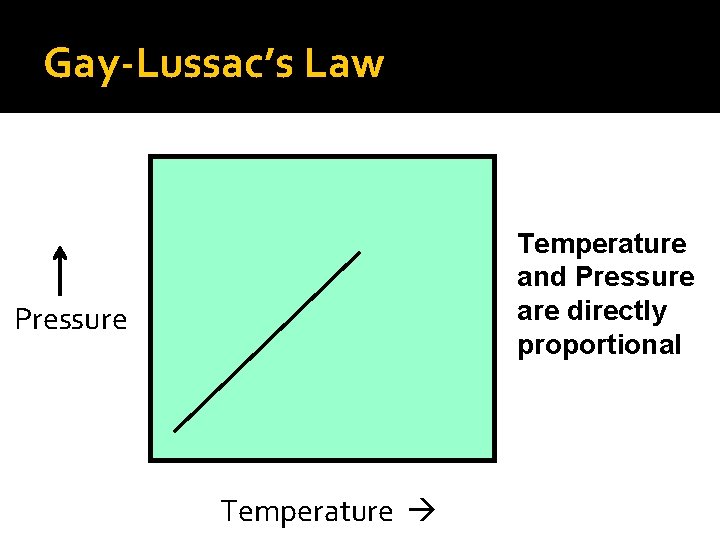
Gay-Lussac’s Law Temperature and Pressure are directly proportional Pressure Temperature

Gay-Lussac’s Law Gay-Lussac’s mathematical law: P 1 = P 2 T 1 T 2

Gay-Lussac’s Law: Example A gas has a pressure of 3. 0 atm at 127⁰C. What is its pressure at 227⁰C?

Gay-Lussac’s Law: Example A gas has a pressure of 3. 0 atm at 127⁰C. its pressure at 227⁰C? What is 1. ) Determine which variables you have: T 1 = 127⁰C + 273 = 400 K P 3. 0 atm T 2 = 227⁰C + 273 = 500 K P 2 = ? 2. ) Determine which law is being used: P 1 = P 2 T and P = Gay-Lussac’s Law T 1 T 2 3. 0 atm = P 2 3. ) Solve for the unknown variable 400 K 500 K P 2 = 3. 8 L

Combined Gas Law mathematical equation: P 1 V 1 = P 2 V 2 T 1 T 2

Combined Gas Law: Example #1 A sample of helium gas has a volume of 0. 180 L, a pressure of 0. 800 atm, and a temperature of 302 K. What is the new temperature (K) of the gas at a volume of 90. 0 m. L and a pressure of 3. 20 atm?

Combined Gas Law: Example #1 A sample of helium gas has a volume of 0. 180 L, a pressure of 0. 800 atm, and a temperature of 302 K. What is the new temperature (K) of the gas at a volume of 900 m. L and a pressure of 3. 20 atm? 1. ) Determine which variables you have: P 1 = 0. 800 atm P 2 = 3. 20 atm V 1 = 0. 180 L V 2 = 90. 0 m. L = 0. 090 L T 1 = 302 K T 2 = ? 2. ) Determine which law is being used: P 1 V 1 = P 2 V 2 P, V, and T = Combined Gas Law T 1 T 2 (0. 800 atm)(0. 180 L) = (3. 20 atm)(0. 090 L) (302 K) T 2 3. ) Solve for the unknown variable T 2 = 604 K

Combined Gas Law: Example #2 A gas has a volume of 0. 675 L at 308 K and 0. 850 atm pressure. What is the temperature in K when the gas has a volume of 0. 315 L and a pressure of 1. 055 atm?

Combined Gas Law: Example #2 A gas has a volume of 0. 675 L at 308 K and 0. 850 atm pressure. What is the temperature in K when the gas has a volume of 0. 315 L and a pressure of 1. 055 atm? 1. ) Determine which variables you have: P 1 = 0. 850 atm P 2 = 1. 055 atm V 1 = 0. 675 L V 2 = 0. 315 L T 1 = 308 K T 2 = ? 2. ) Determine which law is being used: P 1 V 1 = P 2 V 2 P, V, and T = Combined Gas Law T 1 T 2 (0. 850 atm)(0. 675 L) = (1. 055 m)(0. 315 L) (308 K) T 2 3. ) Solve for the unknown variable T 2 = 178 K

Avogadro’s Law Avogadro described the relationship between the volume and amount of a gas. http: //www. google. com/imgres

Avogadro’s Law How does adding more molecules of gas change the volume of the air in a tire? If a tire has a leak, how does the loss of air (gas) molecules change the volume?

Avogadro’s Law How does adding more molecules of gas change the volume of the air in a tire? The volume will increase. If a tire has a leak, how does the loss of air (gas) molecules change the volume? The volume will decrease.

Avogadro’s Law As the amount of gas increases, the volume increases.
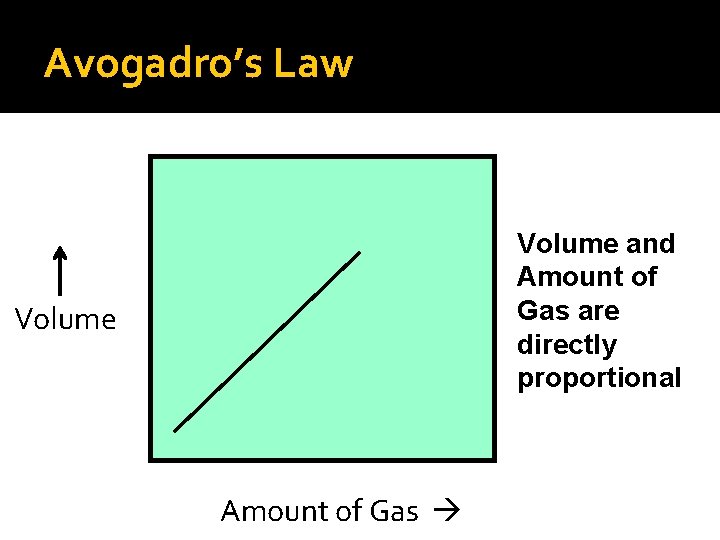
Avogadro’s Law Volume and Amount of Gas are directly proportional Volume Amount of Gas

Avogadro’s Law Avogadro’s mathematical law: V 1 = V 2 n 1 n 2

Avogadro’s Law: Example A 0. 15 mole sample of neon gas has a volume of 3. 0 L. What happens to the volume if the amount of neon gas is increased to 3. 0 moles?

Avogadro’s Law: Example A 0. 15 mole sample of neon gas has a volume of 3. 0 L. What happens to the volume if the amount of neon gas is increased to 3. 0 moles? 1. ) Determine which variables you have: V 1 = 3. 0 L V 2 = ? n 1 = 0. 15 mol n 2 = 3. 0 mol 2. ) Determine which law is being used: V and n = Avogadro’s Law 3. ) Solve for the unknown variable V 1 = V 2 n 1 n 2 (3. 0 L) = V 2 0. 15 mol 3. 0 mol V 2 = 60. 0 L

Ideal Gas Law Benoît Paul Émile Clapeyron first defined the Ideal Gas Law. http: //en. wikipedia. org/wiki/Beno %C 3%AEt_Paul_%C 3%89 mile_ Clapeyron

Ideal Gases No real gas is actually ideal. The ideal gas equation describes most real gases at room temperature and pressure of 1 atm or less. In real gases, particles attract each other reducing the pressure. Real gases behave more like ideal gases as pressure approaches zero.

Ideal Gas Law mathematical equation: PV = n. RT R = ideal gas constant = 0. 0821 L-atm mol-K

Ideal Gas Law: Example #1 N 2 O, laughing gas, is used by dentists as an anesthetic. If 2. 86 mol of gas occupies a 20. 0 L tank at 296 K, what is the pressure (atm) in the tank in the dentist office?

Ideal Gas Law: Example #1 N 2 O, laughing gas, is used by dentists as an anesthetic. If 2. 86 mol of gas occupies a 20. 0 L tank at 296 K, what is the pressure (atm) in the tank in the dentist office? 1. ) Determine which variables you have: P=? V = 20. 0 L n = 2. 86 mol T = 296 K 2. ) Determine which law is being used: P, V, n, and T = Ideal Gas Law R = 0. 0821 L-atm mol-K PV = n. RT 3. ) Solve for the unknown variable L-atm P(20. 0 L) = (2. 86 mol)(0. 0821 mol-K)(296 K) P = 3. 48 atm

Ideal Gas Law: Example #2 A 5. 0 L cylinder contains oxygen gas at 293 K and 0. 967 atm. How many moles of oxygen are in the cylinder?

Ideal Gas Law: Example #2 A 5. 0 L cylinder contains oxygen gas at 293 K and 0. 967 atm. How many moles of oxygen are in the cylinder? 1. ) Determine which variables you have: P = 0. 967 atm V = 5. 0 L n=? T = 293 K 2. ) Determine which law is being used: P, V, n, and T = Ideal Gas Law R = 0. 0821 L-atm mol-K PV = n. RT 3. ) Solve for the unknown variable L-atm (0. 967 atm)(5. 0 L) = n(0. 0821 mol-K)(293 K) P = 0. 20 mol
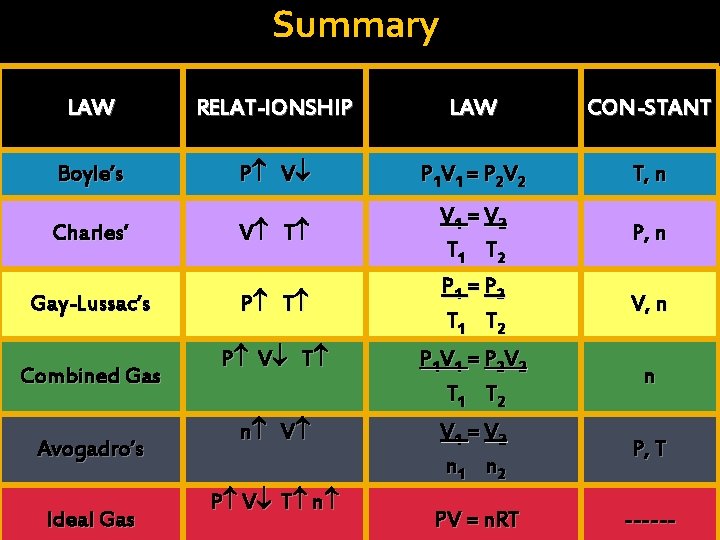
Summary LAW RELAT-IONSHIP LAW CON-STANT Boyle’s P V P 1 V 1 = P 2 V 2 T, n Charles’ V T Gay-Lussac’s P T Combined Gas Avogadro’s Ideal Gas V 1 = V 2 T 1 T 2 P 1 = P 2 T 1 T 2 P, n V, n P V T P 1 V 1 = P 2 V 2 T 1 T 2 n n V V 1 = V 2 n 1 n 2 P, T PV = n. RT ------ P V T n
- Slides: 59