State the Following Laws Boyles Law Charles Law

State the Following Laws • Boyle’s Law • Charles’ Law • Gay Lussac’s Law • Dalton’s Law • Graham’s Law

Drill: • Calculate the volume at STP of CO 2 released when 23. 4 g of aluminum carbonate is decomposed 75 % through heat

AP Test Changes • No Choices in Problems • Only 3 reactions with predicting products – Balance rxns – No choices – Explain rxns

Kinetic Theory

• All matter is made up of tiny particles • The particles are in constant motion • All collisions are elastic

Pressure

• Force per unit area • Caused by collisions against a surface • Gas measured in pressure

Units of Pressure • k. Pa: kilopascal (Std Unit) • Pascal: newton/sq. meter • Atmosphere (Atm): • mm Hg:

Standard Pressure • 101. 3 k. Pa (to be changed) • 1. 00 Atm • 760 mm Hg or Torrs • 30. 0 inches Hg • 1013 millibars

Gas Laws

Boyle’s Law

• The pressure & volume of a gas at constant temperature are inversely proportioned P 1 V 1 = P 2 V 2 = K

Charles’ Law

• The volume and temperature of a gas at constant pressure are directly proportioned V 1/T 1 = V 2/T 2 = K

Guy Lussac’s Law

• The Pressure and temperature of a gas at constant volume are directly proportioned P 1/T 1 = P 2/T 2 = K

Combined Gas Law

• Combination of the three formulas P 1 V 1 /T 1 = P 2 V 2/T 2

Calculate the new volume of 5. 0 L of gas when its pressure is doubled and its temperature is tripled:

Common Sense • The volume of a gas is directly proportioned to the number of moles of gas V 1/n 1 = V 2/n 2 = K
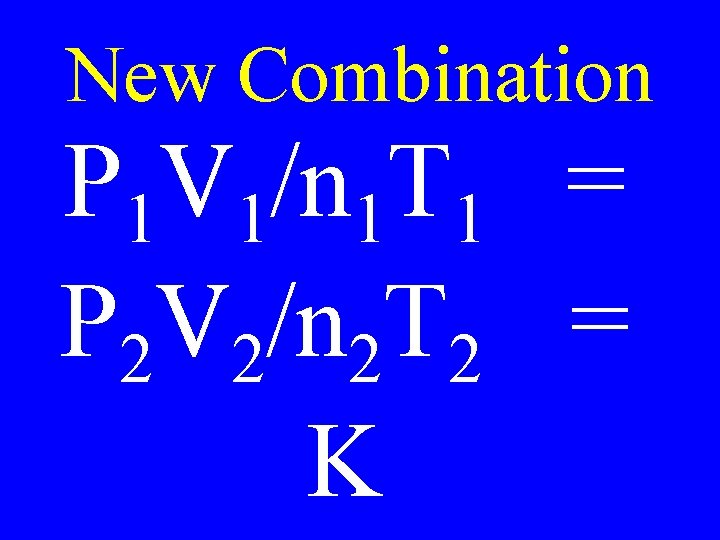
New Combination P 1 V 1/n 1 T 1 = P 2 V 2/n 2 T 2 = K

Ideal Gas Law PV = n. RT

Calculate the volume of 2. 0 moles of gas at o 77 C under 83. 1 k. Pa pressure.

Calculate the number of moles of gas occupying 831 m. L under 250 kpa at o 227 C

Calculate the number of moles of gas occupying 831 m. L under 80. 0 k. Pa at o 127 C

Calculate the mass of CO 2 occupying 83. 1 m. L under 25 GPa at o 227 C

Combined Gas Law PV = n. RT

Related Formulas m D or r = V m/n MW =

Calculate the molecular mass of 5. 0 g of gas occupying 831 m. L under 250 Mpa o at 227 C

Calculate the density of carbon dioxide at o 27 C under 83. 1 k. Pa pressure

Dalton’s Law

• The total pressure = the sum of the partial pressures PT = P 1 + P 2 + etc

The total pressure of a system is 120. 0 k. Pa. The partial pressure of gas A is 112. 0 k. Pa. Determine the pressure of gas B

Graham’s Law
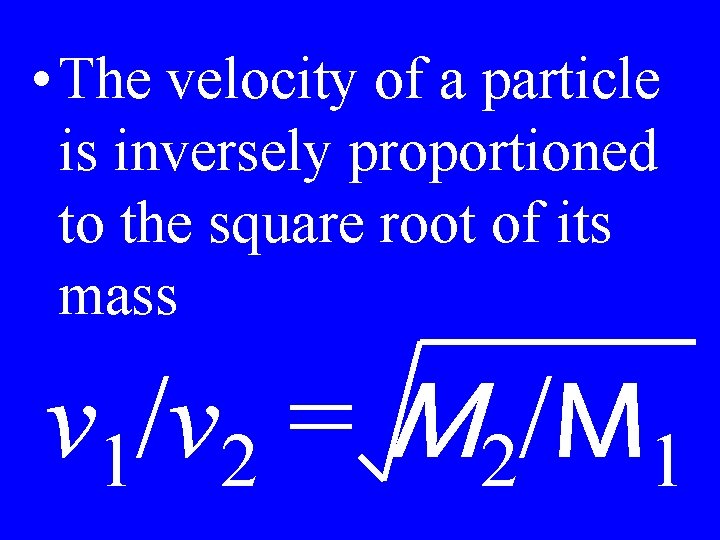
• The velocity of a particle is inversely proportioned to the square root of its mass v 1/v 2 = M 2/M 1

Drill: Calculate the ratio of the velocities of He gas to HCl gas:

Calculate the volume of a gas at STP when it occupies 80. 0 m. L at o 127 C under 303. 9 k. Pa pressure:

Drill: Calculate the volume of 4. 0 moles of gas under 83. 1 k. Pa o pressure at 127 C:

Calculate the mass of 831 m. L of CO 2 o at 27 C under 150 k. Pa pressure:

Calculate the volume of a gas at STP when it occupies 80. 0 m. L at o 127 C under 303. 9 k. Pa pressure:

Calculate the volume of 4. 0 moles of gas under 83. 1 k. Pa o pressure at 127 C:

Calculate the molecular mass of 50 g of gas occupying 831 m. L under 250 o MPa at 227 C

Calculate the mass of 831 m. L of CO 2 o at 167 C under 150 k. Pa pressure:

The total pressure of a system is 120. 0 k. Pa. The partial pressure of gas A is 112. 0 k. Pa. Determine the pressure of gas B

The total pressure of a system is 150. 0 k. Pa. The system contains 50 % A, 30 % B, & 20 % C. Determine the pressure of each gas.

Drill: Calculate the mass of CO 2 occupying 83. 1 m. L under 25 MPa at o 477 C

Calculate the density of carbon o dioxide at 27 C under 83. 1 k. Pa pressure

Calculate the ratio of the velocities of He gas to HCl gas:

Calculate the velocity HBr when the velocity Be is 270 m/s:

Calculate the final volume that 3. 0 L of gas will obtain when the absolute temperature is tripled & the pressure is halved.

Calculate the mass of CO occupying o 831 k. L at 227 C under 2. 50 Mpa pressure.

Calculate the volume of o H 2 formed at 27 C under 150 k. Pa when 6. 8 mg NH 3 decomposes making N 2 & H 2.
- Slides: 52