Chapter 16 Section 3 GAS BEHAVIOR Pressure Definition

Chapter 16 Section 3 GAS BEHAVIOR

Pressure! � Definition of pressure is: �Amount of force exerted on an area �P = F/A �Unit in SI: Pascal (Pa) �One Pascal is one Newton force on an area 1 square meter � Gases exert pressure due to KE of their particles
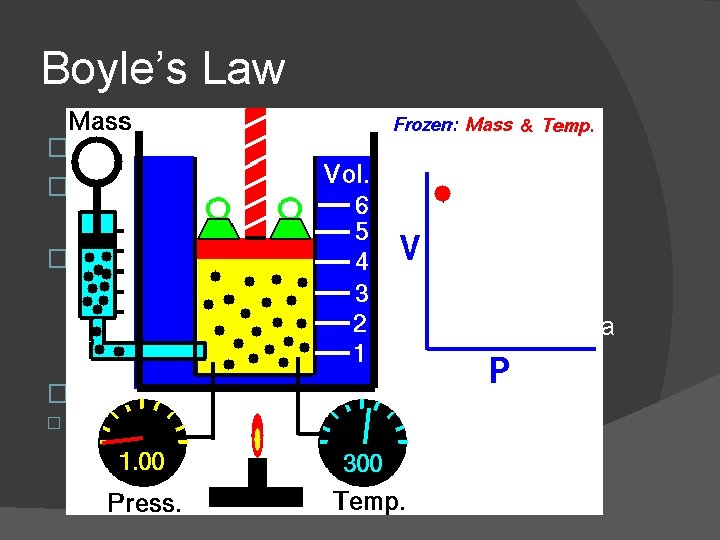
Boyle’s Law � Robert Boyle (1627 -1691) � Studied relationship between volume and pressure of gases � Found: as volume goes up, pressure goes down �Product of pressure and volume of a gas is a constant if temperature does not change � As � an equation: PV=constant http: //www. grc. nasa. gov/WWW/K 12/airplane/Animation/frglab. html

Boyle’s Law � More commonly, we compare pressure and volume of the same gas � Since PV is constant, we can say: �P 1 V 1 = P 2 V 2 for any gas � To see this in action, let’s do Math Skills Activity p 505!

Math Skills Activity! �A balloon has a volume of 10. 0 L at a pressure of 101 k. Pa. What will the balloon’s new volume be if the pressure changes to 43 k. Pa? � What we know: �P 1 = 101 k. Pa �V 1 = 10. 0 L �P 2 = 43 k. Pa �V 2 = ?

� What equation? �P 1 V 1 = P 2 V 2 �So V 2 = P 1 V 1 P 2 � Put in the values! �V 2 = (101 k. Pa)(10. 0 L) (43 k. Pa) � Do the math! �V 2 = 23. 488 L � Check units and math!

Another Problem: �A volume of helium occupies 11. 0 L at 98. 0 k. Pa. What is the new volume if the pressure increases to 101. 0 k. Pa? � What do we know? �P 1 = 98. 0 k. Pa �V 1 = 11. 0 L �P 2 = 101. 0 k. Pa �V 2 = ?

� What equation? �P 1 V 1 = P 2 V 2 �So V 2 = P 1 V 1 P 2 � Put in the numbers! �V 2 = (98. 0 k. Pa)(11. 0 L) (101. 0 k. Pa) � Do the math! �V 2 = 10. 67 L � Check your math and units.
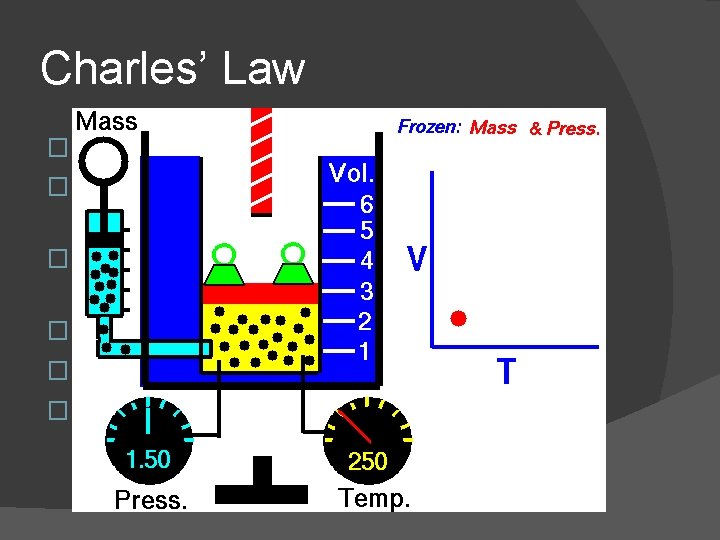
Charles’ Law � Jaques Charles (1746 -1823) � Studied relationship of temperature and volume with constant pressure � As temperature goes up, volume increases � Volume/Temperature is a constant � As an equation: K = V/T � This relationship is the basis for the Kelvin temperature scale

Charles’ Law � More commonly, we compare one situation to another for a gas � V 1 = V 2 With constant pressure T 1 T 2 Temperatures must be used in Kelvins!

Charles’ Law Problem � If a balloon has a volume of 2. 0 L at 25 o. C, what is the volume of the balloon at 3 o. C? � What do we know? �T 1 = 25 o. C = 298 K �V 1 = 2. 0 L �T 2 = 3 o. C = 276 K �V 2 = ?

Charles’ Law Problem � What equation? � V 1 = V 2 T 1 T 2 So V 2 = V 1 T 2 T 1 � Put in the numbers! Remember, temperature must be in Kelvins! � V 2 = (2. 0 L)(276 K) (298 K) � Solve! � V 2 = 1. 85 L � Check for math and units.
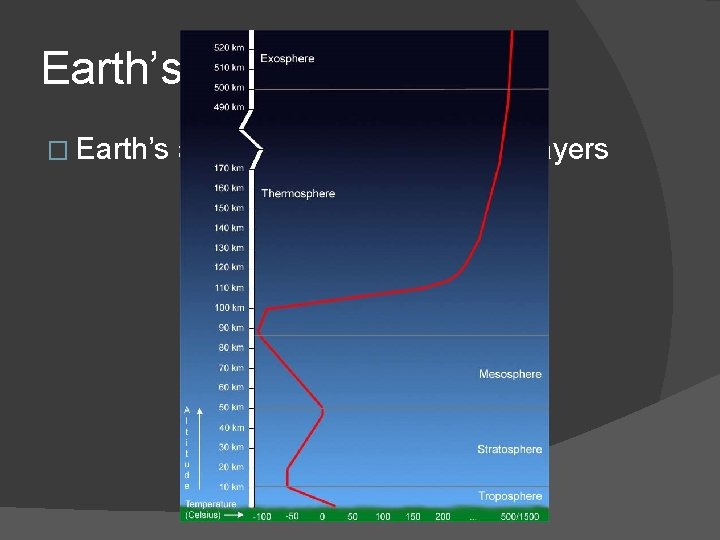
Earth’s Atmosphere � Earth’s atmosphere divided into 5 layers

Earth’s Atmosphere � Exosphere: outer layer � Thermosphere � Mesosphere � Stratosphere � Troposphere: layer we live in!

Exosphere � Very thin outer portion of atmosphere � Negligible pressure
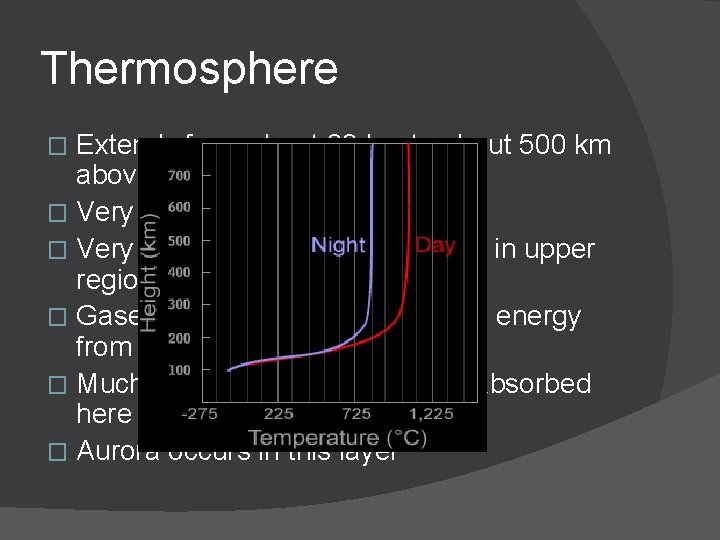
Thermosphere Extends from about 90 km to about 500 km above Earth � Very low pressure � Very high temperatures (1000 o. C) in upper regions � Gases here broken into atoms by energy from sun � Much of X-ray and UV radiation absorbed here � Aurora occurs in this layer �
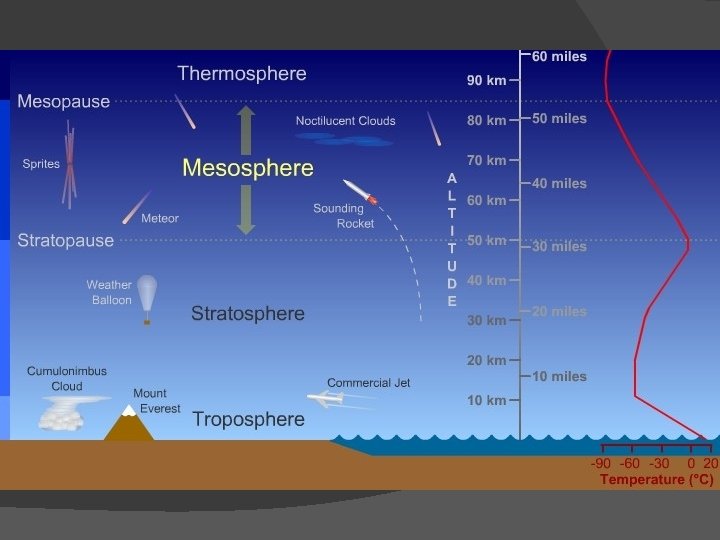
Mesosphere � About 50 to 85 km above Earth’s surface � Temperatures get very cold (-25 o. C to -90 o. C) � Air is mixed by currents � Most meteors burn up in this layer
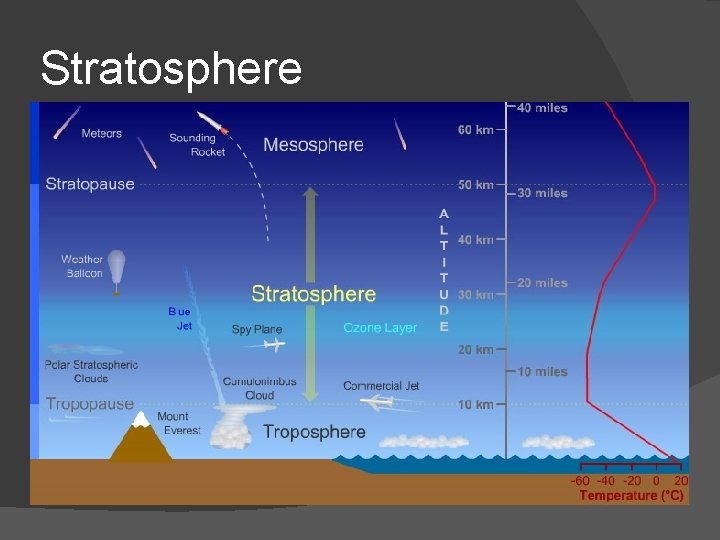
Stratosphere � Altitude of the bottom of this layer varies with latitude and season � 16 km at equator � 8 km at poles � Ozone heats this layer as it absorbs UV from sun � Very few clouds, very dry air
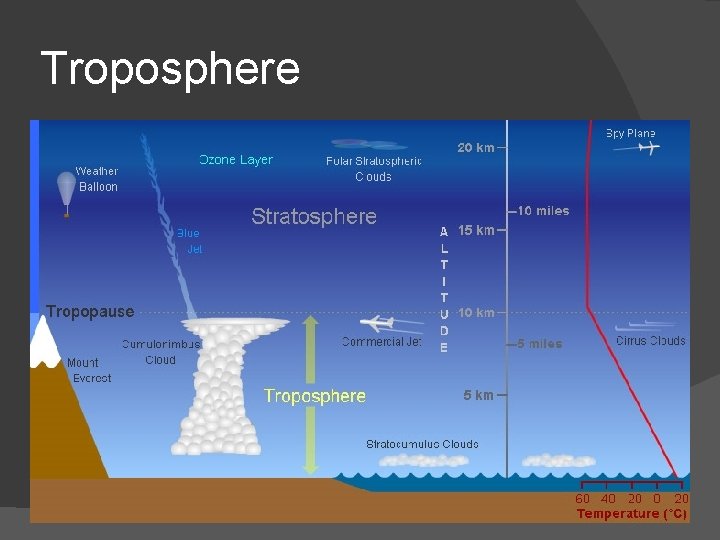
Troposphere � Layer we live in! � From surface up to 7 to 20 km altitude � Almost all weather occurs in this layer � Almost all dust particles and water vapor in this layer � This layer heated from below, so warmer at lower altitudes than higher

Gases are fun!
- Slides: 20