Gas Law Kinetic Theory Summary Objectives 1 State

Gas Law & Kinetic Theory Summary

Objectives 1. State the P-V relation, the p-T relation, V-T relation and the general gas law for a fixed mass of gas 2. Describe an experiment to verify each gas law (or relations) 3. Apply gas laws to solve problems 4. Express temperatures in the Kelvin scale 5. Describe the physical significance of the absolute zero of temperature 6. Explain gas pressure in terms of the kinetic theory
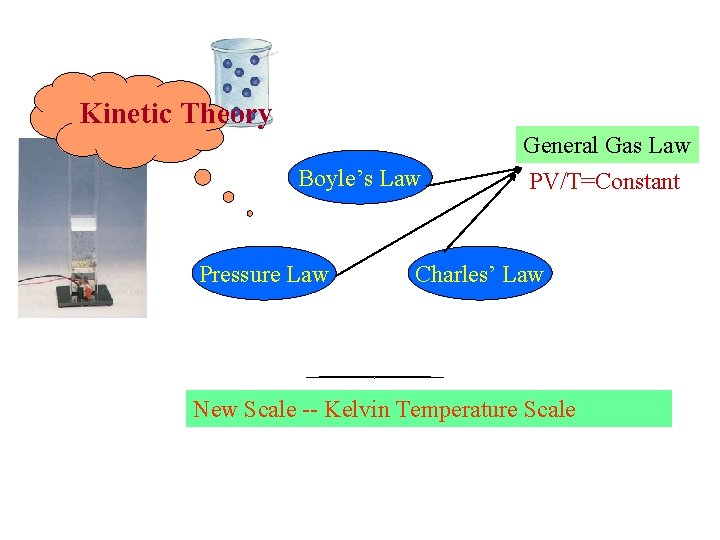
Kinetic Theory Boyle’s Law Pressure Law General Gas Law PV/T=Constant Charles’ Law New Scale -- Kelvin Temperature Scale

Describe an experiment to verify each gas law (or relations) Charles’ Law V/T=Constant Pressure Law Boyle’s Law P/T=Constant PV=Constant

General Gas Equation Boyle’s Law: PV = constant Charles’ Law: V/T = constant Pressure Law: P/T = constant PV/T = constant P 1 V 1/ T 1 = P 2 V 2/T 2

• Explain, in terms of the kinetic theory i. why the pressure of a gas increases when it is compressed at constant temperature? ii. why the pressure of a fixed volume of gas increases with temperature? iii. why the volume of a gas increases when heated at constant pressure?

i. why the pressure of a gas increases when it is compressed at constant temperature? Kinetic theory and Boyle’s Law • When a gas is compressed at constant temperature, • the molecules have less volume to move and hit the wall of the container more frequently. • As a result, pressure will increases.
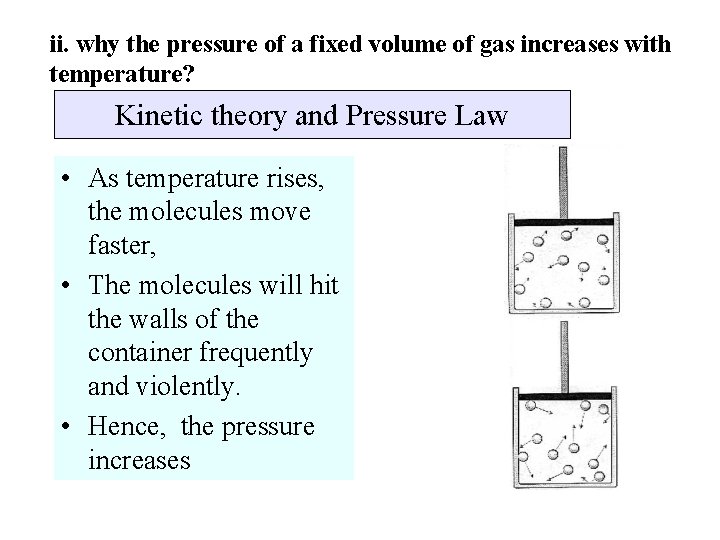
ii. why the pressure of a fixed volume of gas increases with temperature? Kinetic theory and Pressure Law • As temperature rises, the molecules move faster, • The molecules will hit the walls of the container frequently and violently. • Hence, the pressure increases
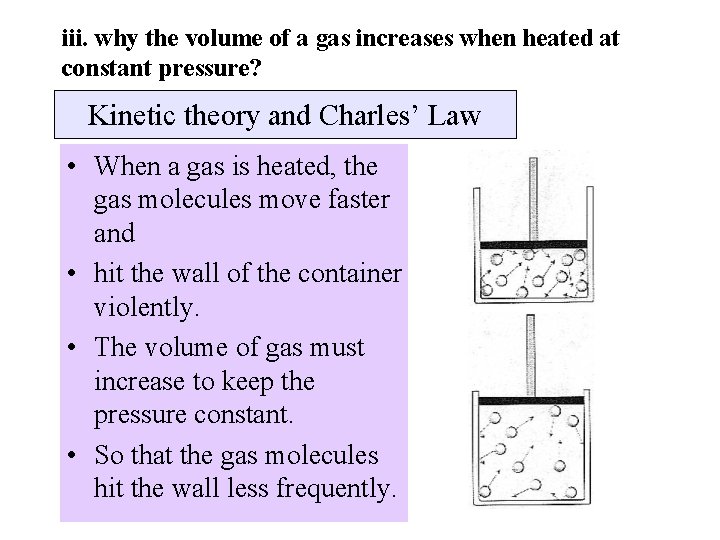
iii. why the volume of a gas increases when heated at constant pressure? Kinetic theory and Charles’ Law • When a gas is heated, the gas molecules move faster and • hit the wall of the container violently. • The volume of gas must increase to keep the pressure constant. • So that the gas molecules hit the wall less frequently.
- Slides: 9