CHEMISTRY Matter and Change Chapter 23 The Chemistry







































































- Slides: 85

CHEMISTRY Matter and Change Chapter 23: The Chemistry of Life

CHAPTER 23 Table Of Contents Section 23. 1 Proteins Section 23. 2 Carbohydrates Section 23. 3 Lipids Section 23. 4 Nucleic Acids Section 23. 5 Metabolism Click a hyperlink to view the corresponding slides. Exit

2 3. 1 SECTION • Describe the structures of amino acids and proteins. • Explain the roles of proteins in cells. Proteins polymer: large molecules composed of many repeating units called monomers

Proteins 2 3. 1 SECTION protein denaturation amino acid enzyme peptide bond substrate peptide active site Proteins perform essential functions, including regulation of chemical reactions, structural support, transport of materials, and muscle contractions.

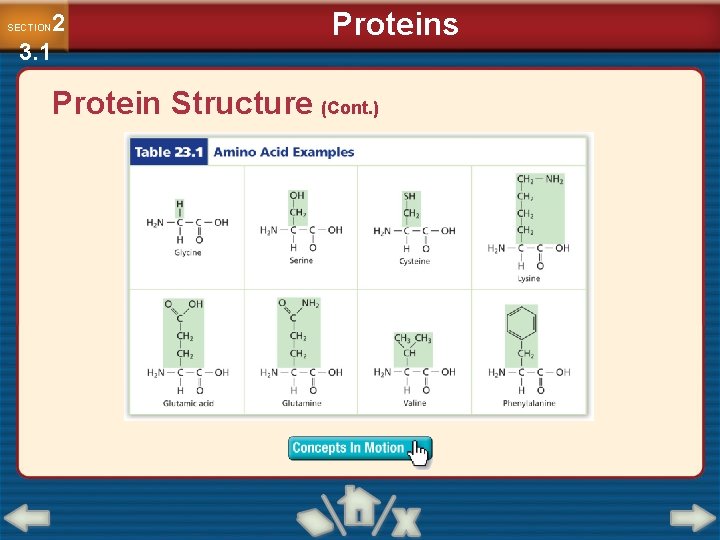
2 3. 1 SECTION Proteins Protein Structure • Proteins are organic polymers made of amino acids linked together in a specific order, not just random chains of amino acids. • Amino acids are organic molecules that have both an amino group and an acidic carboxyl group.

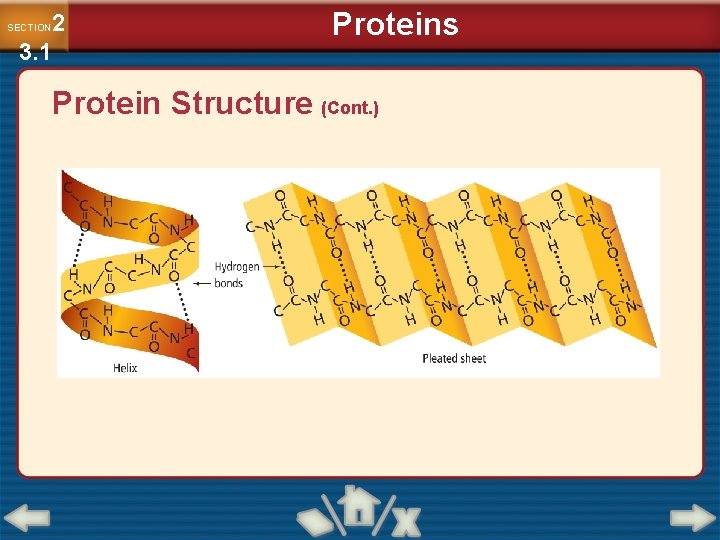
2 3. 1 SECTION Proteins Protein Structure (Cont. ) • Each amino acid has a central carbon atom, with four groups arranged around it: an amino group, carboxyl group, hydrogen atom, and variable side chain.

2 3. 1 SECTION Proteins Protein Structure (Cont. )

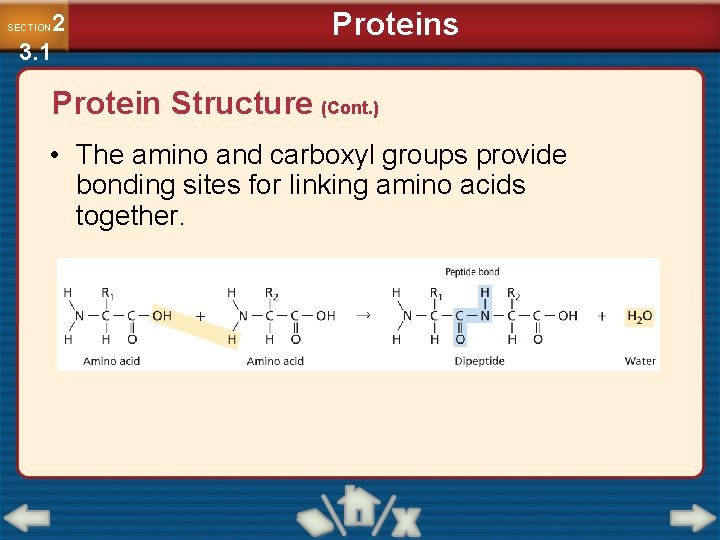
2 3. 1 SECTION Proteins Protein Structure (Cont. ) • The amino and carboxyl groups provide bonding sites for linking amino acids together.

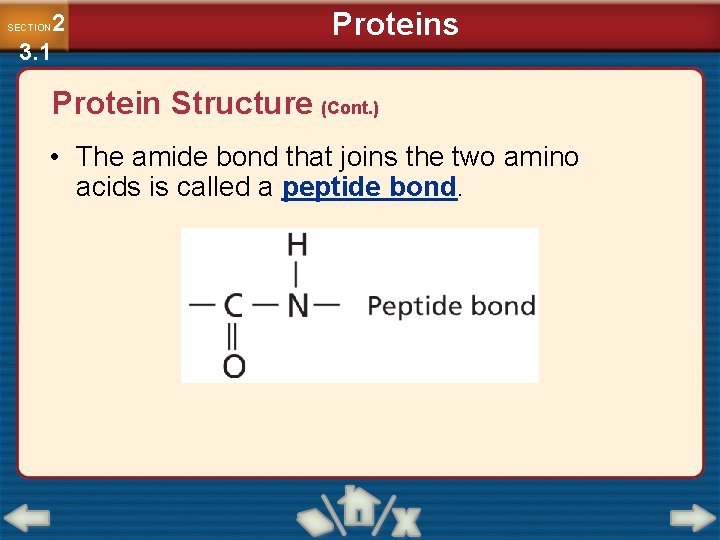
2 3. 1 SECTION Proteins Protein Structure (Cont. ) • The amide bond that joins the two amino acids is called a peptide bond.

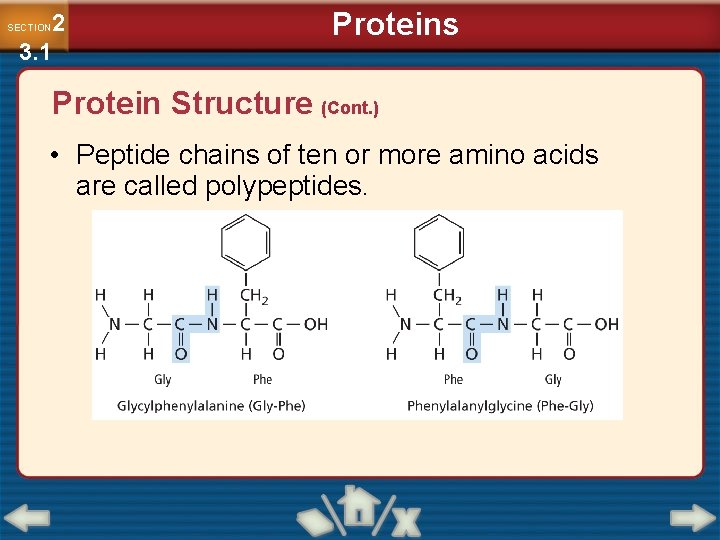
2 3. 1 SECTION Proteins Protein Structure (Cont. ) • Peptide chains of ten or more amino acids are called polypeptides.

2 3. 1 SECTION Proteins Protein Structure (Cont. ) • When a chain reaches 50 or more amino acids, it is called a protein. • Only 20 different amino acids exist.

2 3. 1 SECTION Proteins Protein Structure (Cont. ) • Long chains of amino acids fold in unique shapes determined by the interaction among amino acids. • Denaturation is the process in which a protein’s natural three-dimensional structure is disrupted. –Changes in temperature, ionic strength, p. H, and other factors result in the unfolding and uncoiling of a protein.

2 3. 1 SECTION Proteins Protein Structure (Cont. )

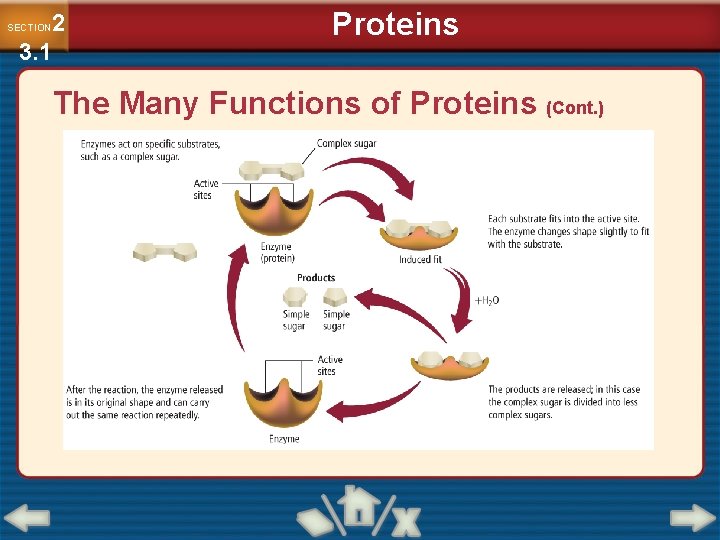
2 3. 1 SECTION Proteins The Many Functions of Proteins • An enzyme is a biological catalyst. • A catalyst lowers the activation energy of a reaction by stabilizing the transition states. • A substrate refers to the reactant in an enzyme-catalyzed reaction. • The spot to which the substrates bind is called the active site. • Substrates must fit the active site in the same way puzzle pieces fit together.

2 3. 1 SECTION Proteins The Many Functions of Proteins (Cont. )

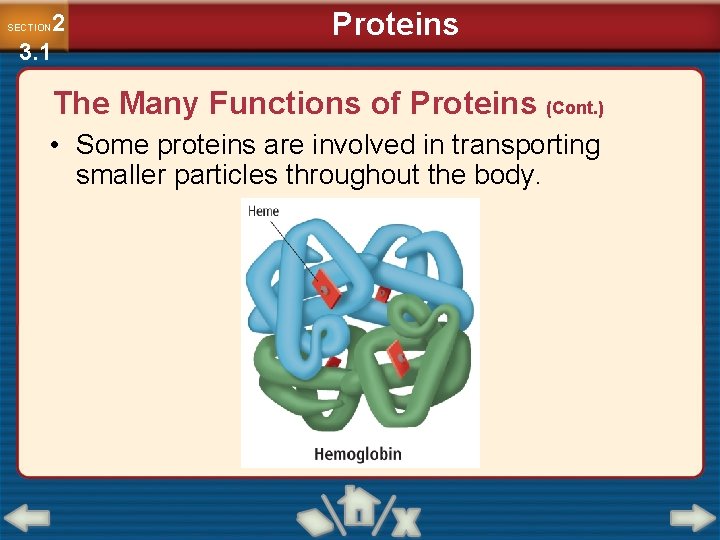
2 3. 1 SECTION Proteins The Many Functions of Proteins (Cont. ) • Some proteins are involved in transporting smaller particles throughout the body.

2 3. 1 SECTION Proteins The Many Functions of Proteins (Cont. ) • Certain proteins’ sole function is to form structures vital to organisms. They are known as structural proteins. – The most abundant structural protein in most animals is collagen, which makes up skin, ligaments, tendons, and bones. – Other structural proteins make up feathers, fur, wool, hooves, fingernails, cocoons and hair.

2 3. 1 SECTION Proteins The Many Functions of Proteins (Cont. ) • Some hormones are proteins and are used as chemical messenger molecules that carry signals from one part of the body to another. –Ex. Insulin, made by pancreas cells, when released into the bloodstream signals the body the blood sugar is abundant and should be stored.

2 3. 1 SECTION Section Check Which best describes a protein? A. a building block B. a biological polymer C. an enzyme D. a catalyst

2 3. 1 SECTION Section Check Amino acids in a protein are linked together by ____. A. ionic bonds B. hydrogen bonds C. peptide bonds D. temporary dipole


2 3. 2 SECTION Carbohydrates • Describe the structures of monosaccharides, disaccharides, and polysaccharides. • Explain the functions of carbohydrates in living things. stereoisomers: a class of isomers whose atoms are bonded in the same order but are arranged differently in space

Carbohydrates 2 3. 2 SECTION carbohydrate monosaccharide disaccharide polysaccharide Carbohydrates provide energy and structural material for living things.

2 3. 2 SECTION Carbohydrates Kinds of Carbohydrates • Carbohydrates are compounds that contain multiple hydroxyl groups as well as carbonyl groups. • Monosaccharides are the simple sugars, composed of five or six carbon atoms.

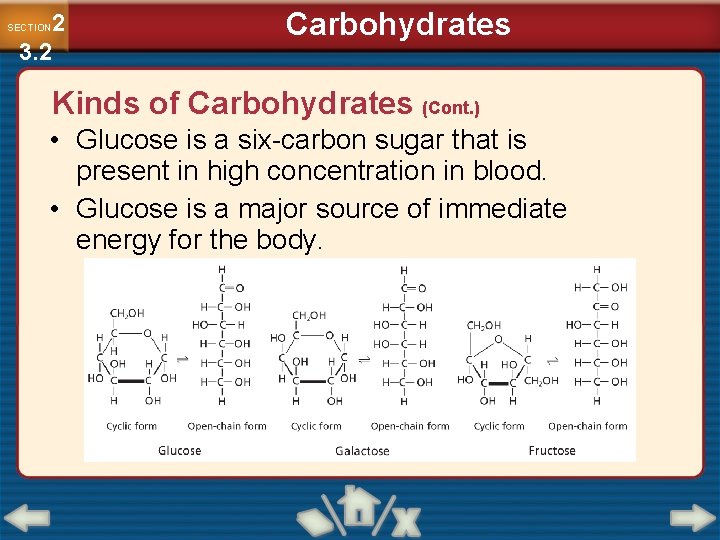
2 3. 2 SECTION Carbohydrates Kinds of Carbohydrates (Cont. ) • Glucose is a six-carbon sugar that is present in high concentration in blood. • Glucose is a major source of immediate energy for the body.

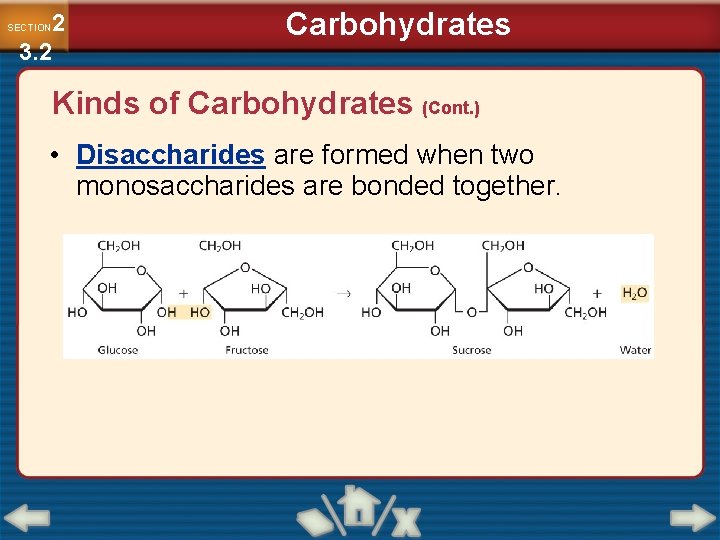
2 3. 2 SECTION Carbohydrates Kinds of Carbohydrates (Cont. ) • Disaccharides are formed when two monosaccharides are bonded together.

2 3. 2 SECTION Carbohydrates Kinds of Carbohydrates (Cont. ) • Polysaccharides are polymers of simple sugars made up of 12 or more monomers. • Glycogen is an important polysaccharide found in animals that is used to store energy. • Starch and cellulose are also important polysaccharides found in starch.

2 3. 2 SECTION Section Check Carbohydrates contain which two functional groups? A. amine and hydroxyl B. carbonyl and amide C. carbonyl and aldehyde D. hydroxyl and carbonyl

2 3. 2 SECTION Section Check Which of the following is considered a monosaccharide? A. glucose B. glycogen C. starch D. cellulose


2 3. 3 SECTION Lipids • Describe the structures of fatty acids, triglycerides, phospholipids, and steroids. • Explain the functions of lipids in living organisms. • Identify some reactions that fatty acids undergo. • Relate the structure and function of cell membranes. nonpolar: without separate positive and negative areas or dipoles

Lipids 2 3. 3 SECTION lipid phospholipid fatty acid wax triglyceride steroid saponification Lipids make cell membranes, store energy, and regulate cellular processes.

2 3. 3 SECTION Lipids What is a lipid? • A lipid is a large, nonpolar biological molecule. • Fatty acids, the building blocks of lipids, are long-chain carboxylic acids. • Fatty acids are placed in two groups: saturated and unsaturated.

2 3. 3 SECTION Lipids What is a lipid? (cont. ) • Saturated fats contain only single bonds. • Unsaturated fats contain one or more double bonds.

2 3. 3 SECTION Lipids What is a lipid? (cont. ) • Triglycerides are formed when three fatty acids are bonded to a glycerol backbone. • Triglycerides can be solids or liquids at room temperature.

2 3. 3 SECTION Lipids What is a lipid? (cont. ) • Saponification is the hydrolysis of a triglyceride using an aqueous solution of a strong base to form carboxylate salts and glycerol. • Saponification is used to make soaps.

2 3. 3 SECTION Lipids What is a lipid? (cont. ) • Phospholipids are triglycerides in which one fatty acid is replaced by a polar phosphate group. • Cell membranes are made up of a double layer of phospholipids, called a lipid bilayer.

2 3. 3 SECTION Lipids What is a lipid? (cont. ) • Waxes are lipids formed by combining a fatty acid with a long-chain alcohol. • Both plants and animals make waxes. • Steroids are lipids that have multiple cyclic rings in their structures.

2 3. 3 SECTION Section Check A triglyceride in which a fatty acid is replaced by a phosphate group is a ____. A. wax B. phospholipid C. steroid D. fatty acid

2 3. 3 SECTION Section Check Fatty acids, the building blocks of lipids, are composed of what? A. triglycerides B. steroids C. amides D. long-chain carboxylic acids


2 3. 4 SECTION Nucleic Acids • Identify the structural components of nucleic acids. • Relate the function of DNA to its structure. genetic information: an inherited sequence of RNA or DNA that causes traits or characteristics to pass from one generation to the next • Describe the structure and function of RNA. nucleic acid nucleotide Nucleic acids store and transmit genetic information.

2 3. 4 SECTION Nucleic Acids Structure of Nucleic Acids • A nucleic acid is a nitrogen-containing biological polymer that is involved in the storage and transmission of genetic information. • The monomer that makes up a nucleic acid is called a nucleotide.

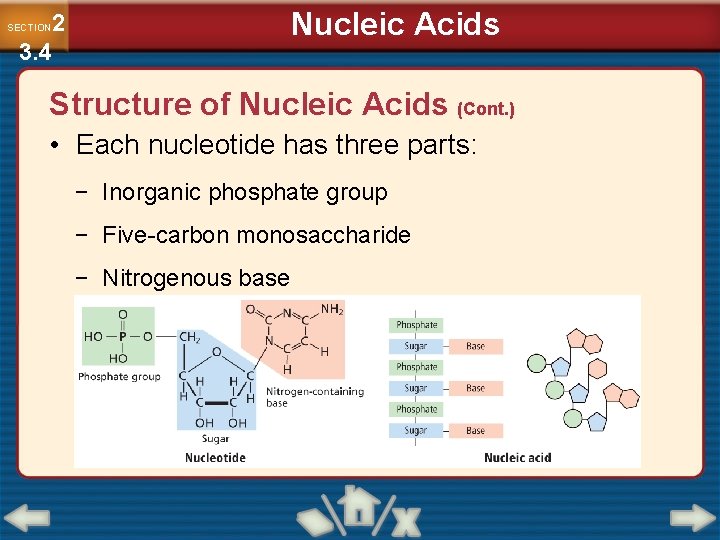
Nucleic Acids 2 3. 4 SECTION Structure of Nucleic Acids (Cont. ) • Each nucleotide has three parts: − Inorganic phosphate group − Five-carbon monosaccharide − Nitrogenous base

2 3. 4 SECTION Nucleic Acids DNA: The Double Helix • DNA consists of two long chains of nucleotides wound together to form a spiral structure. • The spiral structure is known as a double helix.

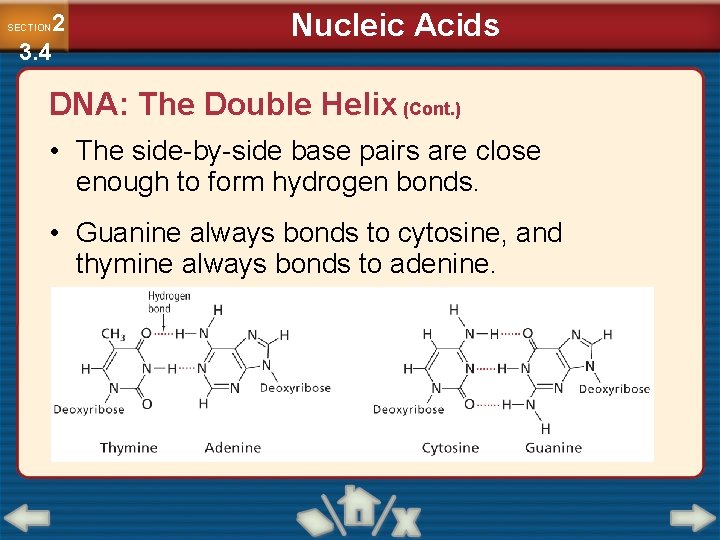
Nucleic Acids 2 3. 4 SECTION DNA: The Double Helix (Cont. ) • DNA contains four different nitrogenous bases. − Adenine − Thymine − Cytosine − Guanine

2 3. 4 SECTION Nucleic Acids DNA: The Double Helix (Cont. ) • The side-by-side base pairs are close enough to form hydrogen bonds. • Guanine always bonds to cytosine, and thymine always bonds to adenine.

2 3. 4 SECTION Nucleic Acids DNA: The Double Helix (Cont. ) • Watson and Crick used their model to predict how DNA’s chemical structure enables it to function. • DNA stores genetic information of a cell in the cell’s nucleus. • The two chains of a DNA helix are complementary. This complementary pairing provides a mechanism by which genetic material of a cell is copied.

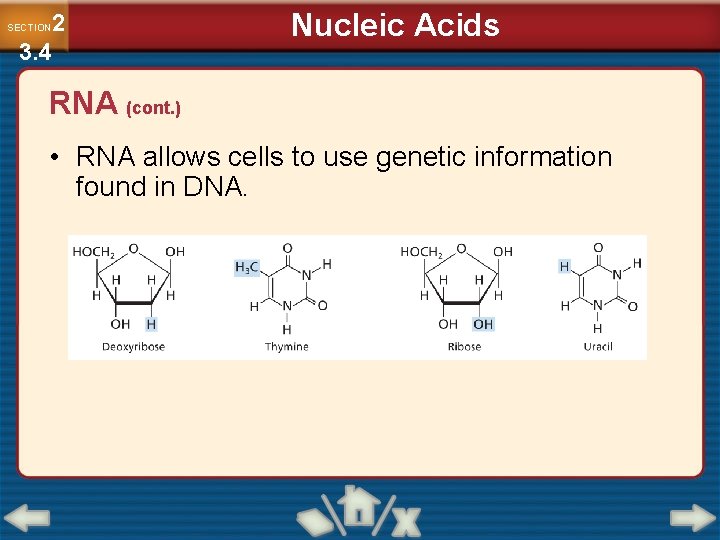
Nucleic Acids 2 3. 4 SECTION RNA • RNA is also a nucleic acid. • The structure of RNA differs from DNA in three ways. − RNA contains adenine, cytosine, guanine, and uracil (but never thymine). − RNA contains sugar ribose instead of sugar deoxyribose. − DNA is a double helix while RNA is a single strand.

2 3. 4 SECTION Nucleic Acids RNA (cont. ) • RNA allows cells to use genetic information found in DNA.

2 3. 4 SECTION Section Check Which is NOT part of a nucleotide? A. nitrogenous base B. lipid C. phosphate group D. sugar

2 3. 4 SECTION Section Check Which is NOT a difference between RNA and DNA? A. DNA is a double helix; RNA a single strand. B. DNA is a nucleic acid; RNA is not. C. DNA has thymine; RNA has uracil. D. DNA contains deoxyribose sugar; RNA contains ribose sugar.


2 3. 5 SECTION Metabolism • Distinguish between anabolism and catabolism. • Describe the role of ATP in metabolism. • Compare and contrast the processes of photosynthesis, cellular respiration, and fermentation. redox process: a chemical reaction in which electrons are transferred from one atom to another

2 3. 5 SECTION Metabolism metabolism photosynthesis catabolism cellular respiration anabolism fermentation ATP Metabolism involves many thousands of reactions in living cells.

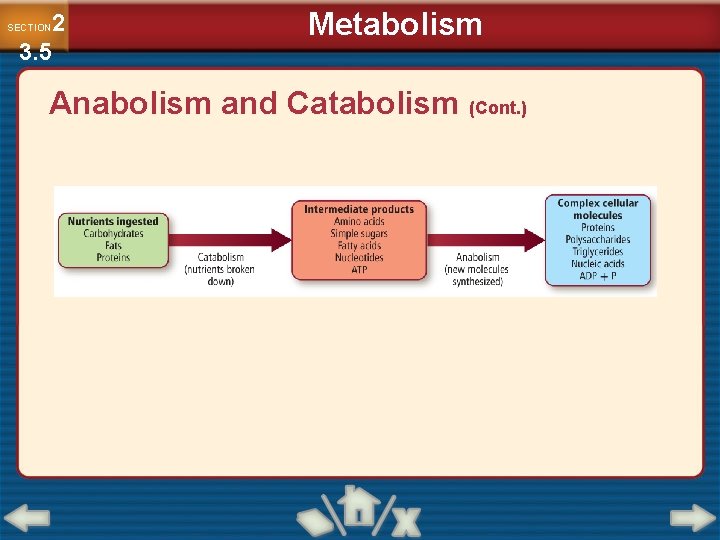
2 3. 5 SECTION Metabolism Anabolism and Catabolism • The set of chemical reactions carried out within an organism is its metabolism. • Catabolism refers to the metabolic processes that break down complex biological molecules such as proteins, polysaccharides, triglycerides, and nucleic acids for the purpose of forming smaller building blocks and extracting energy.

2 3. 5 SECTION Metabolism Anabolism and Catabolism (Cont. ) • Anabolism refers to the metabolic reactions that use energy and small building blocks to synthesize complex molecules needed by an organism. • Catabolism and anabolism are linked by common building blocks that catabolic reactions produce and anabolic reactions use.

2 3. 5 SECTION Metabolism Anabolism and Catabolism (Cont. )

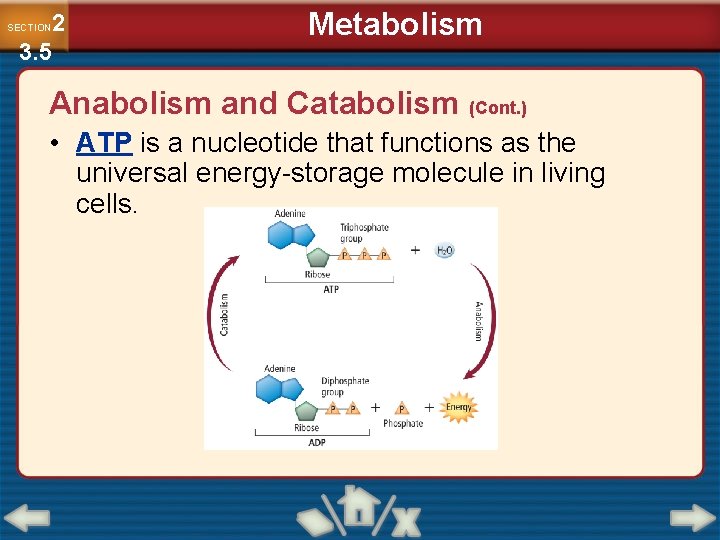
2 3. 5 SECTION Metabolism Anabolism and Catabolism (Cont. ) • ATP is a nucleotide that functions as the universal energy-storage molecule in living cells.

2 3. 5 SECTION Metabolism Photosynthesis • The process that converts energy from sunlight to chemical energy in the bonds of carbohydrates is called photosynthesis. • Photosynthesis results in the reduction of carbon atoms in carbon dioxide as glucose is formed.

2 3. 5 SECTION Metabolism Cellular Respiration • Oxygen produced during photosynthesis is used by living things during cellular respiration, the process in which glucose is broken down to form carbon dioxide, water, and large amounts of energy. • Cellular respiration is the major energyproducing process in living organisms.

2 3. 5 SECTION Metabolism Fermentation • Cells can extract energy from glucose in the absence of oxygen. • Cellular respiration produces 38 mol of ATP per 1 mol glucose. • Fermentation produces 2 mol ATP per 1 mol of glucose.

2 3. 5 SECTION Metabolism Fermentation (cont. ) • Fermentation is the process by which glucose is broken down in the absence of oxygen. • There two common kinds of fermentation: alcoholic and lactic acid fermentation.

2 3. 5 SECTION Metabolism Fermentation (cont. ) • In alcoholic fermentation, yeast and some bacteria can ferment glucose to produce ethanol. • Alcoholic fermentation is used to make bread, form tofu, and produce ethanol in alcoholic beverages.

2 3. 5 SECTION Metabolism Fermentation (cont. ) • In lactic acid fermentation, when the oxygen supply is depleted, cellular respiration stops. • Animal cells produce lactic acid and a small amount of energy from lactic acid fermentation of glucose. • Build-up of lactic acid is what results in burning pain in the muscles during strenuous exercise.

2 3. 5 SECTION Section Check Which process is the major energyproducing process in living organisms? A. photosynthesis B. cellular respiration C. alcoholic fermentation D. lactic acid fermentation

2 3. 5 SECTION Section Check What process breaks down glucose in the absence of oxygen? A. anabolism B. catabolism C. cellular respiration D. fermentation


The Chemistry of Life CHAPTER 23 Resources Chemistry Online Study Guide Chapter Assessment Standardized Test Practice

Proteins 2 3. 1 SECTION Study Guide Key Concepts • Proteins are biological polymers made of amino acids that are linked by peptide bonds. • Protein chains fold into intricate three-dimensional structures. • Proteins have many functions in the human body, including functions within cells, functions between cells, and functions of structural support.

Carbohydrates 2 3. 2 SECTION Study Guide Key Concepts • Carbohydrates are compounds that contain multiple hydroxyl groups (–OH) and a carbonyl functional group (C=O). • Carbohydrates range in size from single monomers to polymers composed of hundreds or thousands of monomers. • Monosaccharides in aqueous solution exist in both open-chain and cyclic structures.

Lipids 2 3. 3 SECTION Study Guide Key Concepts • Fatty acids are long-chain carboxylic acids that usually have between 12 and 24 carbon atoms. • Saturated fatty acids have no double bonds; unsaturated fatty acids have one or more double bonds. • Fatty acids can be linked to glycerol backbones to form triglycerides. • Steroids are lipids that have multiple-ring structures.

Nucleic Acids 2 3. 4 SECTION Study Guide Key Concepts • Nucleic acids are polymers of nucleotides, which consist of a nitrogen base, a phosphate group, and a sugar. • DNA and RNA are the information-storage molecules of a cell. • DNA is double stranded, and RNA is single stranded.

Metabolism 2 3. 5 SECTION Study Guide Key Concepts • Living organisms undergo catabolism and anabolism. • Photosynthesis directly or indirectly provides all living things with energy. • The net equation for cellular respiration is the reverse of the net equation for photosynthesis.

CHAPTER 23 The Chemistry of Life Chapter Assessment What are biological polymers made of amino acid monomers called? A. enzymes B. peptides C. nucleotides D. proteins

CHAPTER 23 The Chemistry of Life Chapter Assessment Carbonyl and hydroxyl groups are found in which group? A. lipids B. fatty acids C. carbohydrates D. proteins

CHAPTER 23 The Chemistry of Life Chapter Assessment Cell membranes are usually made of what? A. fatty acids B. cellulose C. triglycerides D. phospholipids

CHAPTER 23 The Chemistry of Life Chapter Assessment Nucleotides are the monomers of what biological polymer? A. nucleic acid B. nitrogenous bases C. polysaccharides D. polypeptides

CHAPTER 23 The Chemistry of Life Chapter Assessment Energy from sunlight is harnessed by which process? A. alcoholic fermentation B. cellular respiration C. photosynthesis D. catabolism

CHAPTER 23 The Chemistry of Life Standardized Test Practice Which of the following is NOT true about cellulose? A. It is a polysaccharide. B. It is easily digestible by humans. C. It is produced by plants. D. It is a carbohydrate.

CHAPTER 23 The Chemistry of Life Standardized Test Practice Which of the following is NOT true about DNA? A. DNA contains the nitrogenous base thymine. B. DNA is usually double stranded. C. DNA contains the nitrogenous base uracil. D. DNA contains deoxyribose sugar.

CHAPTER 23 The Chemistry of Life Standardized Test Practice What is the condensed structural formula of 1 pentanol? A. CH 3(CH 2)4 OH B. CH 3(CH 2)3 COOH C. CH 3(CH 2)3 CH=O D. CH 2 -O-(CH 2)3 CH 3

CHAPTER 23 The Chemistry of Life Standardized Test Practice What is the oxidation number of Zn in Zn(NO 3)2? A. +2 B. +3 C. +5 D. +6

CHAPTER 23 The Chemistry of Life Standardized Test Practice Glucose is considered a ____. A. simple sugar B. polysaccharide C. fatty acid D. nucleotide

End of Custom Shows This slide is intentionally blank.