CHEMISTRY PROPERTIES OF MATTER CLASSIFYING MATTER v Matter

































- Slides: 37

CHEMISTRY PROPERTIES OF MATTER

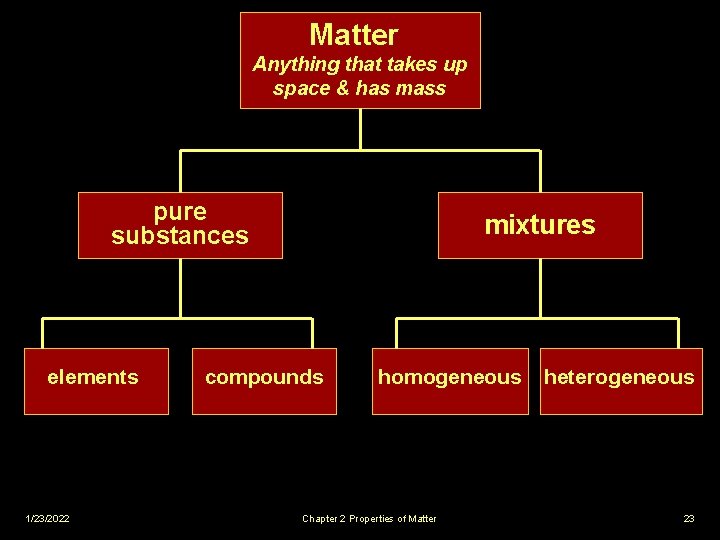
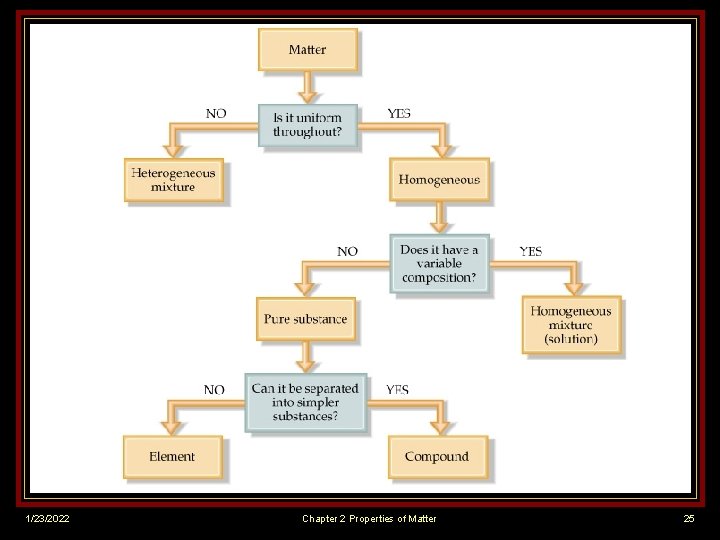
CLASSIFYING MATTER v Matter - Anything that takes up space and has mass v Based on their compositions, materials can be divided into pure substances and mixtures. 1/23/2022 Chapter 2 Properties of Matter 2

PURE SUBSTANCES (BOX 1) Pure substance (or simply a substance) matter that always has exactly the same composition. v Every sample has same properties. v Ex. Salt (Na. Cl); Pure Gold (Au) v v Can be classified into 2 categories: elements and compounds 1/23/2022 Chapter 2 Properties of Matter 3

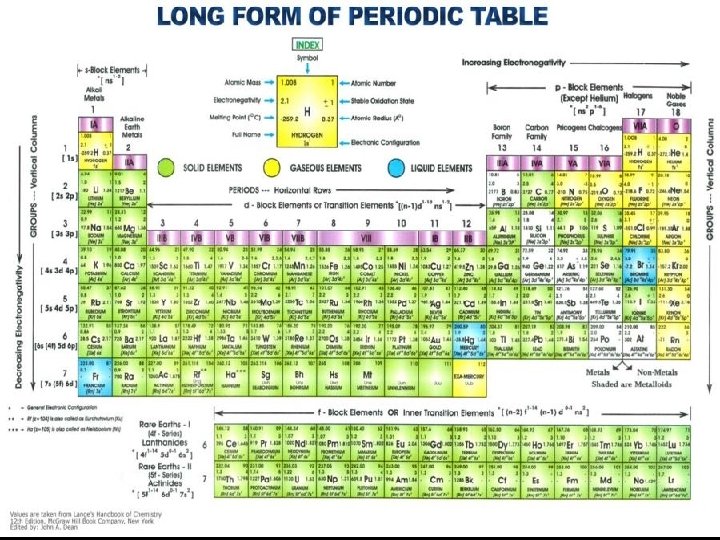
ELEMENTS (BOX 2) v Millions of known substances, but only about 100 elements. v Element - Substance that CANNOT be broken down into simpler substances. 1/23/2022 § Only 1 type of atom § No 2 elements contain same type of atom Chapter 2 Properties of Matter 4

ELEMENTS v At room temperature (20°C, 68°F, 300 K) 1/23/2022 n most are solids n some are gases (i. e. oxygen & nitrogen) n only 2 are liquids, bromine and mercury Chapter 2 Properties of Matter 5

ELEMENTS v 1813, Jons Berzelius suggested use of symbols to represent elements. v Helps scientists to communicate without confusion 1/23/2022 Chapter 2 Properties of Matter 6

ELEMENTS 1 or 2 letters: 1 st capitalized, 2 nd not Ex. Oxygen (O); Chlorine (Cl) v v Some based on Latin names n v Symbol for gold is Au because Latin name for gold is aurum Sometimes name gives clue to properties n 1/23/2022 hydrogen from Greek words hydro (water) and genes (forming) Chapter 2 Properties of Matter 7

1/23/2022 Chapter 2 Properties of Matter 8

COMPOUNDS (BOX 3) v Compound - substance that is made from two or more simpler substances and CAN be broken down into those simpler substances. Simpler substances are elements. Compounds always contain 2 or more elements joined in a fixed proportion – Ex. H 2 O v 1/23/2022 Chapter 2 Properties of Matter 9

COMPOUNDS (BOX 3) v Always contains 2 or more elements joined in a fixed proportion. v Properties of a compound differ from those of the substances from which they are made. 1/23/2022 Chapter 2 Properties of Matter 10

COMPOUNDS (BOX 3) v 1/23/2022 Example: Water (H 2 O) Chapter 2 Properties of Matter 11

COMPOUNDS (BOX 3) v Example: Water (H 2 O) § Compound of 2 elements hydrogen (H) and oxygen (O) H H O 1/23/2022 Chapter 2 Properties of Matter 12

COMPOUNDS (BOX 3) v Example: Water (H 2 O) § H 2 & O 2 both gases at room temperature § H 2 can fuel a fire & O 2 can keep a fire burning § 1/23/2022 H 2 O does not burn or help other substances burn. Chapter 2 Properties of Matter Water decomposes into its component elements, hydrogen & oxygen, when direct electrical current is passed through it. Volume of hydrogen (right) is twice the volume of oxygen (left). 13

COMPOUNDS n Example: silicon dioxide (Si. O 2) n n 1/23/2022 Always 2 O atoms for each silicon (Si) atom n O colorless gas n Si gray solid Si. O 2 is a colorless, transparent solid Compound found in most light-colored grains of sand Chapter 2 Properties of Matter 14

MIXTURES - (Box 4) n n n 1/23/2022 Matter that doesn‘t always have the same composition. Mixtures NEVER become uniform. Tend to retain some of properties of their individual substances. Chapter 2 Properties of Matter 15

MIXTURES (Box 4) n 1/23/2022 The properties of a mixture can vary because the composition of a mixture is not fixed. Chapter 2 Properties of Matter 16

MIXTURES (Box 4) n No matter how well you stir, substances that make up mixture will not be evenly distributed. n Can be classified by how well parts are distributed throughout mixture. 1/23/2022 n Homogeneous n Heterogeneous Chapter 2 Properties of Matter 17

MIXTURES: HOMOGENEOUS (BOX 5) n Homogeneous Mixture - substances are so evenly mixed that it is difficult to see one substance in the mixture from another. n Appears to have only one type of substance. n Example: swimming pool water H 2 O + substances that dissolve in water Coffee n 1/23/2022 Chapter 2 Properties of Matter 18

MIXTURES: HETEROGENEOUS (BOX 6) n Greek words hetero (“different”) and genus (“kind”) Heterogeneous Mixture - parts of the mixture are noticeably different from one another. Example: Salad, Rocks and Sand n 1/23/2022 Chapter 2 Properties of Matter 19

MIXTURES: HOMOGENEOUS & HETEROGENEOUS 1/23/2022 Chapter 2 Properties of Matter 20

MIXTURES: HOMOGENEOUS Stainless steel serving spoon is a homogeneous mixture of iron, chromium, and nickel. It is difficult to distinguish one substance from another. 1/23/2022 Chapter 2 Properties of Matter 21

MIXTURES: HETEROGENEOUS Sand is a heterogeneous mixture. It is not the same throughout. 1/23/2022 Chapter 2 Properties of Matter 22

Matter Anything that takes up space & has mass pure substances elements 1/23/2022 mixtures compounds homogeneous Chapter 2 Properties of Matter heterogeneous 23

Matter Anything that takes up space & has mass pure substances Matter that always has exactly the same composition elements compounds substances that substances CAN’T be made from 2 or broken down more simpler into simpler substances & substances. CAN be broken down. 1/23/2022 mixtures Matter that doesn’t always have the same composition homogeneous Evenly mixed and difficult to see one substance in the mixture from another. Chapter 2 Properties of Matter heterogeneous parts of the mixture are noticeably different from one another. 24

1/23/2022 Chapter 2 Properties of Matter 25

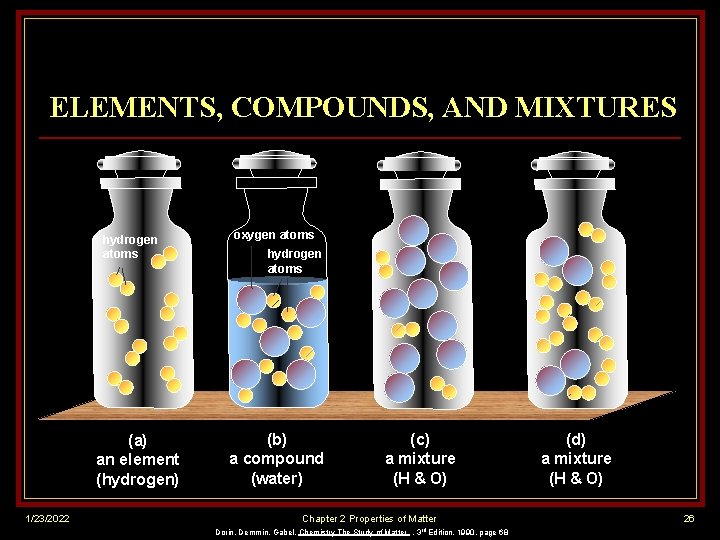
ELEMENTS, COMPOUNDS, AND MIXTURES hydrogen atoms (a) an element (hydrogen) 1/23/2022 oxygen atoms hydrogen atoms (b) a compound (water) (c) a mixture (H & O) Chapter 2 Properties of Matter Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3 rd Edition, 1990, page 68 (d) a mixture (H & O) 26

Chart Examining Some Components of Air Nitrogen consists of molecules consisting of two atoms of nitrogen: N 2 Oxygen consists of molecules consisting of two atoms of oxygen: O 2 Water consists of molecules consisting of two hydrogen atoms and one oxygen atom: H 2 O Argon consists of individual argon atoms: Ar Carbon dioxide consists of molecules consisting of two oxygen atoms and one carbon atom: CO 2 Neon consists of individual neon atoms: Ne Helium consists of individual helium atoms: He 1/23/2022 Chapter 2 Properties of Matter Zumdahl, De. Coste, World of Chemistry 2002, page 35 27

SOLUTIONS, SUSPENSIONS, AND COLLOIDS n The size of the particles in a mixture has an effect on the properties of that mixture. Key Concept: n Based on the size of its largest particles, a mixture can be classified as a solution, a suspension, or a colloid. 1/23/2022 Chapter 2 Properties of Matter 28

SOLUTIONS n Solution - mixture that forms when substances dissolve and form a homogeneous mixture n Example: Sugar dissolved in water Spoonful of sugar in a glass of hot water & stir, the sugar dissolves in the water homogeneous mixture of sugar & water. 1/23/2022 Chapter 2 Properties of Matter 29

SOLUTIONS n Properties of liquid solutions: Particles small so… n do not separate into distinct layers over time. n none of the substances in the solution are trapped in the filter. n can see through because light passes through them without being scattered in all directions. n Particles in a solution are too small to settle out of the solution, be trapped by a filter, or scatter light. 1/23/2022 Chapter 2 Properties of Matter 30

SUSPENSIONS n Suspension - heterogeneous mixture that separates into layers over time n Examples: sand mixed with water 1/23/2022 Chapter 2 Properties of Matter 31

SUSPENSIONS n 1/23/2022 Properties of a suspension: Because suspended particles are large… n suspended particles settle out of mixture (form layers) n can use a filter to separate out suspended particles n can scatter more light in all directions (cloudy) Chapter 2 Properties of Matter 32

COLLOIDS n Colloid - contains some particles that are intermediate in size between the small particles in a solution and the larger particles in a suspension n i. e. : homogenized milk, fog (water droplets in air) 1/23/2022 Chapter 2 Properties of Matter 33

COLLOIDS n 1/23/2022 Properties of a colloid: n does not separate into layers n can’t use a filter to separate the parts n scatters light (cloudy, opaque) Chapter 2 Properties of Matter 34

Key Concepts for Solutions, Suspensions & Colloids n 1/23/2022 Properties of liquid solutions: Particles small so… n Particles will not separate into layers over time. n None of the substances in the solution can be trapped in a filter. n Can see through the solution because light passes through them without being scattered in all directions. Chapter 2 Properties of Matter 35

Key Concepts for Solutions, Suspensions & Colloids n 1/23/2022 Properties of a suspension: Because suspended particles are large… n Suspended particles settle out of a mixture (Parts WILL separate). n CAN use a filter to separate out suspended particles. n Can scatter more light in all directions (cloudy) Chapter 2 Properties of Matter 36

Key Concepts for Solutions, Suspensions & Colloids n 1/23/2022 Properties of a colloid: n Does NOT separate into layers. n Can not use a filter to separate the parts. n Scatters light (cloudy, opaque). The light will be reflected by the larger particles in a colloid. Chapter 2 Properties of Matter 37