OrganicBio Chemistry Inorganic vs Organic Inorganic chemistry compound

Organic/Bio. Chemistry

Inorganic vs. Organic

Inorganic chemistry: • compound which usually does not contain carbon AND hydrogen –Ex. O 3, Na. Cl, H 2 O, CO 2

Inorganic Chemistry: • Compounds that do not need to contain: –Carbon: C AND –Hydrogen: H

Biochemical Compounds A. K. A Organic chemistry: • compounds usually found in Living or once living organisms • contains both Carbon and Hydrogen • other elements found in living things are: C, H, O, N, Phosphorous • Carbon = “element of LIFE”

Carbon: The element of LIFE

Biochemical Compounds Types of Organic Compounds found in all living things include: 1. Carbohydrates KNOW 2. Lipids THESE!!! 3. Protei. Ns 4. Nucleic Acids

Organic Chemistry: • Organic Compounds (Carbs, lipids, Nucleic Acids, and Proteins) are chemically bonded together or broken down by: – Dehydration Synthesis: – Hydrolysis:

Dehydration Synthesis: • process of joining (bonding) molecules to form large molecules called: MACROMOLECULES • macromolecules are formed by REMOVING WATER!! – Hence the name dehydration… as in dehydrate

Hydrolysis: • process of breaking down macromolecules into smaller molecules called MONOMERS • smaller molecules are formed by ADDING WATER!
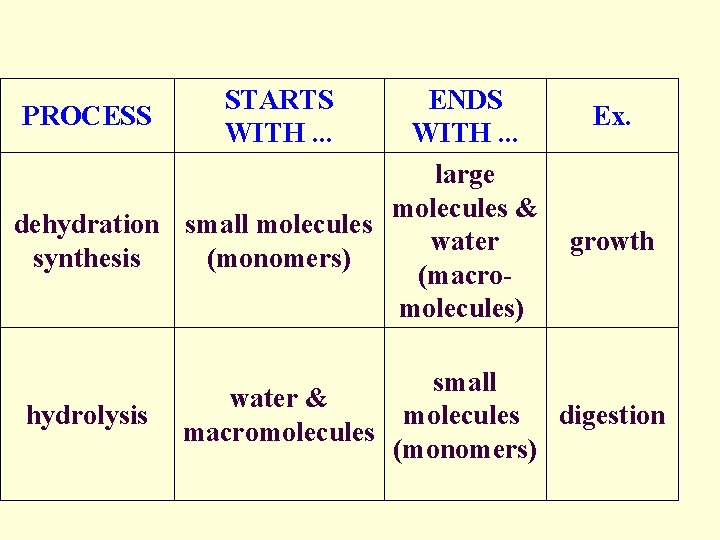
STARTS WITH. . . ENDS PROCESS Ex. WITH. . . large molecules & dehydration small molecules growth water synthesis (monomers) (macromolecules) hydrolysis small water & molecules digestion macromolecules (monomers)
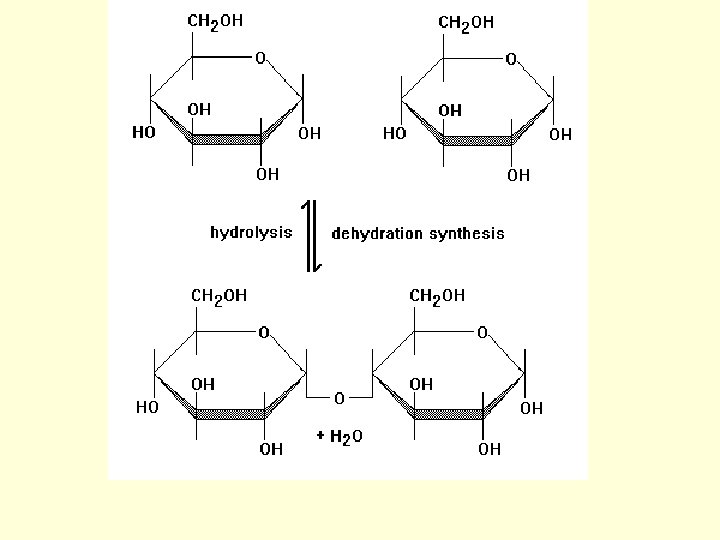

• dehydration synthesis and hydrolysis

Organic Compounds Cont… Carbohydrates: • Made of the elements Carbon, Hydrogen & Oxygen • Usually the Carbon, Hydrogen and Oxygen have a 1: 2: 1 ratio • Used in organisms for quick energy

Carbohydrate: a. Monosaccharide: –simple sugar (1 - ring) – “ose” ending = sugar –ex. Fructose, glucose:
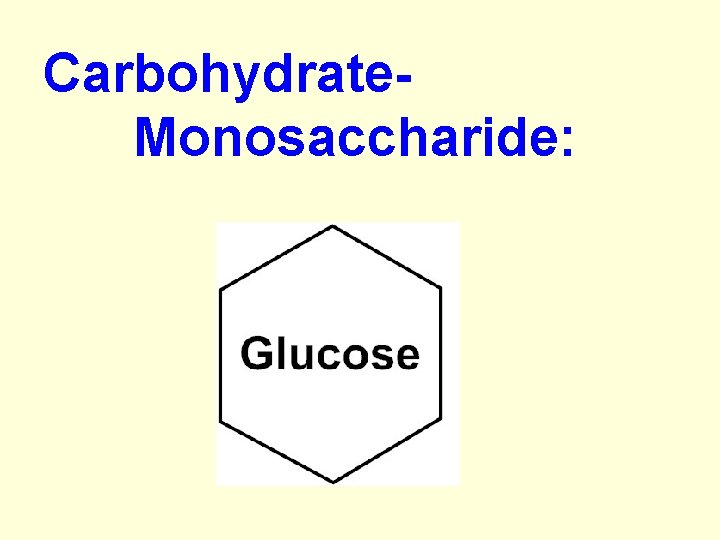
Carbohydrate- Monosaccharide:

Carbohydrates: b. Disaccharide • 2 simple sugars are chemically bonded together • ex. Sucrose, lactose
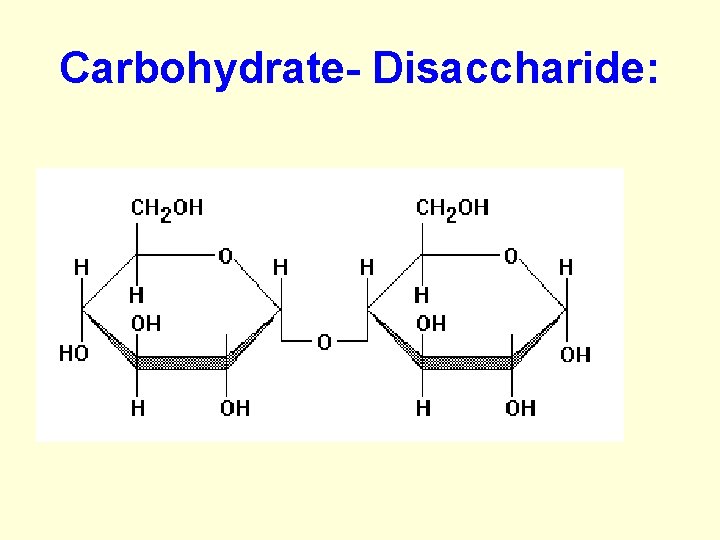
Carbohydrate- Disaccharide:

Carbohydrates: C. Polysaccharide: • many sugars chemically bonded together • form long chains = polymers • are macromolecules (lg molecules) • ex. Starches, Chitin, and cellulose (found in fruits and veggies)
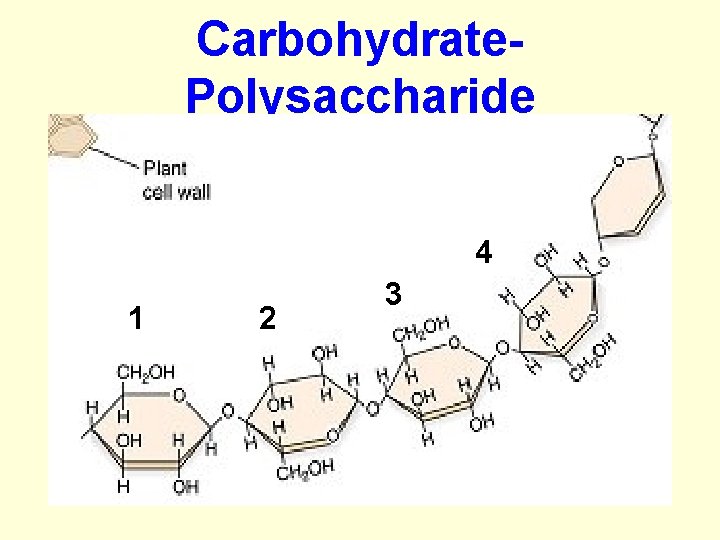
Carbohydrate- Polysaccharide 4 1 2 3
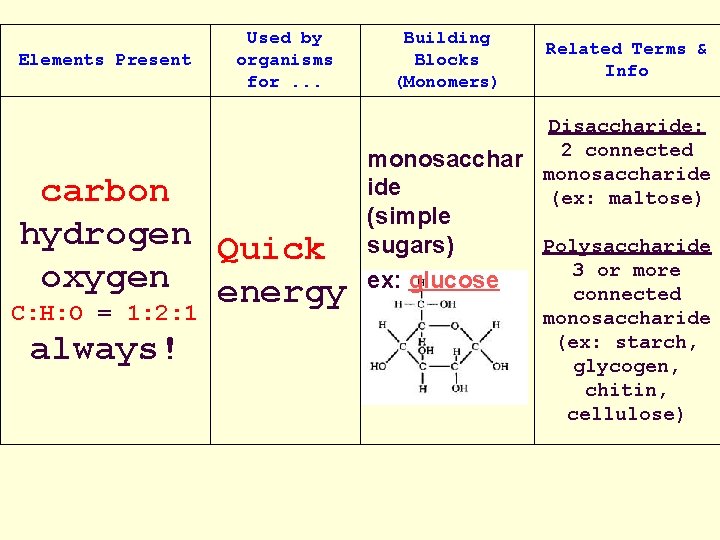
Elements Present Used by organisms for. . . carbon hydrogen Quick oxygen energy C: H: O = 1: 2: 1 always! Building Blocks (Monomers) monosacchar ide (simple sugars) ex: glucose Related Terms & Info Disaccharide: 2 connected monosaccharide (ex: maltose) Polysaccharide 3 or more connected monosaccharide (ex: starch, glycogen, chitin, cellulose)

Organic Compounds cont. Lipids: • Fats, cholesterol, waxes, oils • Lipids + H 2 O 3 fatty acids +glycerol • H to O ratio is much greater than 2: 1 • Do not EMULSIFY - break apart in H 2 O • Used as Stored energy in organisms
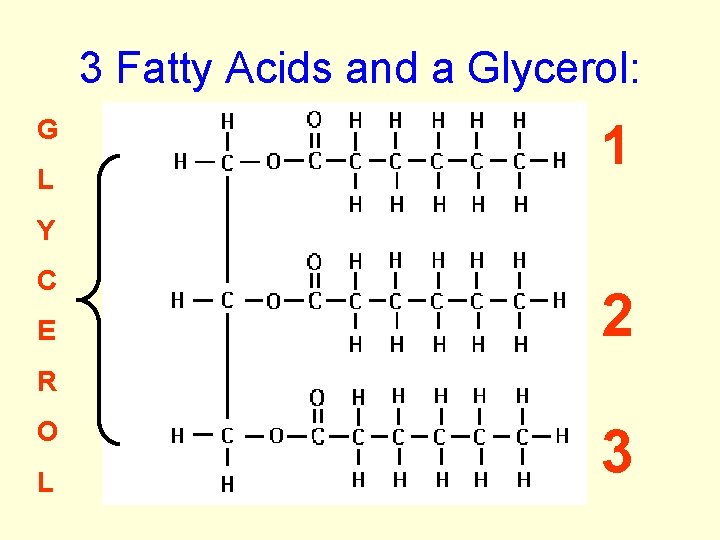
3 Fatty Acids and a Glycerol: G L 1 Y C E 2 R O L 3

Lipids • 2 classifications of fats: 1. Saturated fats 2. Unsaturated fats (which includes polyunsaturated fats)

Lipids: • Saturated fats: • solid at room temperature • linked to cardiovascular disease • have many H-C bonds

Lipids: Unsaturated fats: • not solids at room temp • not associated with cardiovascular disease • have 1 or more double C-C bonds • polyunsaturated fats have more than 1 double or triple C-C bond
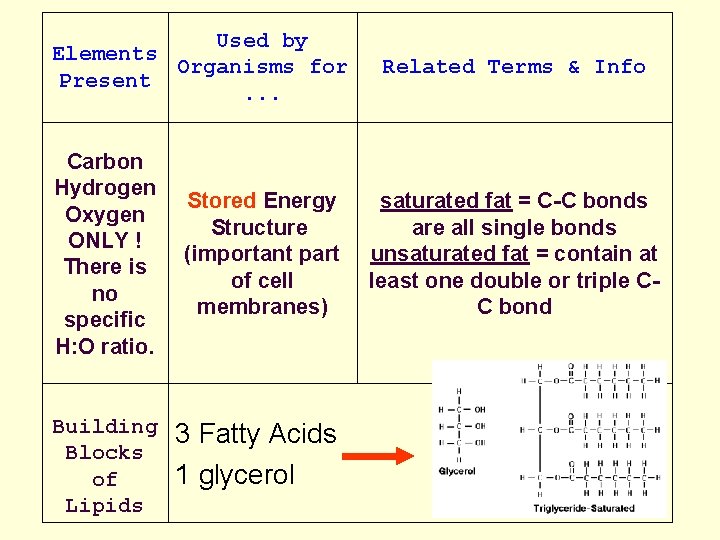
Used by Elements Organisms for Present. . . Related Terms & Info Carbon Hydrogen Stored Energy Oxygen Structure ONLY ! (important part There is of cell no membranes) specific H: O ratio. saturated fat = C-C bonds are all single bonds unsaturated fat = contain at least one double or triple CC bond Building Blocks of Lipids 3 Fatty Acids 1 glycerol

STOP!!! CK POINT Topic: “chemistry of life”: 1. What elements are found in carbohydrates and lipids? 2. Explain 2 differences between carbohydrates and lipids. 3. Describe the function for each? 4. How are monomers bonded together? Broken apart?

Organic Compounds: NUCLEIC ACIDS: DNA & RNA We will save the nitty, gritty details of DNA & RNA for later in the year (Genetics). But for now, you should know there functions & basic structure, and how DNA compares to RNA.

DNA & RNA are polymers (many units) --- long chains of smaller repeating units. The repeating unit (monomers) in nucleic acids is called a nucleotide.
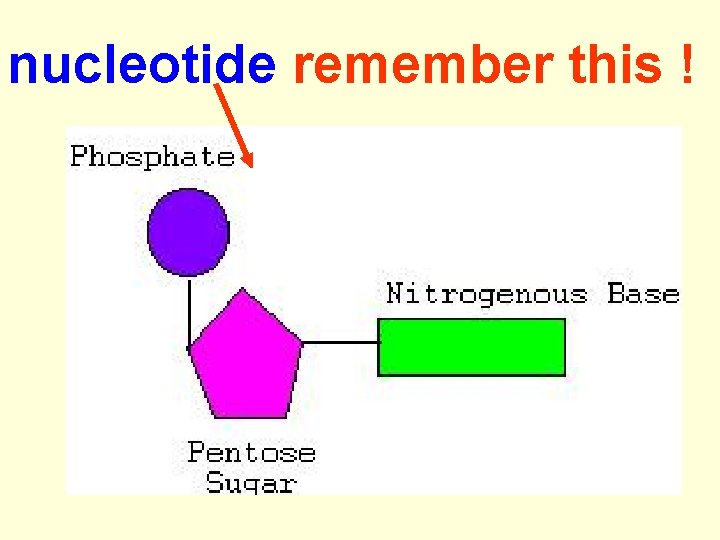
nucleotide remember this !
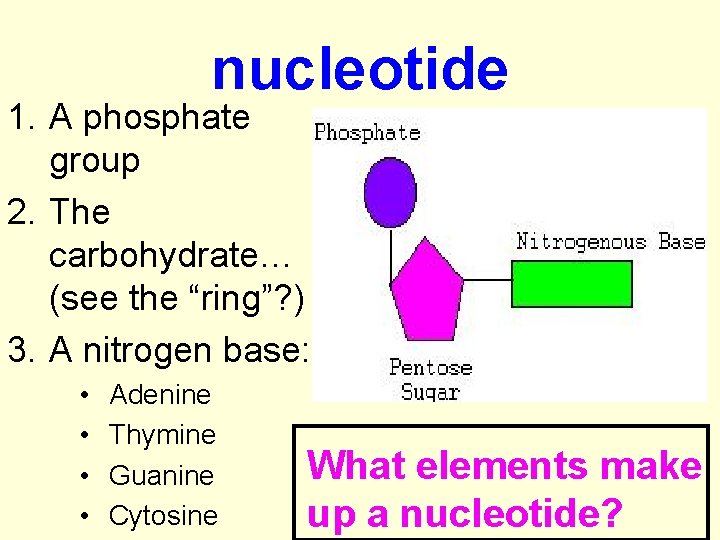
nucleotide 1. A phosphate group 2. The carbohydrate… (see the “ring”? ) 3. A nitrogen base: • • Adenine Thymine Guanine Cytosine What elements make up a nucleotide?
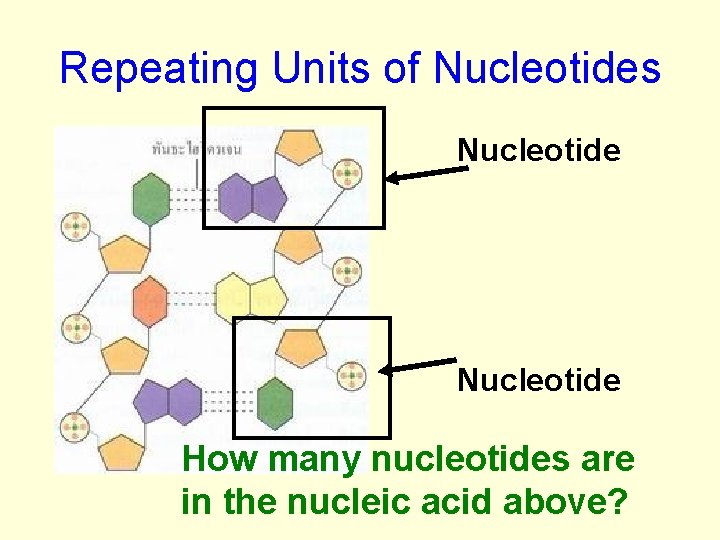
Repeating Units of Nucleotides Nucleotide How many nucleotides are in the nucleic acid above?
Nucleic Acid: DNA
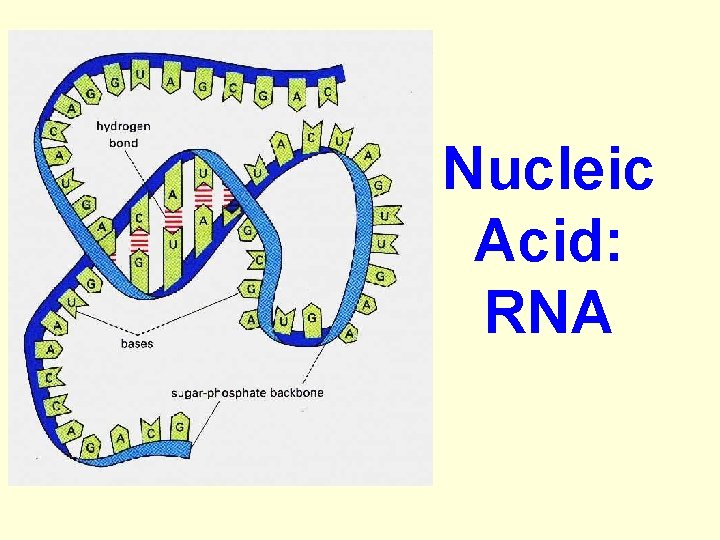
Nucleic Acid: RNA
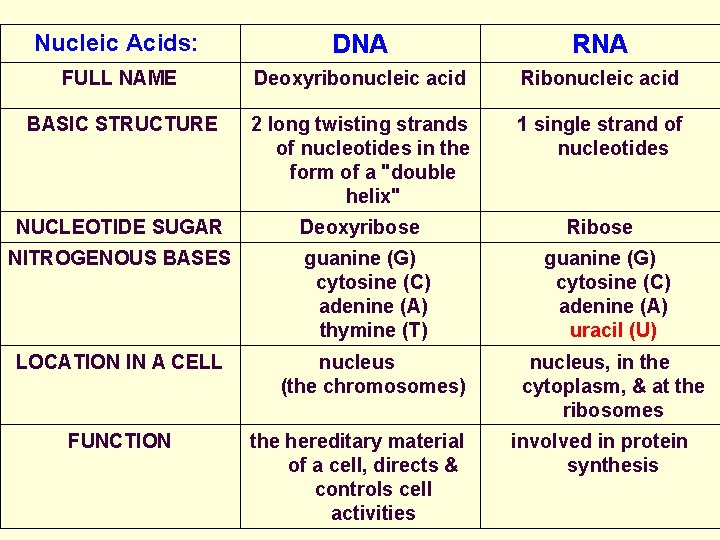
Nucleic Acids: DNA RNA FULL NAME Deoxyribonucleic acid Ribonucleic acid BASIC STRUCTURE 2 long twisting strands of nucleotides in the form of a "double helix" 1 single strand of nucleotides NUCLEOTIDE SUGAR Deoxyribose Ribose NITROGENOUS BASES guanine (G) cytosine (C) adenine (A) thymine (T) guanine (G) cytosine (C) adenine (A) uracil (U) LOCATION IN A CELL nucleus (the chromosomes) nucleus, in the cytoplasm, & at the ribosomes FUNCTION the hereditary material of a cell, directs & controls cell activities involved in protein synthesis

Organic Compounds: Protei. Ns' • Contain the elements: Carbon, Hydrogen, Oxygen and Nitrogen • take the shape of coils, helixes and globules • ex. Collagen, hormones, muscle tissue, enzymes, Hemoglobin
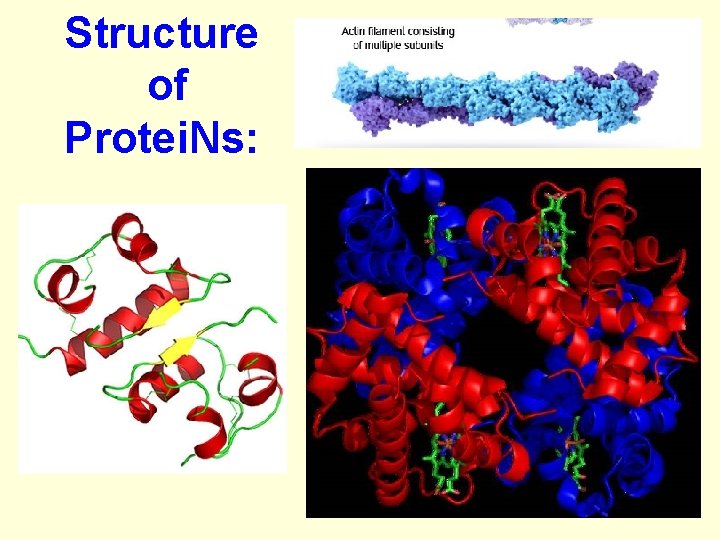
Structure of Protei. Ns:

Protei. Ns: Made up of basic building blocks MONOMERS called: AMINO ACIDS!!!!

Amino Acids: • are the structural units (monomers) of protein • bond together to form proteins • The bond between amino acids are called Peptide bonds

Amino Acids: • The order/sequence and size of amino acid determines the protein made • 2 amino acids bonded together = dipeptide • amino acids form long chains called a polypeptide chains

Elements Present carbon hydrogen oxygen & NITROGEN (sometimes Sulfur) monomers of Proteins: Used by organisms for. . . muscles enzymes antibodies hormones Pigments Hemoglobin AMINO ACIDS!!! Related Terms & Info peptide bond = the bond that holds amino acids together in protein molecules dipeptide = 2 connected amino acids polypeptide = 3 or more connected amino acids

STOP!!! CK POINT Topic: “NA and Protei. Ns”: 1. What are 2 types of Nucleic Acids? 2. What is the “monomer” of a nucleic acid? 3. Describe the primary functions of nucleic acids. 4. Identify elements found in Protei. Ns. 5. What is the monomer for Protei. Ns?

Group Activity: • Group leaders to get a marker • As a group, and when instructed, you will go to a poster in a designated area in the room • When the timer begins WRITE 1 ACCURATE FACT • Wait until instructed, move to the next poster. • As you move to each poster, read each statement then add an additional fact

Enzymes: • Are forms of protein!!!!!!! • Usually end in “ASE” • Are not changed during a chemical rxn (a substance affects a reaction w/o being changed is called a CATALYST)

Enzymes: • Are organic catalysts : – Control the rate of rxn within cells – Catalysts allow rxns to occur at a faster rate • Are used over and over • For each chem. rxn, there is a specific enzyme to initiate the rxn

HOW ENZYMES WORK: • Substrate– the substance the enzyme acts upon • Enzyme Active– the region on the enzyme • Substrate and Enzyme active site are specific to each other. • The substrate fits the enzyme active site like a puzzle called: enzymesubstrate complex

HOW ENZYMES WORK: • When the enzyme and substrate come together: • The enzyme: –may cause 2 molecules to join together –may cause bonds to break
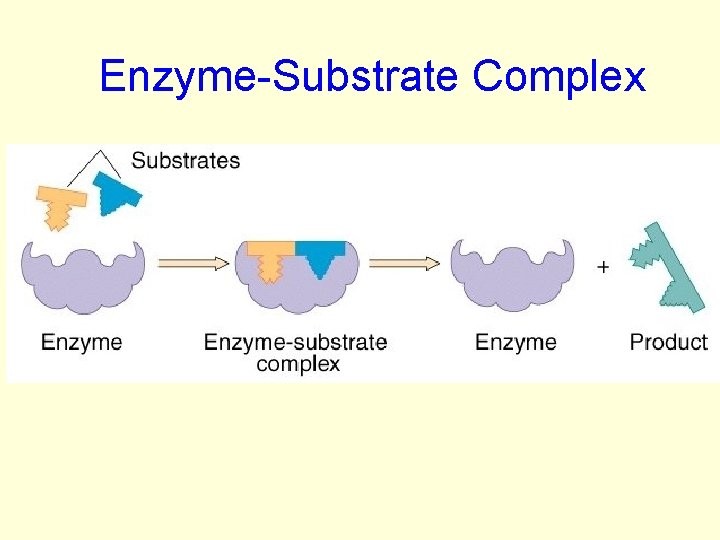
Enzyme-Substrate Complex
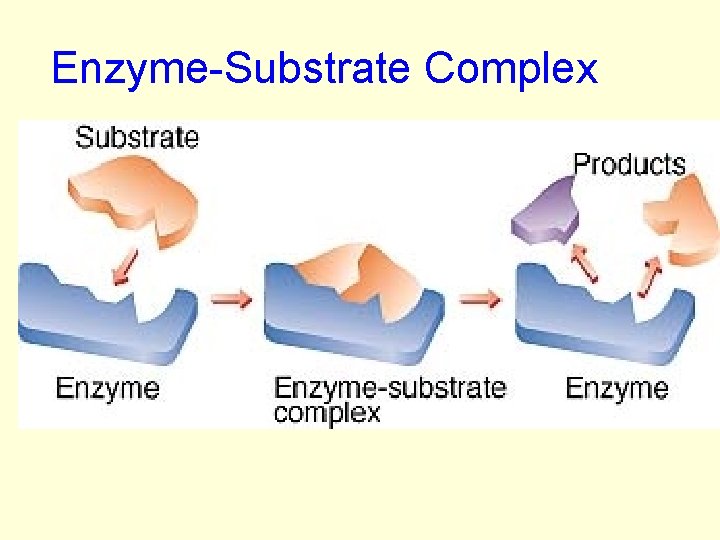
Enzyme-Substrate Complex

Factors Affecting Enzyme Action: 1. Concentration and surface area 2. Temperature 3. p. H 4. Co-enzymes
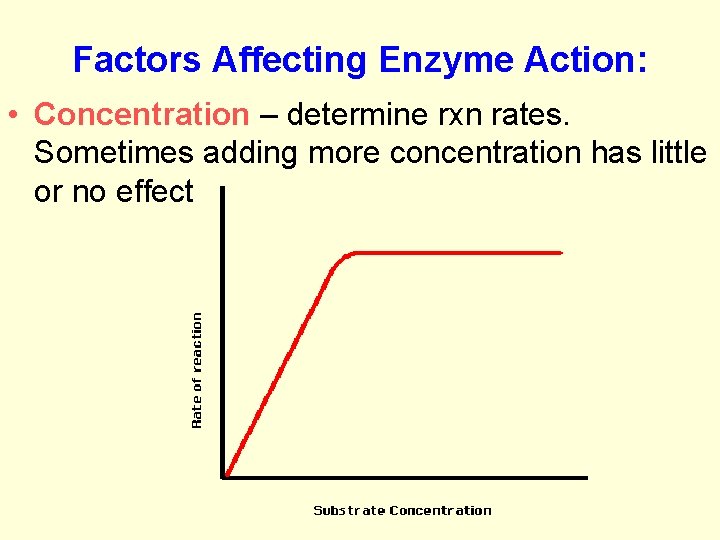
Factors Affecting Enzyme Action: • Concentration – determine rxn rates. Sometimes adding more concentration has little or no effect
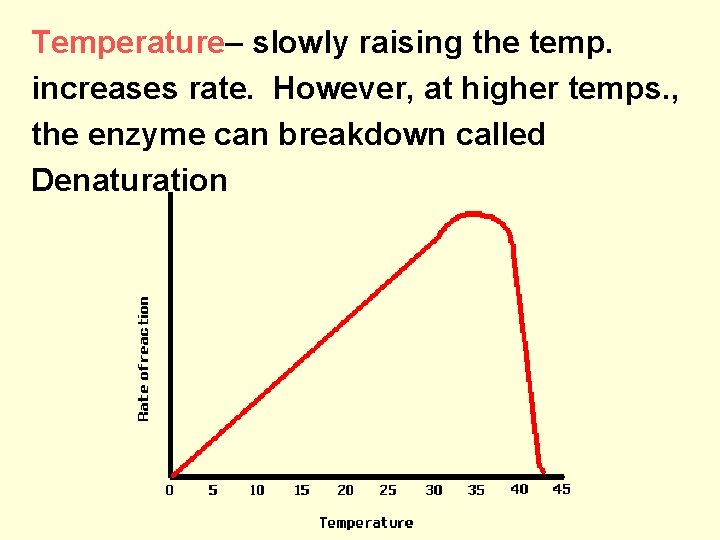
Temperature– slowly raising the temp. increases rate. However, at higher temps. , the enzyme can breakdown called Denaturation

• temp and enzymes
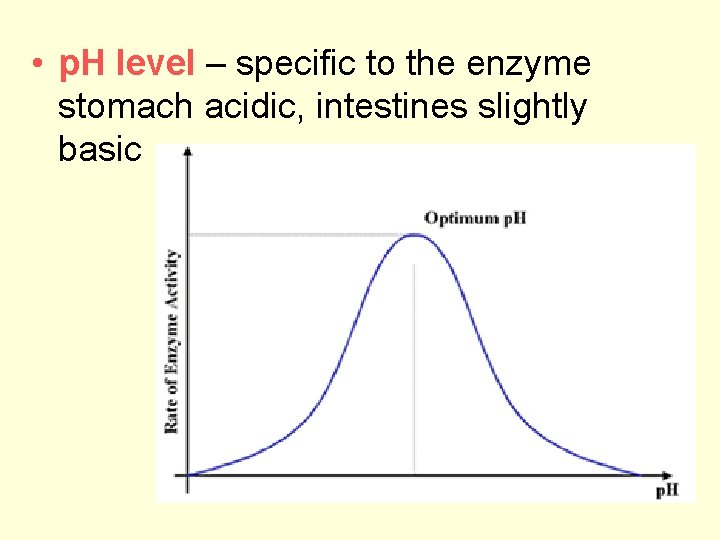
• p. H level – specific to the enzyme stomach acidic, intestines slightly basic

• Presence of coenzymes (vitamins) allows an enzyme to perform

Carbon Compounds 4 groups of carbon compounds found in living things are carbohydrates, lipids, nucleic acids, and protein. Living things use carbohydrates as their main source of energy. Plants and some animals also use carbohydrates for structural purposes. Lipids can be used to store energy. Some lipids are important parts of cell membranes and waterproofing. Nucleic acids store and transmit hereditary, or genetic, information. Proteins: • control the rate of reactions and regulate cell processes. • build tissues such as bone and muscle. Others transport materials or help to fight disease.

Chemical Reactions and Enzymes Chemical rxns always involve the breaking of bonds in reactants and the formation of new bonds in products. Cells use enzymes to speed up chemical reactions that take place in cells.
- Slides: 58