Classifying Chemical Reactions Chemistry Classifying Chemical reactions Essential










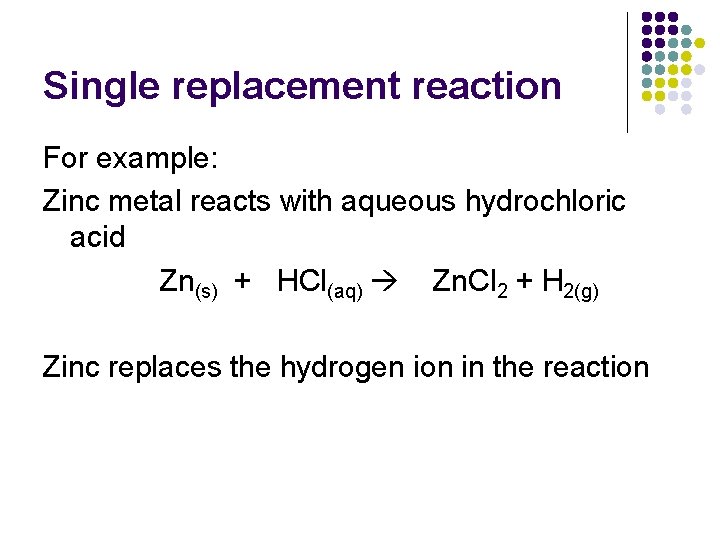































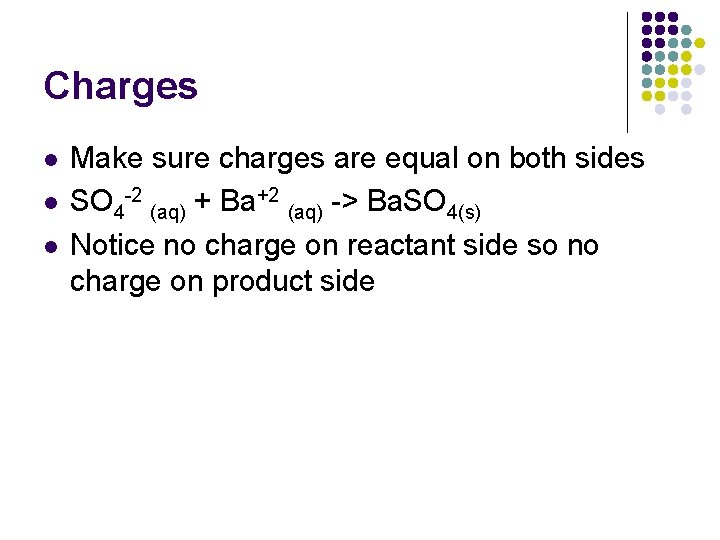

- Slides: 50

Classifying Chemical Reactions Chemistry

Classifying Chemical reactions Essential Questions 1. 2. Is there a way to organize chemical reactions to make them easier to understand? And maybe easier to balance?

Types of Reactions l l We classify reactions to make predicting the products easier Each type of reaction follows a certain pattern which make balancing easier: ) But…The 5 reactions we talk about are not the only types And …each reaction does not have to fit into just one type l Maybe more than one maybe none

5 types of reactions 1) 2) 3) 4) 5) Synthesis Decomposition Single Replacement Double Replacement Combustion Video: http: //www. youtube. com/watch? v=i. HHvx 1 VC_8 (1: 29 min)

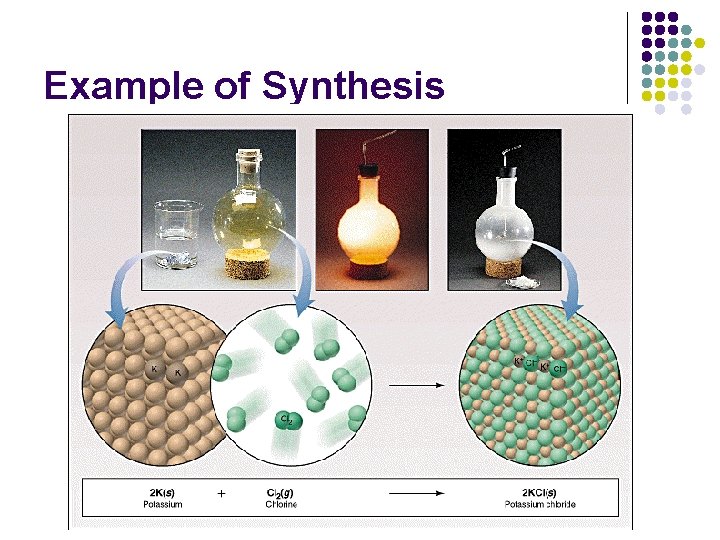
(1) Synthesis Reactions Synthesis means “to put together” A + B AB Two or more reactants react to form one product Examples: l l Ca. O (s) + H 2 O (l) Ca(OH)2 (s) 2 SO 2(g) + O 2 (g) 2 SO 3 (g)

Example of Synthesis

Let’s practice synthesis Predict the products. Write and balance the following synthesis reaction equations. • Sodium metal reacts with chlorine gas Na(s) + Cl 2(g) • Solid Magnesium reacts with fluorine gas Mg(s) + F 2(g) • Aluminum metal reacts with fluorine gas Al(s) + F 2(g)

(2) Decomposition Reactions Opposite of synthesis reactions A single compound breaks down into two or more elements or simpler compounds AB A + B Often need an input of energy (such as heat, light, or electricity) to make the reaction happen

Decomposition Reactions Example: Car air bags Sodium azide pellets decompose to produce sodium and nitrogen gas hat inflates the airbag! 2 Na. N 3(s) 2 Na(s) + 3 N 2(g)

Let’s practice decomposition Predict the products. Then, write and balance the following decomposition reaction equations: • Solid Lead (IV) oxide decomposes Pb. O 2(s) • Aluminum nitride decomposes Al. N(s)

Replacement Reactions Aka displacement reactions Two types: Single replacement and Double replacement

(3) Single Replacement l l l A single element reacts with a compound and displaces another less reactive element from the compound Like they switch places Example: Mg + Zn(NO 3)2 --> Mg(NO 3)2 + Zn
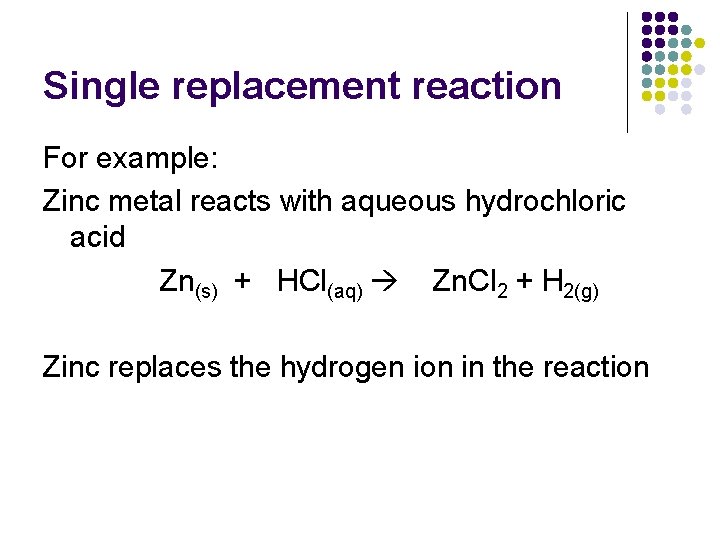
Single replacement reaction For example: Zinc metal reacts with aqueous hydrochloric acid Zn(s) + HCl(aq) Zn. Cl 2 + H 2(g) Zinc replaces the hydrogen ion in the reaction

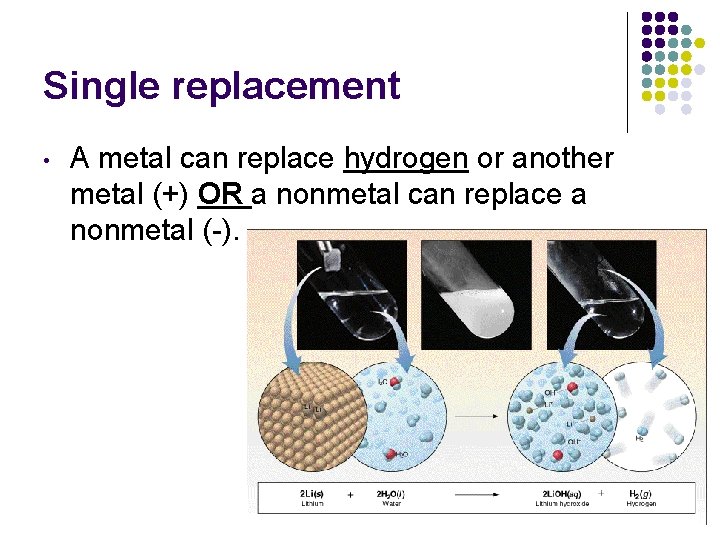
Single replacement • A metal can replace hydrogen or another metal (+) OR a nonmetal can replace a nonmetal (-).

Single Replacement Do single replacement reactions always occur? l NO! A metal will only replace a less reactive metal. l The activity series is a way to predict whether or not certain reactions will occur. l Any specific metal can replace any metal listed below it that is in a compound. l It cannot replace any metal listed above it!

What is the Activity Series? l l l Activity series a series of elements that have similar properties and that are arranged in descending order of chemical activity (meaning most reactive at the top) Elements can only “switch” places with elements below it on the activity series

Activity Series for Metals
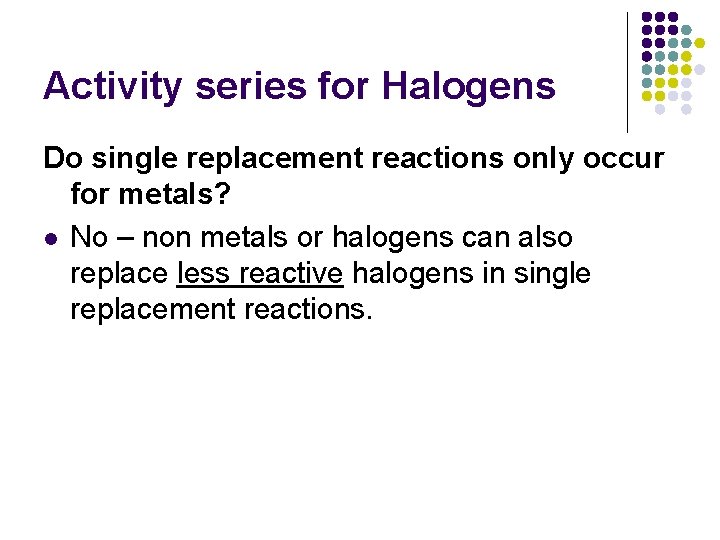
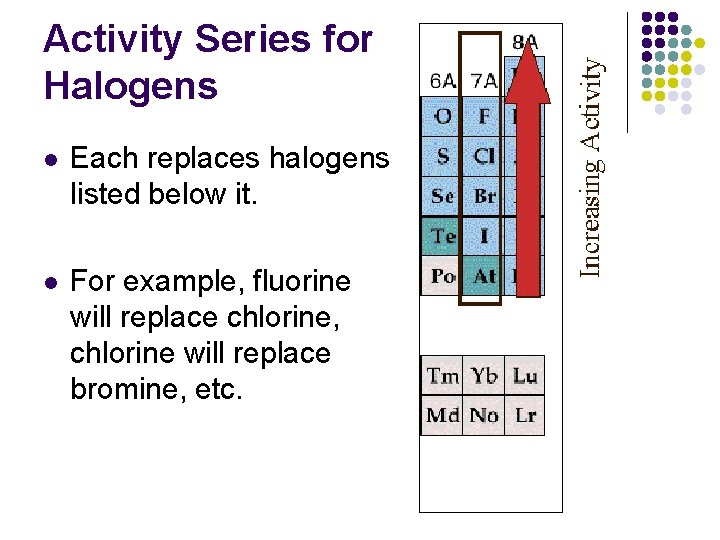
Activity series for Halogens Do single replacement reactions only occur for metals? l No – non metals or halogens can also replace less reactive halogens in single replacement reactions.

Activity Series for Halogens l Each replaces halogens listed below it. l For example, fluorine will replace chlorine, chlorine will replace bromine, etc.

Step for doing single replacement reactions 1) Identify the reactants Determine if element is metal or halogen Determine the element that might be displaced from compound 2) Check the activity series Determine which element is more active 3) Write the products and balance the equation If the more active element is already part of the compound then NO reaction will happen 4) Check your work Make sure the equation is balanced (just like always)

Check for Understanding Will the following react? If yes, write the balanced equation. If no, write NR for no reaction! 1. Cu + Zn. SO 4 (compare copper and zinc) 2. Fe + Cu. NO 3 (compare iron and copper) (iron takes +2 charge) 1. Al + Fe 2 O 3 (compare aluminum and iron)

Check for Understanding Will the following react? If yes, write the balanced equation. If no, write no reaction! 1. Cu + Zn. SO 4 no reaction 2. Fe + 2 Cu. NO 3 Fe(NO 3)2 + 2 Cu 3. 2 Al + Fe 2 O 3 Al 2 O 3 + 2 Fe

STOP! l Work on identifying and writing the first three types of reactions on the next worksheet both sides.

(4) Double- replacement Reactions l l When a metal replaces a metal in a compound a nonmetal replaces a nonmetal in a compound A reaction in which a gas, a solid precipitate, or a molecular compound forms from the apparent exchange of atoms or ions between two compounds Happens in aqueous solutions Must end up with a solid precipitate, gas, or molecular compound (like water)

Double replacement

Double Replacement • Think about it like “foil”ing in algebra, first and last ions go together + inside ions go together • Ag. NO 3(aq) + Na. Cl(s) Ag. Cl(s) + Na. NO 3(aq)

Special DD – Acid/Base Reaction l l Type of double displacement reaction that takes place when an acid and base react with each other. The H+ ion in the acid reacts with the OH- ion in the base, causing the formation of water. Product of this reaction is typically an ionic salt and water: HA + BOH ---> H 2 O + BA

Special DD – Acid/Base Reaction • • In double- displacement reactions water, as a product, is often written as HOH so you can more easily see the exchange of atoms/compounds HCl(aq) + Na. OH(aq) -> HOH(l) + Na. Cl(aq) This is a double replacement reaction • • The H and the Na changed partners You formed water (that’s one of the signs!!)

Practice double replacement 1. HCl(aq) + Ag. NO 3(aq) (hint: silver takes a +1 charge) 2. Ca. Cl 2(aq) + Na 3 PO 4(aq) (hint: You can tell that phosphate takes a -3 charge) 3. Pb(NO 3)2(aq) + Ba. Cl 2(aq) (hint: You can tell that lead takes a +2 charge) 4. HBr + Na. OH

(5) Combustion Reactions Combustion reaction l. Reaction of a hydrocarbon with oxygen l. AKA “burning!” l. Products are carbon dioxide and water vapor l. Releases energy in the form of heat & light l l Ex: the lighting in here is because of the combustion of coal Do you have any others?

Hydrocarbons l Just a fancy way to say the compound only has hydrogen and carbon in it Let’s try some: ___C 3 H 8 + ___O 2 ___CO 2 + ___H 2 O ___C 5 H 12 + ___O 2 ___ C 10 H 22 + ___ O 2

How to identify the type of reaction! Follow this series of questions. When you can answer "yes" to a question, then stop! 1) Does your reaction have oxygen as one of its reactants and carbon dioxide and water as products? If yes, then it's a combustion reaction 2) Does your reaction have two (or more) chemicals combining to form one chemical? If yes, then it's a synthesis reaction

3) Does your reaction have one large molecule falling apart to make several small ones? If yes, then it's a decomposition reaction 4) Does your reaction have any molecules that contain only one element? If yes, then it's a single displacement reaction 5) If you haven't answered "yes" to any of the questions above, then you've got a double displacement reaction

Check for understanding List what type the following reactions are: 1) Na. OH + KNO 3 --> Na. NO 3 + KOH 2) CH 4 + 2 O 2 --> CO 2 + 2 H 2 O 3) 2 Fe + 6 Na. Br --> 2 Fe. Br 3 + 6 Na 4) Ca. SO 4 + Mg(OH)2 --> Ca(OH)2 + Mg. SO 4 5) Pb + O 2 --> Pb. O 2 6) Na 2 CO 3 --> Na 2 O + CO 2

1) 2) 3) 4) 5) 6) double displacement combustion single displacement double displacement synthesis decomposition

Pharaoh's Snakes l Now for something fun l Check this out… http: //www. youtube. com/watch? v=ritaljhhk 7 s

Pharaoh's Snakes Igniting mercury(II) thiocyanate Hg(SCN)2 causes it to decompose into an insoluble brown mass that is primarily tricarbon tetranitride, mercury(II) sulfide and carbon disulfide. Write the decomposition reaction and balance it! 2 Hg(SCN)2 → 2 Hg. S + CS 2 + C 3 N 4

Pharaoh's Snakes Then flammable carbon disulfide combusts in oxygen to produce carbon dioxide and sulfur dioxide: Write the combustion reaction and balance it! CS 2 + 3 O 2 → CO 2 + 2 SO 2

Stop! Let’s practice what we have learned with some worksheets!

“Writing Net Ionic Equations” Goals l l Write total ionic equations for reactions in aqueous solutions Identify spectator ions and write ionic equations for reactions in aqueous solutions

Ionic Equations l l l When ionic compounds dissolve in water, the ions separate form each other and spread throughout the solution So when you see KI(aq) what it really means is K+(aq) + I-(aq) Or if you see Pb(NO 3)2(aq) it means Pb+2(aq) + 2 NO 3 -(aq)

More Correct l l When we react the two compounds on the last slide we would do this: KI(aq)+ Pb(NO 3)2(aq) -> Pb. I 2(s) + 2 KNO 3(aq) But we should put the charges on them and then balance the charges and masses K+(aq) + I-(aq) + Pb+2(aq) + 2 NO 3 -(aq) -> Pb. I 2(s) + 2 K+ (aq) + 2 NO 3 -(aq)

Most Correct l When you mix two solutions all of the ions are present in the combined solution l l l But not all of the ions will react with each other Spectator ions=> ions that are present in a solution in which a reaction is taking place but that do not participate in the reaction These spectator ions can be taken out of the total ionic equation

Example l Bottom of page 286

Writing Net Ionic Equations l These equations only show the compounds that change

Displacement Net Ionic l Happens in displacement reactions too l Zn(s)+Cu+2(aq)+SO 4 -2(aq)-> Cu(s)+Zn+2 (aq)+ SO 4 -2(aq) l Which is the spectator ion? How would we correctly write this? l

Skills Toolkit Page 288 l 1) List what you know l l l 2) Write a balanced equation l l l Predict products Include states (next slide shows how to know!!!) 3) Write total ionic equation l l l ID each chemical described ID type of reaction Write separated aqueous ions for each aqueous ionic substance DO NOT split up anything that is a solid, liquid, or gas 4) Find the net ionic equation l l Cancel out spectator ions Double check numbers and charges

Solubility l l l All group 1 compounds and NH 4 All nitrates All halides (but Ag+ and Pb+2) All sulfates (but Ca+2, Sr+2, Ba+2, Hg+2, and Pb+2) All carbonates (except group 1 and NH 4) are INSOLUBLE!
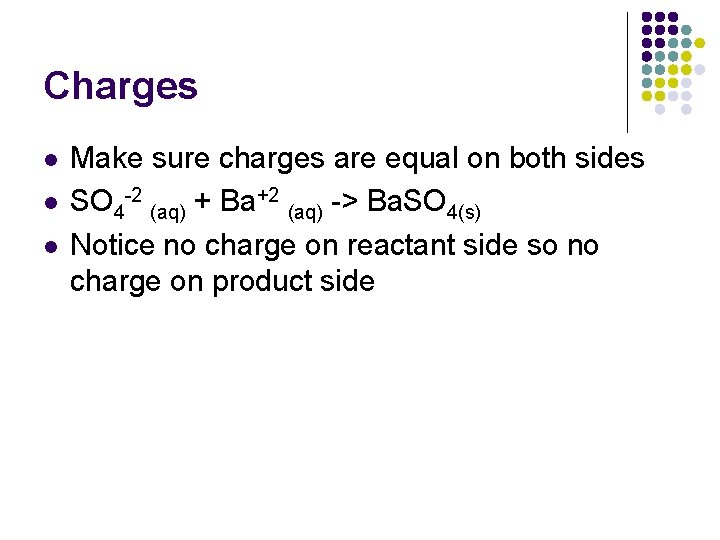
Charges l l l Make sure charges are equal on both sides SO 4 -2 (aq) + Ba+2 (aq) -> Ba. SO 4(s) Notice no charge on reactant side so no charge on product side

Practice l l Br 2(l) + Na. I(aq) -> Na. Br(aq) + I(s) Br 2(l) + 2 Na+(aq) + 2 I-(aq) -> 2 Na+(aq) + 2 Br -(aq) + I 2(s)