Properties Of Matter Classifying Matter Phases of Matter
































































- Slides: 69



Properties Of Matter Classifying Matter Phases of Matter Separating Matter Energy Take a Chance $400 $400 $800 $800 $1200 $1200 $1600 $1600 $2000 $2000

Final Jeopardy Particle Diagrams

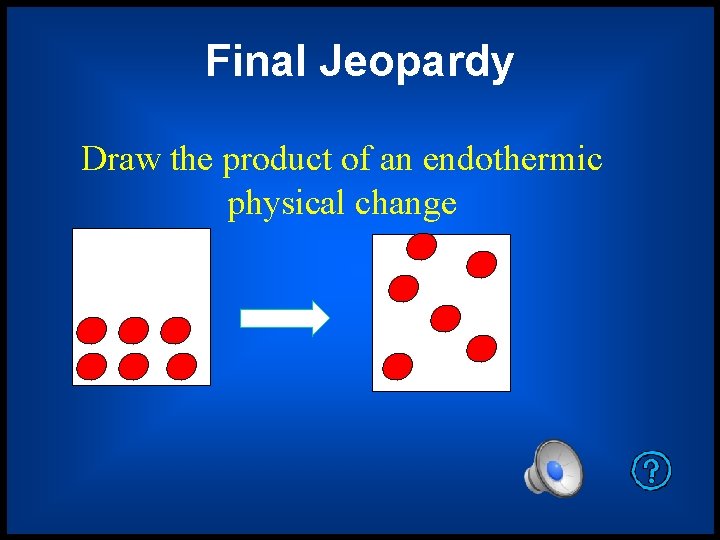
Final Jeopardy Draw the product of an endothermic physical change

$200 answer question

$400 answer question

$600 answer question

$800 answer question

$1000 answer question

$200 answer question

$400 answer question

$600 answer question

$800 answer question

$1000 answer question

$200 answer question

$400 answer question

$600 answer question

$800 answer question

$1000 answer question

$200 answer question

$400 answer question

$600 answer question

$800 answer question

$1000 answer question

$200 answer question

$400 answer question

$600 answer question

$800 answer question

$1000 answer question

$200 answer question

$400 answer question

$600 answer question

$800 answer question

$1000 answer question

$400 3. melting point of 1930 C Which of the following would be considered a physical property? 1. reacts with an acid 2. flammable 3. melting point of 1930 C 4. corrodes easily

$800 4. flammable Which of the following would be considered a chemical property 1. malleable 2. boils at 1000 C 3. low melting point 4. flammable

$1200 physical Is dissolving Na. Cl in water a physical or chemical change?

$1600 Chemical = identity Physical = appearance (look) A chemical change occurs when the _______ of a substance changes and a physical change occurs when the _____ of a substance changes.

$2000 Formation of a gas (bubble), color change, and formation of a percipitate Two evidences that a chemical reaction has occurred are the presence of heat and light. What are the other 3?

$400 Pure substance Matter can be classified as a pure substance or a mixture. If it can NOT be physical separated, it is a ______

$800 3. Salt and sand Which of the following is a mixture? 1. salt (Na. Cl) 2. gold (Au) 3. salt and sand (Na. Cl + Si. O 2) 4. helium gas (He (g))

$1200 Elements: He, N 2, Ag Compounds: Na. Cl Element or Compound 2. Place the following under the correct column salt (Na. Cl) Nitrogen (N 2) silver (Ag) helium gas (He (g))

$1600 air Which of the following is a mixture? 1. salt (Na. Cl) 2. sugar (C 12 H 22 O 11) 3. lead (Pb) 4. air

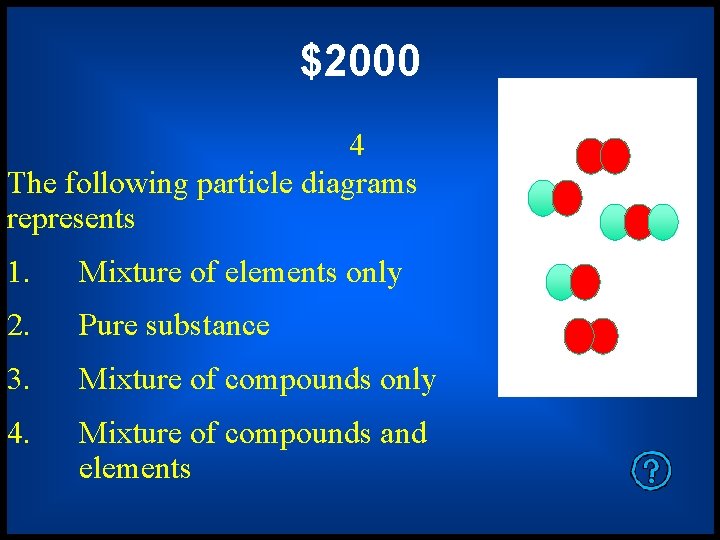
$2000 4 The following particle diagrams represents 1. Mixture of elements only 2. Pure substance 3. Mixture of compounds only 4. Mixture of compounds and elements

$400 Has mass and takes up space Define Matter

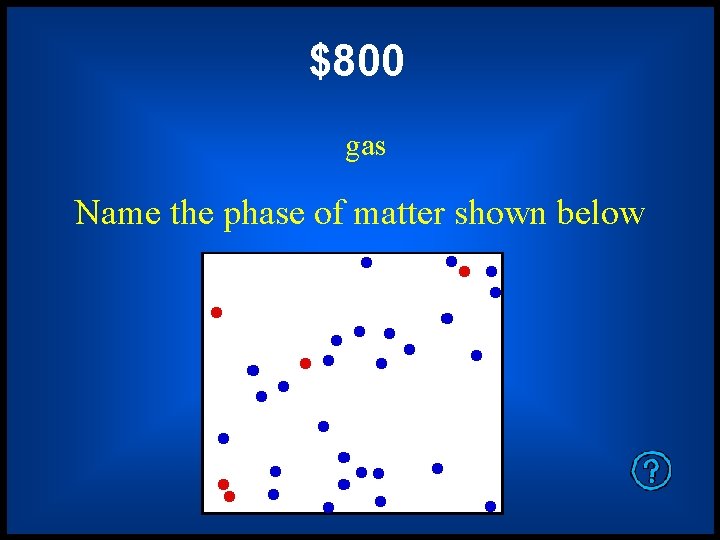
$800 gas Name the phase of matter shown below

$1200 3 Solids have 1. definite shape and indefinite volume 2. indefinite shape and indefinite volume 3. definite shape and definite volume 4. indefinite shape and definite volume

$1600 Definite volume and indefinite shape Use the word(s) indefinite and/or definite Liquids have ____ volume _____ shape

$2000 1. Physical change 2. freezing 2 parts 1. When a liquid becomes a solid, what type of change has taken place (physical or chemical)? 2. What is this phase change called?

$400 sorting Peas and carrots can be separated by this technique

$800 Filtration This mixture can be separated using which technique?

$1200 3. Salad dressing An example of a heterogeneous mixture is: 1. water 2. vegetable oil 3. salad dressing 4. vinegar

$1600 physical Mixture can be separated based on _____ properties

$2000 homogeneous When salt it dissolved in water, the individual salt particles are too small to see, so this type of mixture is called a _______ mixture

$400 energy _____ is the ability to do work

$800 J (joule) The SI unit of energy is _______

$1200 Exothermic Endothermic or Exothermic When energy exits the system and warms the environment, it’s considered to be _______

$1600 endothermic Endothermic or Exothermic When energy enters the system it (and it becomes warm) and the environment cools, it’s considered to be _______

$2000 1. Endo 2. Exo 3. Exo Name the following as endothermic or exothermic 1. Solid liquid 2. Gas liquid 3. Liquid solid

$400 compound A substance that consists of only 2 or more type of elements is called a _______

$800 element A substance that consists of only one type of atom is called a(n) ______

$1200 PE State whether the following describe potential energy (PE) or kinetic energy (KE) a carrot ____________

$1600 PE State whether the following describe potential energy (PE) or kinetic energy (KE) a dam holding back a river of water ____________

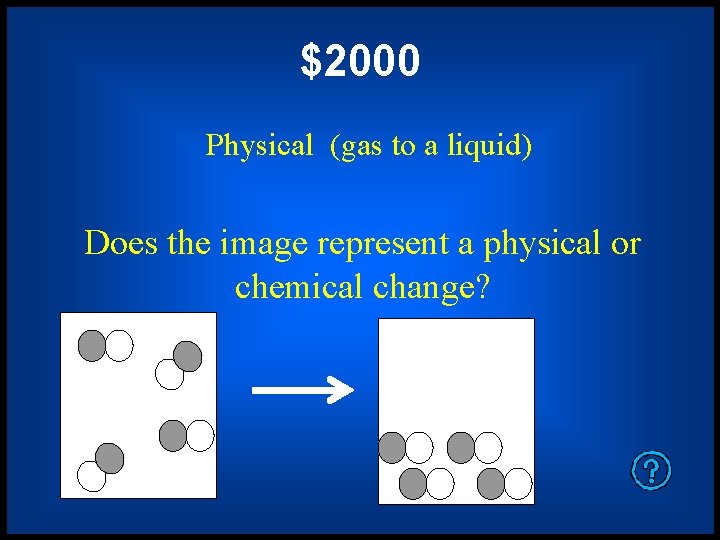
$2000 Physical (gas to a liquid) Does the image represent a physical or chemical change?

Daily Double answer question

Daily Double answer question

Daily Double answer question

The Jeopardy champion!