PROPERTIES AND CHANGES OF MATTER S 8 P
















- Slides: 19

PROPERTIES AND CHANGES OF MATTER S 8 P 1. Students will examine the scientific view of the nature of matter. d. Distinguish between physical and chemical properties of matter as physical (i. e. , density, melting point, boiling point) or chemical (i. e. , reactivity, combustibility). e. Distinguish between changes in matter as physical (i. e. , physical change) or chemical (development of a gas, formation of precipitate, and change in color). SPS 2. Students will explore the nature of matter, its classifications, and its system for naming types of matter. a. Calculate density when given a means to determine a substance’s mass and volume. SPS 7. Students will relate transformations and flow of energy within a system. c. Determine the heat capacity of a substance using mass, specific heat, and temperature.

PHYSICAL PROPERTIES q A characteristic of a substance that can be observed or measured without changing its identity q Used to identify matter q Examples: Boiling point, melting point, odor, shape, hardness, color, magnetism, electrical conductivity, mass, weight, volume, texture

PHYSICAL PROPERTIES Other Examples q Thermal conductivity- the rate at which a substance transfers heat q State- the physical form in which a substance exists: solid, liquid, gas, and plasma q Density- the amount of matter in a given space or volume (m/v) q Solubility- the ability of a substance to dissolve in another substance q Ductility- the ability of a substance to be pulled into a wire q Malleability- the ability of a substance to be rolled or hammered into thin sheets q Magnetism – the ability to attract magnetically

PHYSICAL PROPERTIES

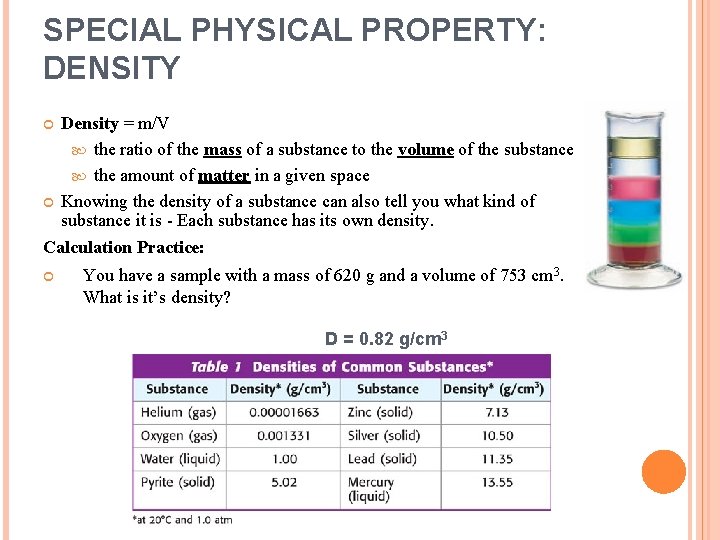
SPECIAL PHYSICAL PROPERTY: DENSITY Density = m/V the ratio of the mass of a substance to the volume of the substance the amount of matter in a given space Knowing the density of a substance can also tell you what kind of substance it is - Each substance has its own density. Calculation Practice: You have a sample with a mass of 620 g and a volume of 753 cm 3. What is it’s density? D = 0. 82 g/cm 3

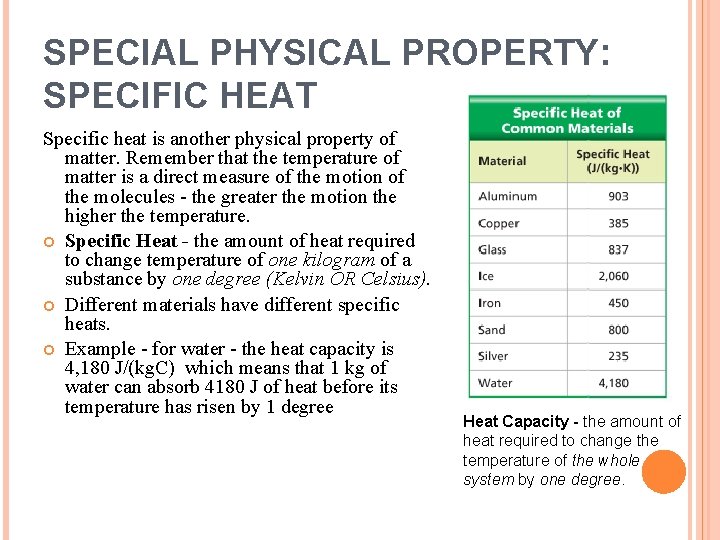
SPECIAL PHYSICAL PROPERTY: SPECIFIC HEAT Specific heat is another physical property of matter. Remember that the temperature of matter is a direct measure of the motion of the molecules - the greater the motion the higher the temperature. Specific Heat - the amount of heat required to change temperature of one kilogram of a substance by one degree (Kelvin OR Celsius). Different materials have different specific heats. Example - for water - the heat capacity is 4, 180 J/(kg. C) which means that 1 kg of water can absorb 4180 J of heat before its temperature has risen by 1 degree Heat Capacity - the amount of heat required to change the temperature of the whole system by one degree.

CHANGES IN THERMAL ENERGY Energy gained or lost by a material is related to its mass, change in temperature and specific heat. Formula: Q = m x D T x C Change in energy in joules (Q) = mass in kg X D T in C X specific heat Problem: How much heat is required to raise the temperature of 5 kg water by 10 degrees Celsius? Q = 5 kg X 4, 180 X 10 C Q = 209, 000 J

MEASURING HEAT CHANGES A calorimeter is a device used to measure the quantity of heat transferred to or from an object. Calorimetry is the science of determining the changes in energy of a system by measuring the heat exchanged with the surroundings. Calorimetry can be used to find relative specific heat values when different substances are mixed with water. A simple design for calorimeter is an insulated container filled with a liquid, usually water. When a hot object is placed in the calorimeter, heat energy is transferred into the water and it heats up. How much energy is transferred can provide information of an object’s specific heat.

A NOTE ABOUT TEMPERATURE & SPECIFIC HEAT Remember that temperature is a measure of the average kinetic energy of the particles in a sample of matter Fahrenheit is an outdated system of recording temperature Celsius is the metric unit of measure and used in science – based on freezing and boiling point of water Kelvin scale uses the same increments as the Celsius scale but begins at absolute zero which is -273 Celsius In determining Specific Heat it doesn’t matter if the units are Kelvins or degrees Celsius as you are measuring CHANGE and both use the same increments but you CANNOT use Fahrenheit as the increments are different!

PHYSICAL CHANGES A change in the form of a substance without changing its identity (chemical properties) When any physical properties of a substance are changed, a physical change has occurred. Do not form new substances Examples Changing the state of matter: freezing water Changing the shape of an object: sanding a piece of wood Dissolving salt in water

PHASE CHANGES ARE PHYSICAL CHANGES

OTHER PHYSICAL CHANGES

CHEMICAL PROPERTIES A characteristic of a substance that describes its ability to undergo a chemical change Chemical changes involve making or breaking of bonds between atoms Description of matter based on its ability to change into new substances Properties are ONLY observable during a chemical change/reaction Examples Flammability- the ability of a substance to burn Combustibility- the ability of a substance to ignite Reactivity- the ability of two or more substances to combine and form one or more new substances with chemicals with light

PHYSICAL VS. CHEMICAL PROPERTIES You can observe physical properties without changing the identity of the substance. Ex. hardness of wood Chemical properties are not always easy to observe. Ex. You can see that wood is flammable only while it is burning no longer wood

CHEMICAL CHANGES A change that occurs when one or more substances change into entirely new substances with different properties A change in the identity of a substance Ex: Burning, rusting, alka-seltzer tablets bubbling, souring of milk, tarnish on silver or copper, cooking food, digestion Signs a chemical change has occurred: a change in color, odor, production of heat, production of a gas or solid (precipitate), fizzing and foaming, and sound or light being given off Chemical properties describe the ability or potential to change whereas the chemical change is the reaction itself.

EXAMPLES OF CHEMICAL CHANGES

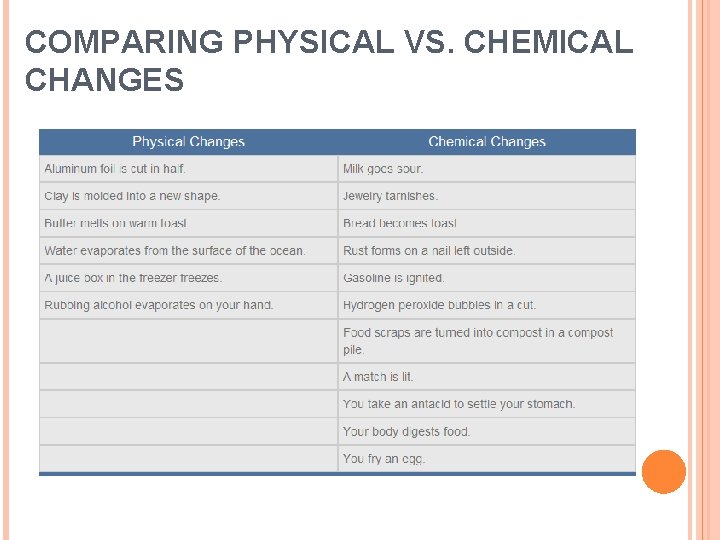
COMPARING PHYSICAL VS. CHEMICAL CHANGES A Change in Composition Physical changes do not change the composition of a substance while chemical changes do. Reversing Changes Many physical changes are easily reversed, but most chemical changes cannot easily be reversed. Ex. An ice cube can be melted and frozen Ex: Baking a cake - you cannot reverse the chemical reactions that have occurred between the ingredients.

COMPARING PHYSICAL VS. CHEMICAL CHANGES

ACTIVITIES! Physical & Chemical Properties & Changes in Matter Worksheet Specific Heat Gizmo Specific Heat Worksheet