Chapter 4 Basic Food Chemistry The Nature of

Chapter 4 Basic Food Chemistry: The Nature of Matter

Objectives: Explain the difference between pure substances and mixtures. Compare physical and chemical reactions in laboratory experiments.
• Discussion question: How does chemistry and food go together?

Name the components that make up foods that you eat. Brainstorm ideas.

Look at a food label and discuss the relationship between chemistry and food. Write down the ingredients you do not know how to pronounce or you do not know what it is!!!

Vocabulary Words Elements Atom Subatomic Particles Compounds Pure Substances Inorganic compounds Molecule Organic compounds Mixture Homogeneous mixture Heterogeneous mixture Solution Solute Solvent Physical change Phase change Chemical change

The Basic Nature of Matter • Everything you encounter, whether plant, animal or mineral is made up of atoms. • Atom: The smallest unit of any elemental substance that maintains the characteristics of that substance.

• Subatomic Particles = a small part of an atom – Proton, neutron, electron, nucleus
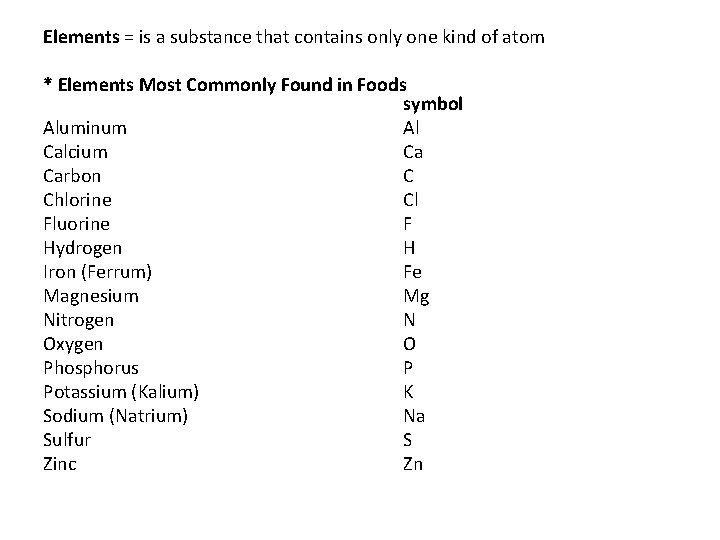
Elements = is a substance that contains only one kind of atom * Elements Most Commonly Found in Foods symbol Aluminum Al Calcium Ca Carbon C Chlorine Cl Fluorine F Hydrogen H Iron (Ferrum) Fe Magnesium Mg Nitrogen N Oxygen O Phosphorus P Potassium (Kalium) K Sodium (Natrium) Na Sulfur S Zinc Zn

The Classification of Matter – matter can be classified into the two general categories of pure substances and mixtures. Pure Substances is the matter in which all the basic units are the same. Can be either: Elements - those needed for good health - examples: iron, calcium & potassium

Compounds - most of the pure substances food scientists work with are compounds -examples: salt (sodium chloride) & baking soda (sodium bicarbonate)

• -chemists divide they study of compounds into two main groups. This method of classifying compounds is based on the source of the compound in nature: living or nonliving substances. Scientists have discovered that all living substances contain carbon. Most nonliving substances do not.

-chemists group compounds into two main categories: • Organic compounds contain chains or rings of carbon - most organic compounds contain hydrogen & oxygen - energy in your diet are organic compounds examples: carbohydrates, fats, & proteins

Inorganic compounds either contain no carbon or have single carbon atoms. examples: table salt, water, & minerals

Mixtures are substances that are put together but not chemically combined. Categories: 1. Homogeneous mixture has a uniform distribution of particles throughout the sample visually cannot tell one part of the mixture from another examples: tea, mayonnaise & soft drinks

-A solution is a homogeneous mixture of one material dissolved in another. The material that is dissolved is called the solute. The material that does the dissolving is the solvent • Ex: in sweetened drinks, the water is the solvent and sugar is the solute.

2. Heterogeneous mixture has a non-uniform distribution of particles. examples: vegetable soup -the substances in heterogeneous mixtures can be separated by mechanical means. (strain vegetable soup, & hand sort vegetables)

*sometimes, homogeneous and heterogeneous mixtures can be hard to tell apart. Hot cocoa appears to be a homogeneous mixture, when a cup of cocoa sits without stirring, the heavier cocoa molecules will gradually settle to the bottom. Create a chart showing the classification of matter.

Physical and Chemical Changes You will observe two basic kinds of changes when looking at foods or chemicals. These are physical changes and chemical changes.

Physical Changes involve changing shape, physical state, size, or temperature without changing the chemical identity. Examples: freeze water to ice, still have H 2 O

A phase change is a physical change in the visible structure of matter without changing the molecular structure. In the solid state, atoms and molecules are close together in a rigid structure. Solids have a definite shape & volume. Examples: salt & ice

As solids lose their structure (through heating) they become liquid. -liquids take on the shape of the container Examples: water, milk & fruit juices

Chemical Change occurs whenever new substances with different chemical and physical properties are formed. Chemical changes can produce changes in color, odor, flavor, or the release of gas. Examples: vinegar & baking soda will foam & fizzle -fermented grape juice to make wine

There are times when it will be difficult to tell physical and chemical changes apart. Physical and chemical changes are similar in that they may or may not be reversible.
- Slides: 24