Classifying Matter HOW CAN MATTER BE CLASSIFIED Matter

Classifying Matter

HOW CAN MATTER BE CLASSIFIED? • Matter can be classified by…. …its state solid liquid gas

HOW CAN MATTER BE CLASSIFIED? • Matter can be classified by…. …its state …its color

HOW CAN MATTER BE CLASSIFIED? • Matter can be classified by…. …its state …its color …its uses

HOW CAN MATTER BE CLASSIFIED? • Matter can be classified by…. …its state …its color …its uses …and any of its other properties • BUT we will be classifying matter according to its _______. composition and _____ distribution of particles
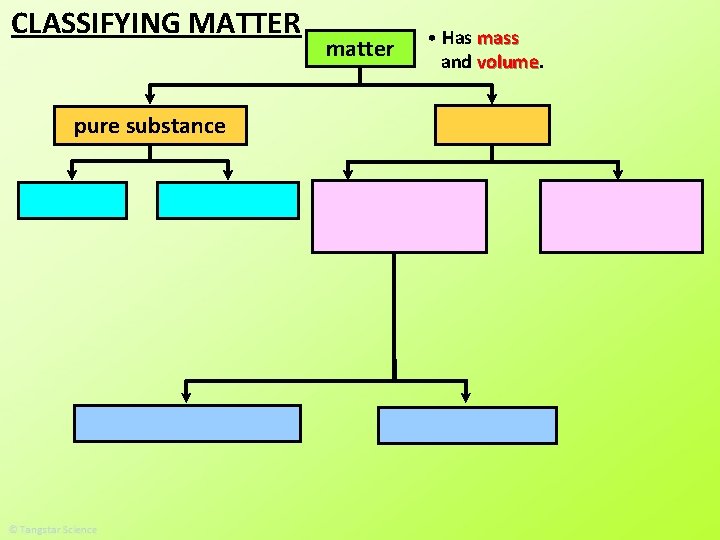
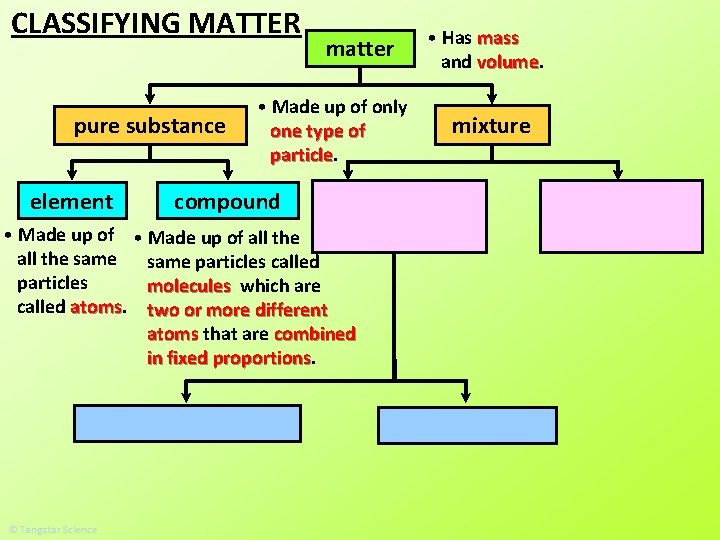
CLASSIFYING MATTER pure substance matter • Has mass and volume

pure substance type of particles. • A pure substance is made up of all the same _____ This is one particle. The rest are all the same particles.
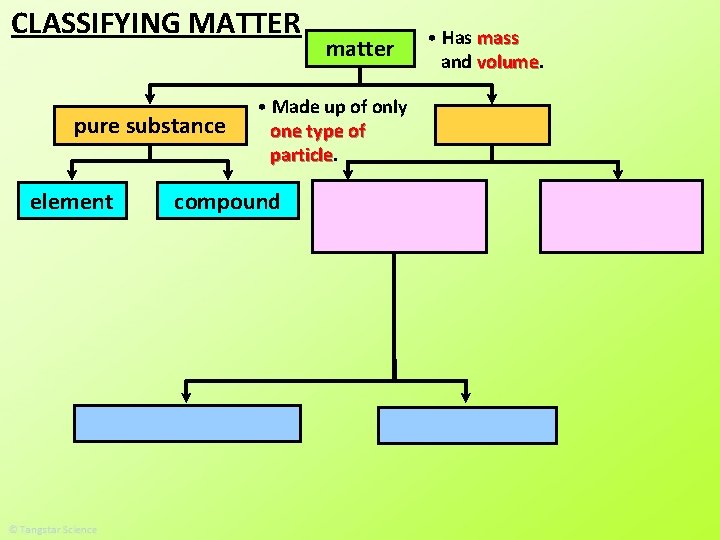
CLASSIFYING MATTER pure substance element matter • Made up of only one type of particle compound • Has mass and volume
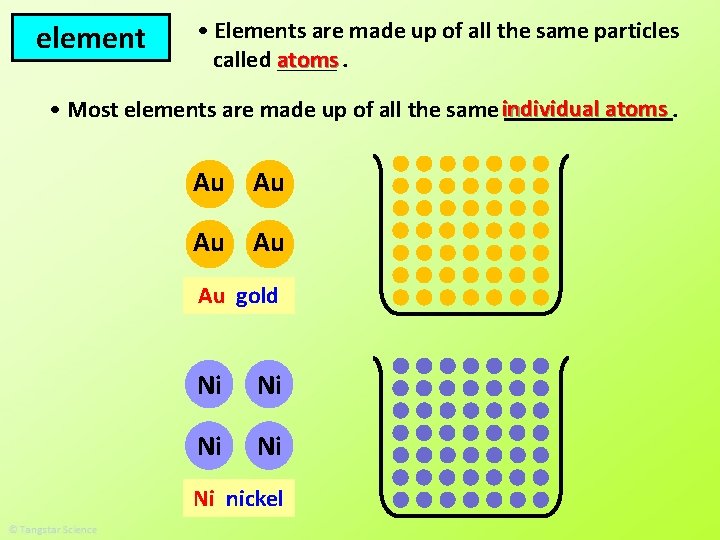
element • Elements are made up of all the same particles called atoms _____. atoms • Most elements are made up of all the same individual _______. Au Au Au gold Ni Ni Ni nickel
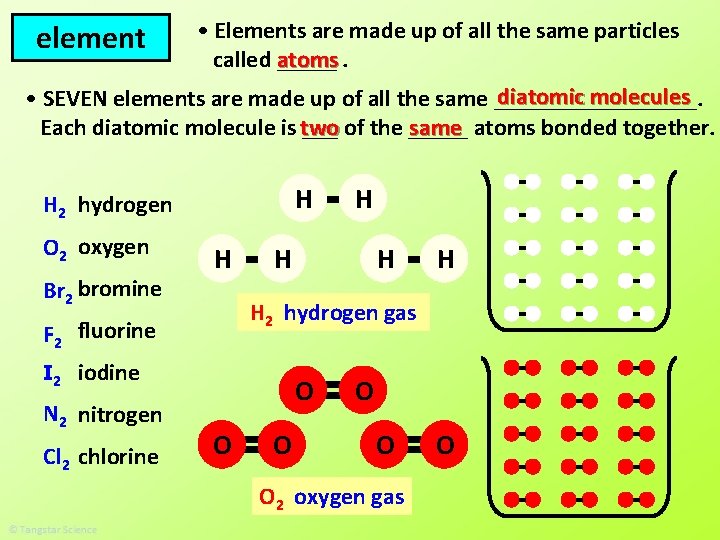
• Elements are made up of all the same particles called atoms _____. diatomic molecules • SEVEN elements are made up of all the same _________. same atoms bonded together. Each diatomic molecule is two ___ of the _____ element H H 2 hydrogen O 2 oxygen Br 2 bromine H H iodine N 2 nitrogen Cl 2 chlorine H H H 2 hydrogen gas F 2 fluorine I 2 H O O O 2 oxygen gas O
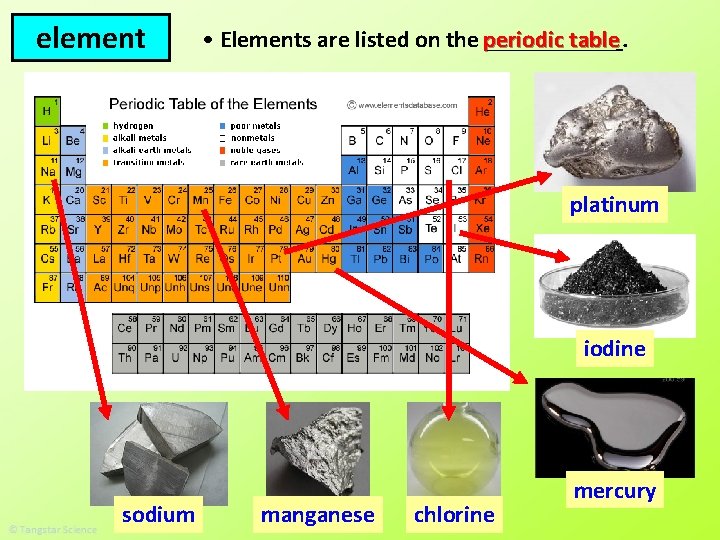
element table. • Elements are listed on the periodic ______ platinum iodine sodium manganese chlorine mercury
CLASSIFYING MATTER pure substance element • Made up of all the same particles called atoms matter • Made up of only one type of particle compound • Has mass and volume
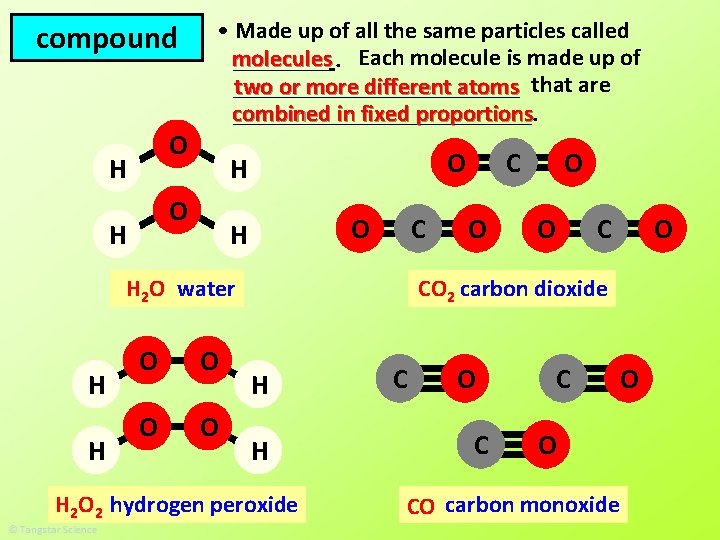
compound O H • Made up of all the same particles called molecules ____. Each molecule is made up of ____________ two or more different atoms that are combined in fixed proportions _____________. O H O H C H 2 O water H H O O O C CO 2 carbon dioxide H H H 2 O 2 hydrogen peroxide C O CO carbon monoxide O
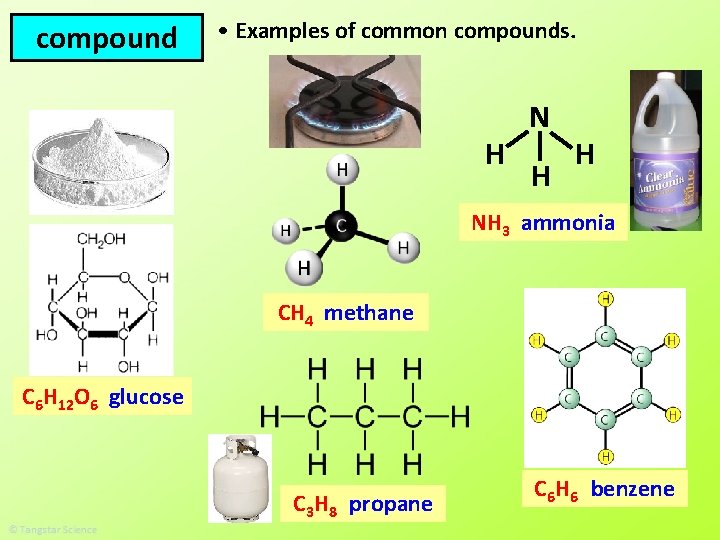
compound • Examples of common compounds. H N H H NH 3 ammonia CH 4 methane C 6 H 12 O 6 glucose C 3 H 8 propane C 6 H 6 benzene
CLASSIFYING MATTER pure substance element matter • Made up of only one type of particle compound • Made up of all the same particles called particles molecules which are called atoms two or more different atoms that are combined in fixed proportions • Has mass and volume mixture
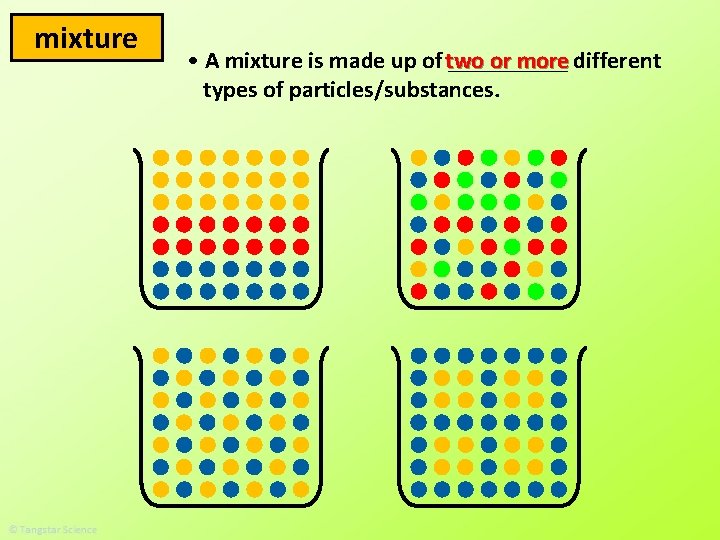
mixture or more different • A mixture is made up of two _____ types of particles/substances.
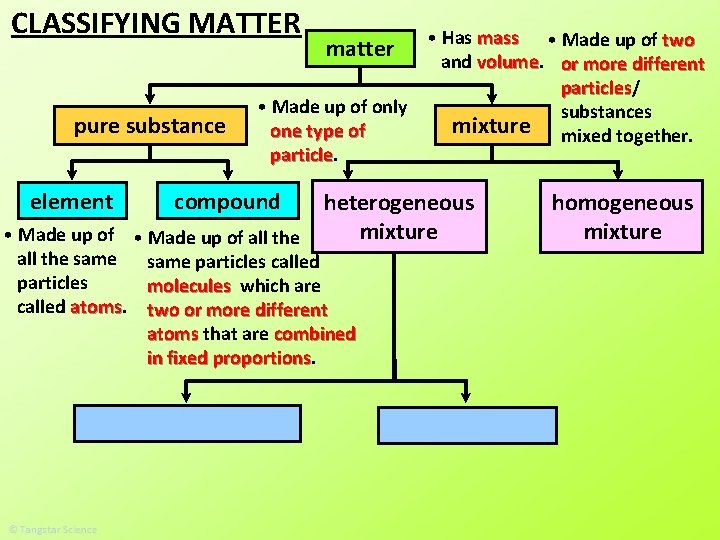
CLASSIFYING MATTER pure substance element • Has mass • Made up of two and volume or more different particles/ particles • Made up of only substances mixture mixed together. one type of particle matter compound heterogeneous mixture • Made up of all the same particles called particles molecules which are called atoms two or more different atoms that are combined in fixed proportions homogeneous mixture
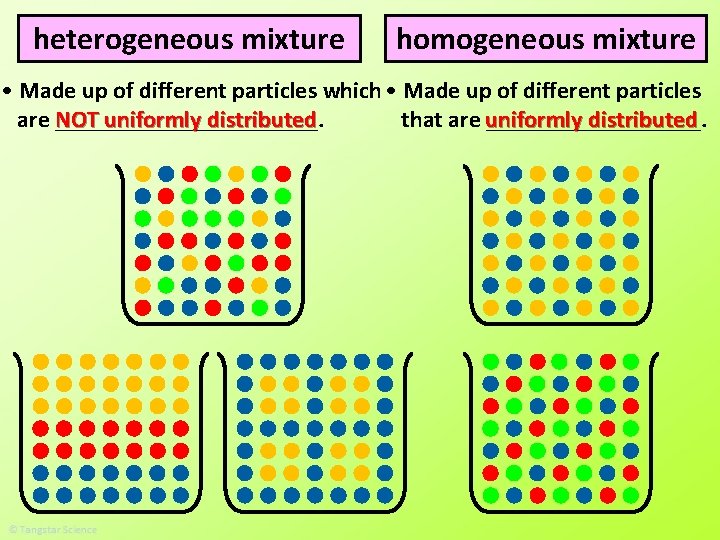
heterogeneous mixture homogeneous mixture • Made up of different particles which • Made up of different particles distributed are NOT ___________. that are uniformly _________. uniformly distributed
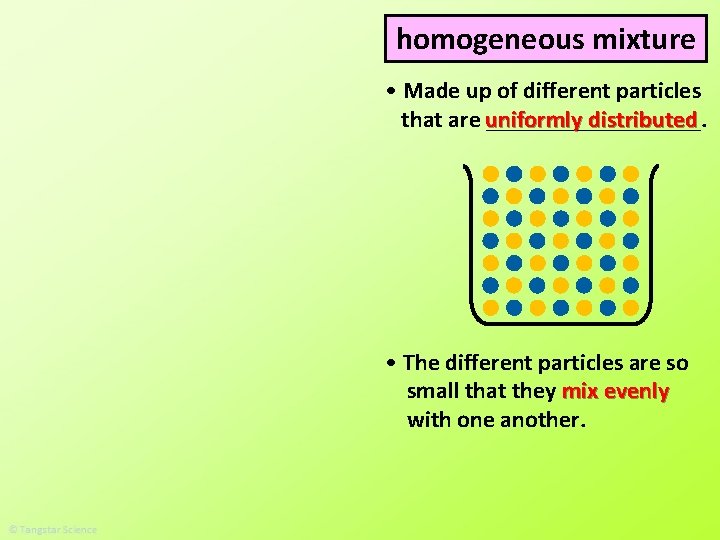
homogeneous mixture • Made up of different particles distributed that are uniformly _________. • The different particles are so small that they mix evenly with one another.
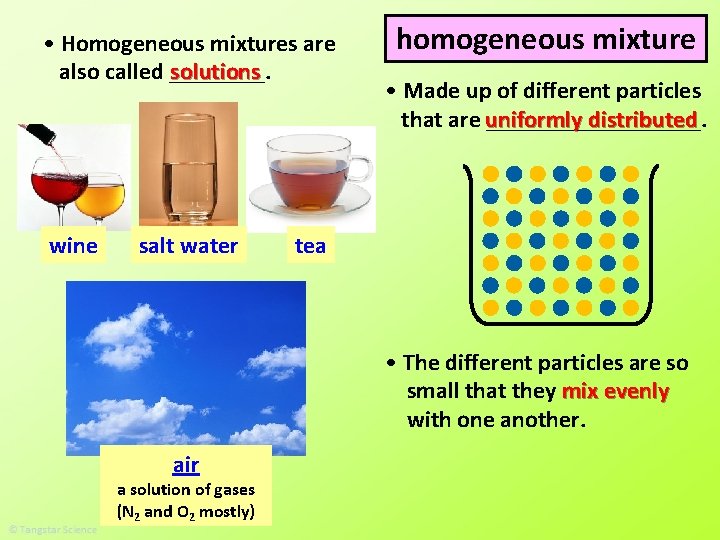
• Homogeneous mixtures are solutions also called ____. wine salt water homogeneous mixture • Made up of different particles distributed that are uniformly _________. tea • The different particles are so small that they mix evenly with one another. air a solution of gases (N 2 and O 2 mostly)
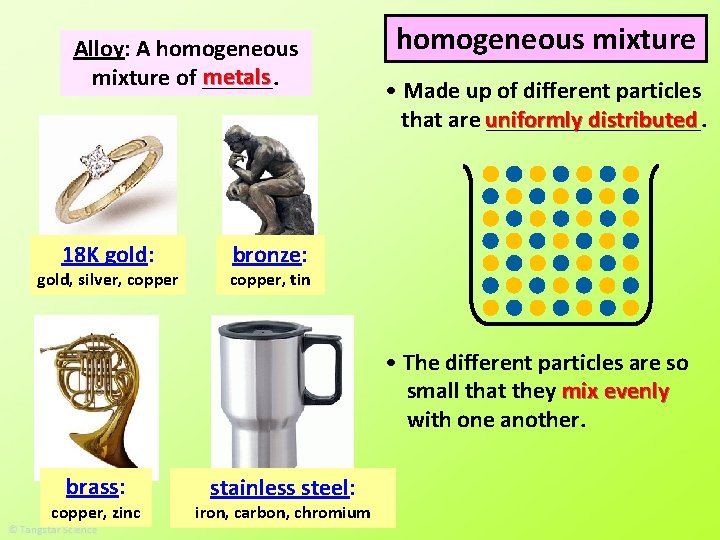
Alloy: A homogeneous metals mixture of ______. 18 K gold: gold, silver, copper homogeneous mixture • Made up of different particles distributed that are uniformly _________. bronze: copper, tin • The different particles are so small that they mix evenly with one another. brass: copper, zinc stainless steel: iron, carbon, chromium
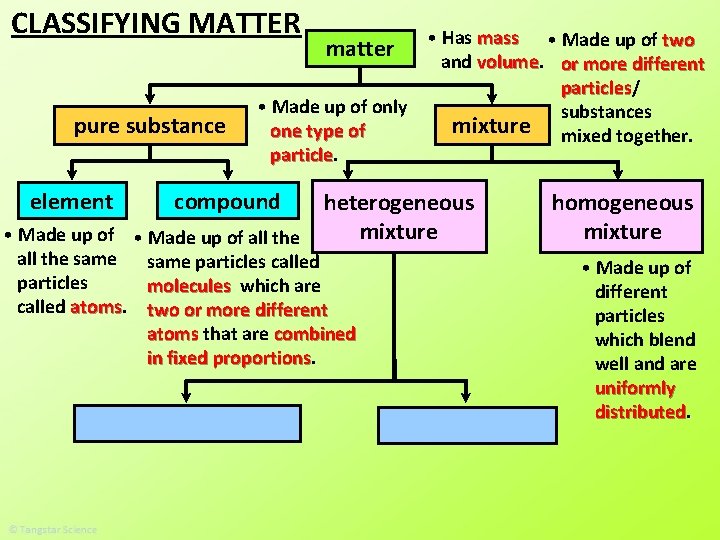
CLASSIFYING MATTER pure substance element • Has mass • Made up of two and volume or more different particles/ particles • Made up of only substances mixture mixed together. one type of particle matter compound heterogeneous mixture • Made up of all the same particles called particles molecules which are called atoms two or more different atoms that are combined in fixed proportions homogeneous mixture • Made up of different particles which blend well and are uniformly distributed.

heterogeneous mixture • Made up of different particles which are NOT ___________. uniformly distributed • The different particles are so large that they do NOT evenly mix with one another (e. g. salt and pepper mixture) or they are chemically unable to mix with one another (e. g. oil and water). However, some appear as if they are evenly mixed even when they are not (e. g. paint).
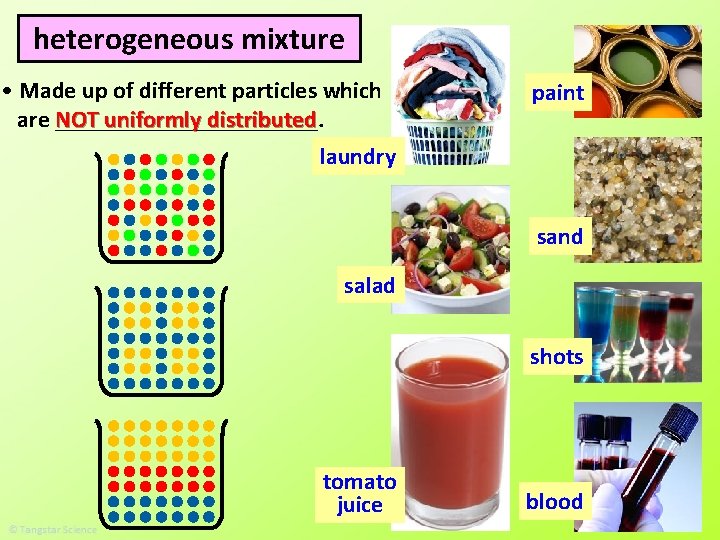
heterogeneous mixture • Made up of different particles which are NOT ___________. uniformly distributed laundry paint sand salad shots tomato juice blood

CLASSIFYING MATTER pure substance element • Has mass • Made up of two and volume or more different particles/ particles • Made up of only substances mixture mixed together. one type of particle matter compound heterogeneous mixture • Made up of all the same particles called particles molecules which are called atoms two or more different atoms that are combined in fixed proportions mechanical mixture homogeneous mixture • Made up of different particles different which don't blend particles which blend are NOT uniformly distributed. well and are uniformly distributed. suspension

mechanical mixture suspension fairly • The different types of particles • The different particles blend _____ well and __________ don’t blend well and separate _______ appear homogeneous _______ into __________. distinctly visible phases out into ____ but will settle ____ different ______ phases over time. Are these mechanical mixtures or suspensions?

mechanical mixture suspension fairly • The different types of particles • The different particles blend _____ well and __________ don’t blend well and separate _______ appear homogeneous _______ into __________. distinctly visible phases out into ____ but will settle ____ different ______ phases over time. tomato juice mud salad shots sand paint laundry flour and water blood oil and vinegar

suspension fairly • The different particles blend _____ well and __________ appear homogeneous ____ out into ____ but will settle ____ different ______ phases over time. • Since particles in a suspension • However, since they contain particles that are larger _____ than those are small they will initially ______ mix in a solution _______, they ______ do not stay together and _______. appear uniform evenly mixed over time. suspension after being shaken suspension after settling
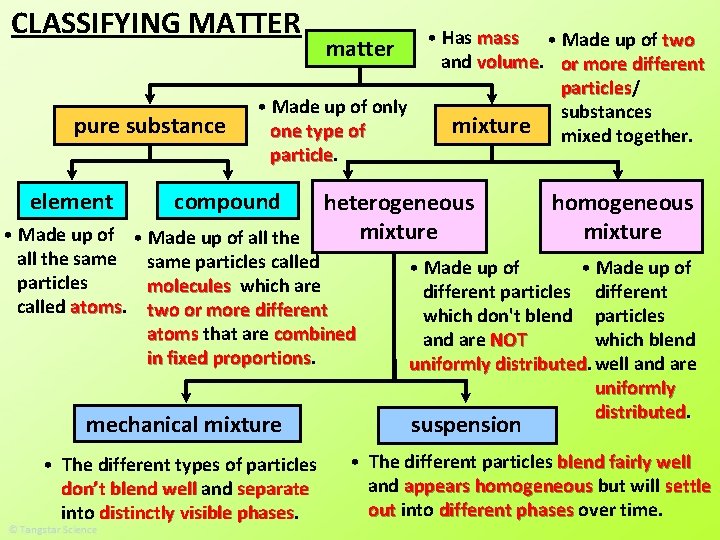
CLASSIFYING MATTER pure substance element • Has mass • Made up of two and volume or more different particles/ particles • Made up of only substances mixture mixed together. one type of particle matter compound heterogeneous mixture • Made up of all the same particles called particles molecules which are called atoms two or more different atoms that are combined in fixed proportions mechanical mixture • The different types of particles don’t blend well and separate into distinctly visible phases homogeneous mixture • Made up of different particles different which don't blend particles which blend are NOT uniformly distributed. well and are uniformly distributed. suspension • The different particles blend fairly well and appears homogeneous but will settle out into different phases over time.

The End!
- Slides: 30