Chemistry Matter and Change Chapter 7 Ionic Compounds











![Group 1 Li: 1 s 22 s 1 or [He] 2 s 1 The Group 1 Li: 1 s 22 s 1 or [He] 2 s 1 The](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-13.jpg)
![Group 1 Na: 1 s 22 p 63 s 1 or [Ne]3 s 1 Group 1 Na: 1 s 22 p 63 s 1 or [Ne]3 s 1](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-14.jpg)

![Group 2 Be: 1 s 22 s 12 or [He] 2 s 2 The Group 2 Be: 1 s 22 s 12 or [He] 2 s 2 The](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-16.jpg)
![Group 2 Mg: 1 s 22 p 63 s 2 or [Ne]3 s 2 Group 2 Mg: 1 s 22 p 63 s 2 or [Ne]3 s 2](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-17.jpg)

![Noble Gas Electron Configuration Ca: [Ar]4 s 2 Ion Symbol Ca 2+ Noble Gas Electron Configuration Ca: [Ar]4 s 2 Ion Symbol Ca 2+](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-19.jpg)
![Group 3 B: 1 s 22 s 2 sp 1 or [He] 2 s Group 3 B: 1 s 22 s 2 sp 1 or [He] 2 s](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-20.jpg)



















- Slides: 44

Chemistry Matter and Change Chapter 7 Ionic Compounds and Metals

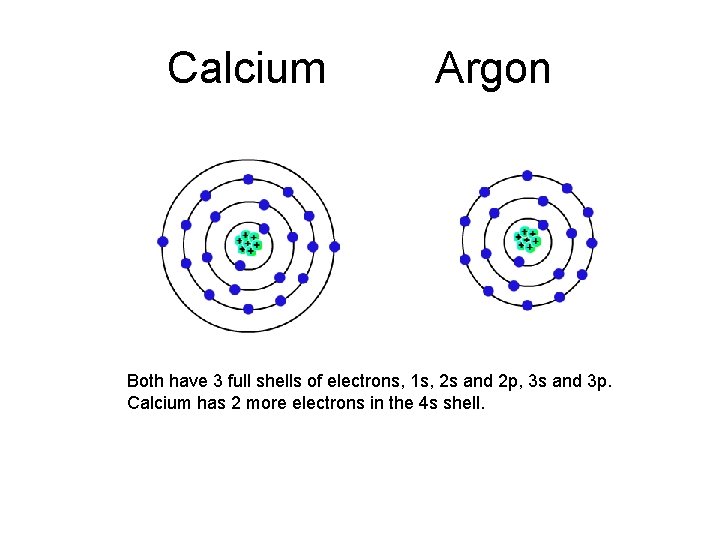
Previous Ca: 1 s 22 p 63 s 23 p 64 s 2 Ar: 1 s 22 p 63 s 23 p 6 Draw the valence shell diagrams. Ca is what element? Ar is what element? Compare Ca to Ar:

Calcium Argon Both have 3 full shells of electrons, 1 s, 2 s and 2 p, 3 s and 3 p. Calcium has 2 more electrons in the 4 s shell.

Previous Ca: 1 s 22 p 63 s 23 p 64 s 2 Ar: 1 s 22 p 63 s 23 p 6 Cl: 1 s 22 p 63 s 23 p 5 What element is Cl? Draw its valence shell diagram. Compare Cl to Ar:

Previous Ca: 1 s 22 p 63 s 23 p 64 s 2 Ar: 1 s 22 p 63 s 23 p 6 Cl: 1 s 22 p 63 s 23 p 5 What will it take for calcium and chlorine to be like argon? Why would calcium and chlorine want to be like argon?

Previous K: 1 s 22 p 63 s 23 p 64 s 1 Ar: 1 s 22 p 63 s 23 p 6 Draw the valence shell diagrams. What element is K? Compare and contrast: Why is argon stable? How can K become “Noble Gas” like?

Previous Ca: 1 s 22 p 63 s 23 p 64 s 2 Ga: 1 s 22 p 63 s 23 p 64 s 23 d 104 p 1 What element is Ga? Draw its valence shell diagram. Compare and contrast:

Important “Noble Gas Like” means to have a full octet of valence electrons in the s and p orbitals. The atoms do not become Noble Gases. Only the atomic number determines the name of the element. The only way to change to another element is nuclear decay, remember the proton and neutrons are in the nucleus not the electrons.

7. 1 Ion Formation How do so many compounds form from the relatively few elements know to exist?

Chemical Bond A chemical bond is the force that holds two atoms together. One way is an attraction between the electron (-) on one atom and the nucleus of another (+). Another way is the attraction between ions.

Valence Electrons in the s and p orbitals only Gp 1 2 13 14 15 16 17 18

Cation Positively charged atom Lose 1 or more electrons Groups 1, 2 and 13
![Group 1 Li 1 s 22 s 1 or He 2 s 1 The Group 1 Li: 1 s 22 s 1 or [He] 2 s 1 The](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-13.jpg)
Group 1 Li: 1 s 22 s 1 or [He] 2 s 1 The outermost shell has 1 electron. Remove 1 electron and lithium is like the Noble Gas helium, called lithium cation 1+ Li
![Group 1 Na 1 s 22 p 63 s 1 or Ne3 s 1 Group 1 Na: 1 s 22 p 63 s 1 or [Ne]3 s 1](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-14.jpg)
Group 1 Na: 1 s 22 p 63 s 1 or [Ne]3 s 1 The outermost shell has 1 electron. Remove 1 electron and sodium is like the Noble Gas neon, called sodium cation 1+ Na

Group 1 K: 1 s 22 p 63 s 23 p 64 s 1 or [Ar]4 s 1 The outermost shell has 1 electron. Remove 1 electron and potassium is like the Noble Gas argon, called potassium cation 1+ K
![Group 2 Be 1 s 22 s 12 or He 2 s 2 The Group 2 Be: 1 s 22 s 12 or [He] 2 s 2 The](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-16.jpg)
Group 2 Be: 1 s 22 s 12 or [He] 2 s 2 The outermost shell has 2 electrons. Remove 2 electrons and beryllium is like the Noble Gas helium 2+ Be
![Group 2 Mg 1 s 22 p 63 s 2 or Ne3 s 2 Group 2 Mg: 1 s 22 p 63 s 2 or [Ne]3 s 2](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-17.jpg)
Group 2 Mg: 1 s 22 p 63 s 2 or [Ne]3 s 2 The outermost shell has 2 electrons. Remove 2 electrons and magnesium is like the Noble Gas neon 2+ Mg

Group 2 Write the Noble Gas electron configuration for Ca Write the ion symbol.
![Noble Gas Electron Configuration Ca Ar4 s 2 Ion Symbol Ca 2 Noble Gas Electron Configuration Ca: [Ar]4 s 2 Ion Symbol Ca 2+](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-19.jpg)
Noble Gas Electron Configuration Ca: [Ar]4 s 2 Ion Symbol Ca 2+
![Group 3 B 1 s 22 s 2 sp 1 or He 2 s Group 3 B: 1 s 22 s 2 sp 1 or [He] 2 s](https://slidetodoc.com/presentation_image_h/98fd8312cc6108adcd49645a43b548b3/image-20.jpg)
Group 3 B: 1 s 22 s 2 sp 1 or [He] 2 s 22 p 1 The outermost shell has 3 electrons. Remove 3 electrons and boron is like the Noble Gas helium 3+ B

Group 3 Write the Noble Gas electron configurations for: Al Ga Write the ion symbols.

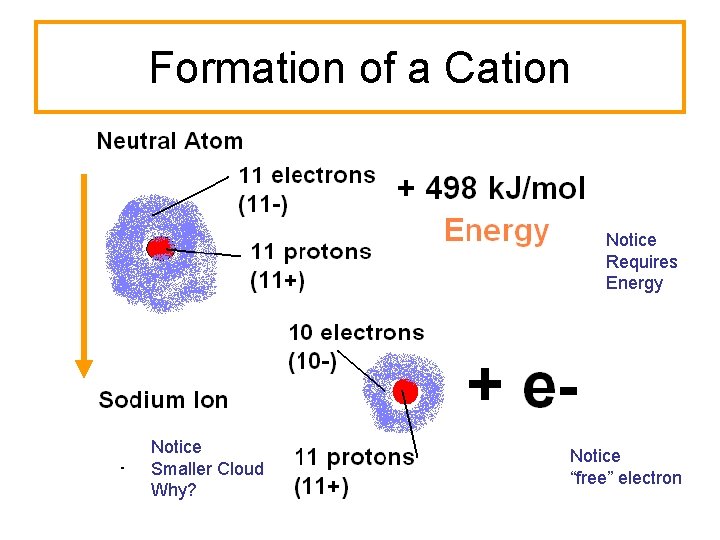
Cations Does removal of an electron from a neutral atom require or release energy?

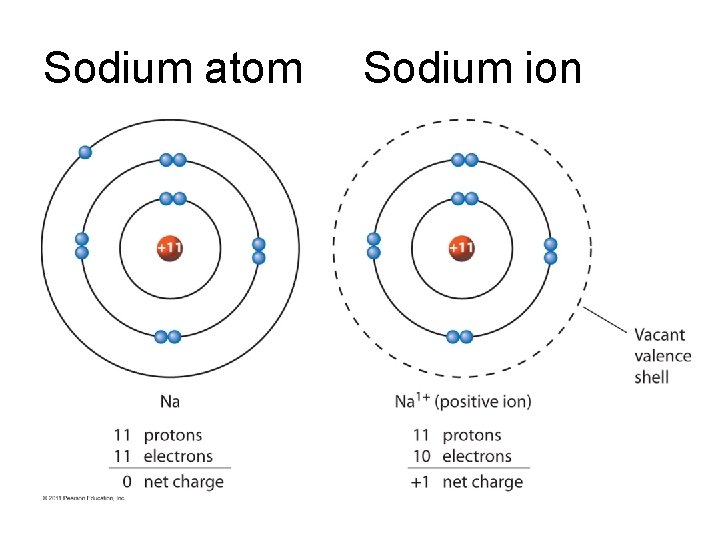
Sodium atom Sodium ion

Formation of a Cation Notice Requires Energy Notice Smaller Cloud Why? Notice “free” electron

Metals • Group 1 and 2 most reactive • Form cations easy

Transition Metals Have full s 2 orbitals Lose these 2 electrons and maybe one electron in the d orbital Sometimes it is difficult to predict how many Important to write cation symbol Fe 2+ and Fe 3+

Anion Negatively Charged Ion Gain one or more electrons Name is changed: __suffex becomes __ide chlorine chloride nitrogen nitride

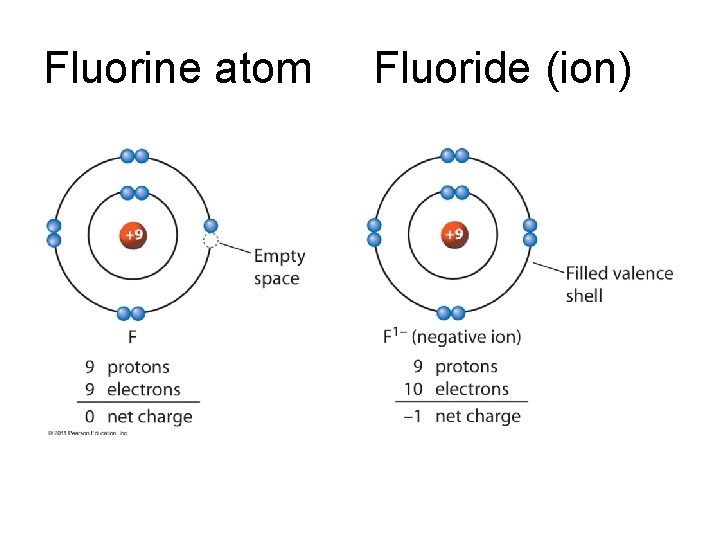
Group 17 Flourine wants to be neon like. Has 7 valence electrons and wants 8 Gains 1 electron and has a -1 charge Flouride F

Group 17 Chlorine wants to be argon like. Has 7 valence electrons and wants 8 Gains 1 electron and has a -1 charge Chloride Cl

Group 17 Bromine wants to be krypton like. Has 7 valence electrons and wants 8 Gains 1 electron and has a -1 charge Bromide Br -

Group 16 Oxygen wants to be neon like Has 6 valence electrons and wants 8 Gains 2 electron and has a -2 charge Oxide 2 O

Group 16 Sulfur wants to be argon like Has 6 valence electrons and wants 8 Gains 2 electron and has a -2 charge Sulfide 2 S

Group 16 Write the Valance Electron Diagram for Selinium, Se What noble gas does it want to be like? How many electrons does it need? Write the name and symbol for the anion.

Valence Electron diagram Wants to be like Krypton, Kr Needs 2 electrons Ion Se 2 -

Group 15 Find the number of valence electrons. What noble gas do these elements want to be like? Draw the electron dot structure for each. Name the anion and write its name. Nitrogen, Phosphorus and Arsenic

Fluorine atom Fluoride (ion)

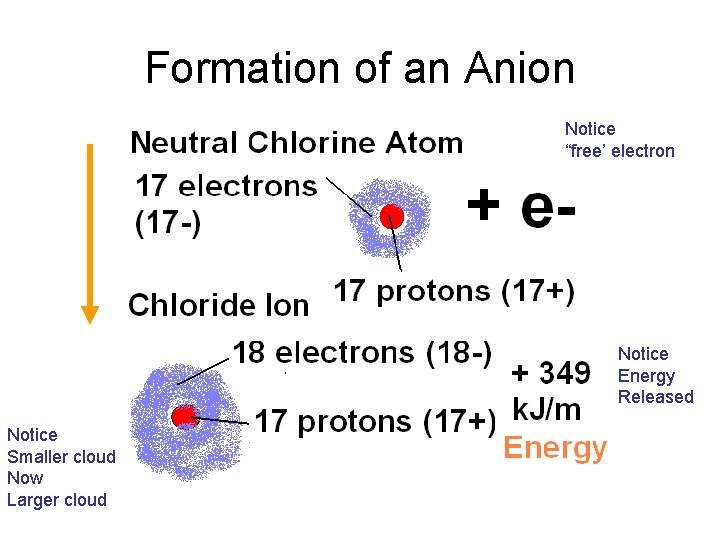
Formation of an Anion Notice “free’ electron Notice Energy Released Notice Smaller cloud Now Larger cloud
Compare How do the energy changes accompanying positive ion and negative ion formation compare?

Questions p. 209 1. Compare the stability of a lithium atom with that of its ion, Li+. 2. Describe two causes of the force of attraction in a chemical bond. 3. Why are all of the elements in group 18 unreactive, whereas those in group 17 are very reactive?

4. Summarize ionic bond formation by correctly paring these terms: cation, anion, electron gain, electron loss. 5. Write out the electron configuration for each atom. Then predict the change that must occur in each to achieve a noblegas configuration. a. nitrogen b. sulfur c. barium d. lithium

6. Draw models to represent the formation of the positive calcium ion and the negative bromide ion.

Fluorine 1. 2. 3. 4. Write out electron configuration Calculate number of valence electrons What ion forms? (symbol) What is the name of the ion?

Each Student Write 5 element symbols on a piece of paper with atomic number 54 or lower. Switch papers Write electron configuration for each element Draw electron dot diagram Determine what type of ion is most likely formed

Exit Ticket Determine the ions formed for Strontium Aluminum Sulfur