Matter Properties and Change What is Matter Matter



















- Slides: 33

Matter: Properties and Change

What is Matter? Matter is anything that takes up space and/or has mass. Matter is made up of atoms and molecules.

What are Atoms? Atoms are the building blocks of matter. They are composed of particles called protons, electrons and neutrons. The smallest particle of matter.

Protons carry a positive electrical charge + Electrons carry a negative electrical charge Neutrons carry no electrical charge at all o – they are NEUTRAL!!! • http: //studyjams. scholastic. com/studyjams/science/

What are Molecules? Molecules are a group of atoms bonded together.

What are Elements? . any material that cannot be broken down into more fundamental substances. Each element has only that specific type of atom. • http: //studyjams. scholastic. com/studyjams/science/matte r/elements-and-compounds. htm


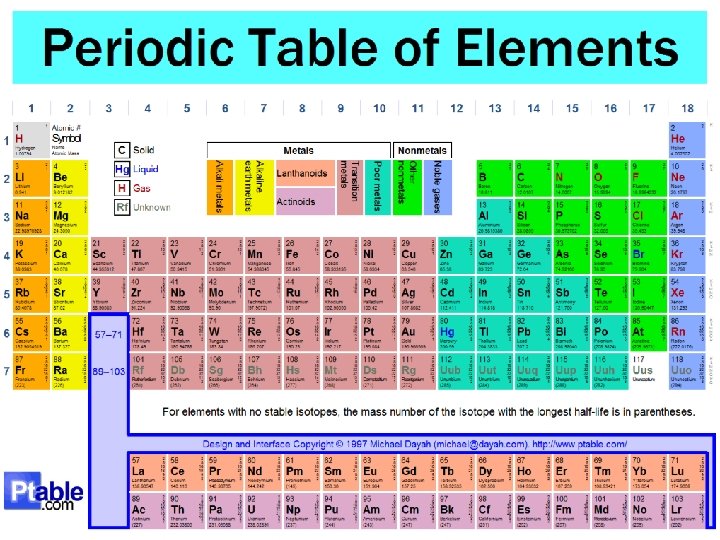
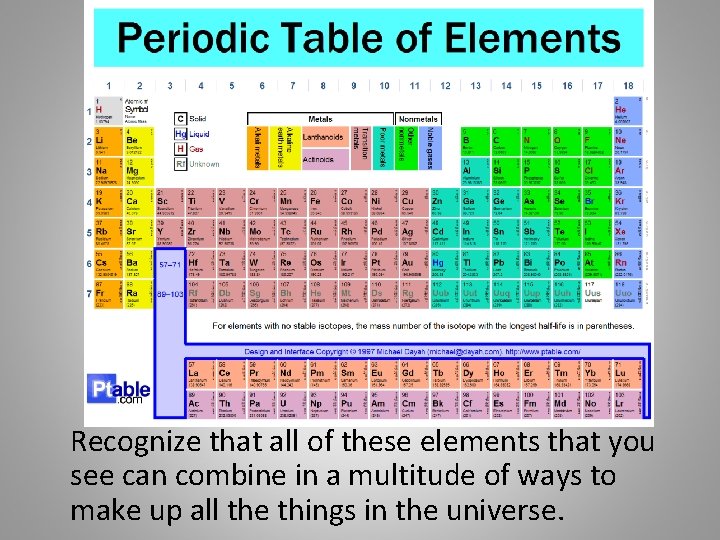
The Periodic Table • The periodic table is a table that lists the element with over 100 elements on it. • The periodic table shows the symbols for the elements, their full names, their atomic numbers and their mass number. • It organizes elements into groups and periods (columns and rows) based on their structure and properties.

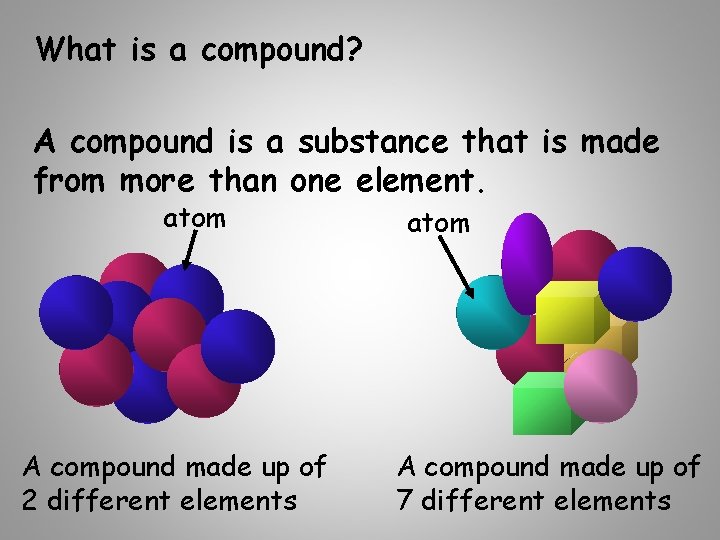
What is a compound? A compound is a substance that is made from more than one element. atom A compound made up of 2 different elements atom A compound made up of 7 different elements

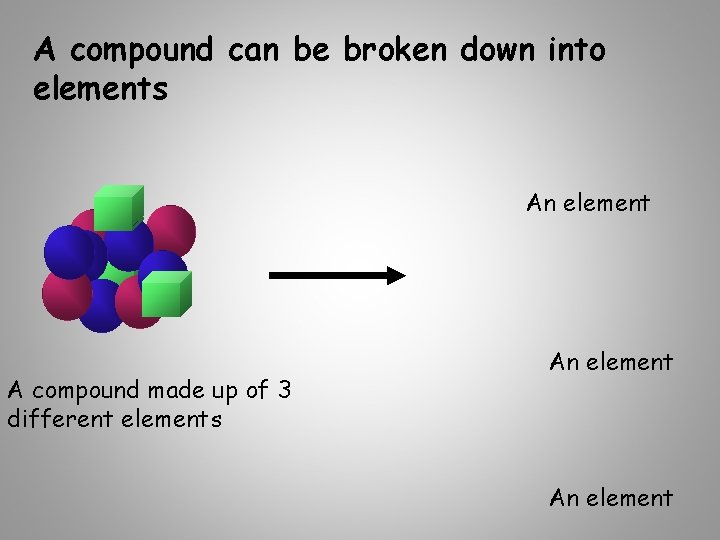
A compound can be broken down into elements An element A compound made up of 3 different elements An element

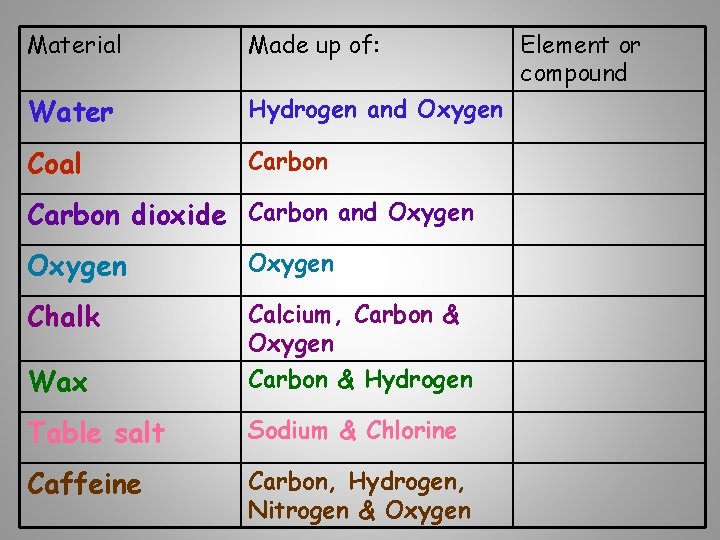
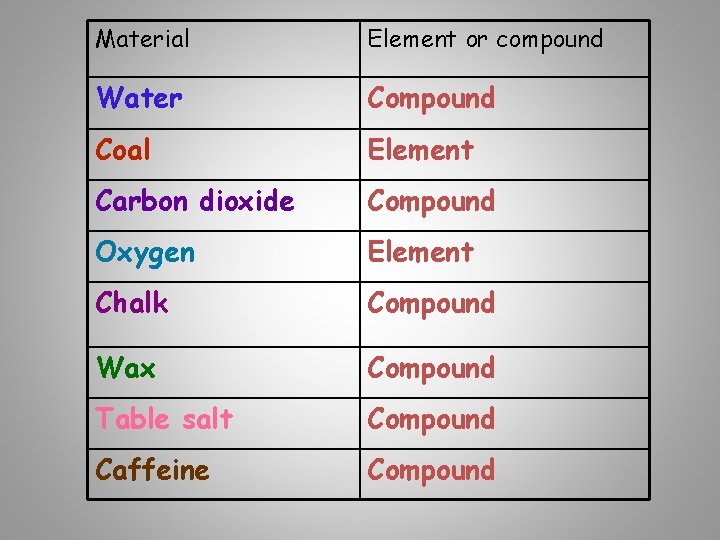
Material Made up of: Water Hydrogen and Oxygen Coal Carbon dioxide Carbon and Oxygen Chalk Wax Calcium, Carbon & Oxygen Carbon & Hydrogen Table salt Sodium & Chlorine Caffeine Carbon, Hydrogen, Nitrogen & Oxygen Element or compound

Material Element or compound Water Compound Coal Element Carbon dioxide Compound Oxygen Element Chalk Compound Wax Compound Table salt Compound Caffeine Compound

Mass is how much matter an object has *Mass is measured in grams (g) or kilograms (kg)

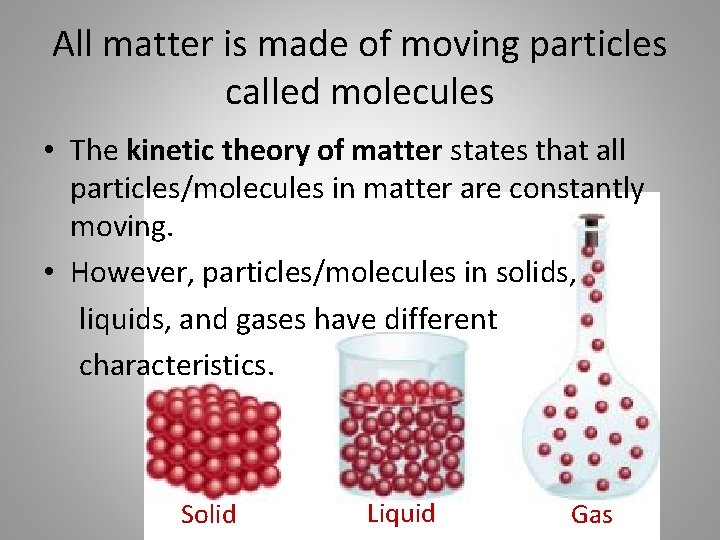
All matter is made of moving particles called molecules • The kinetic theory of matter states that all particles/molecules in matter are constantly moving. • However, particles/molecules in solids, liquids, and gases have different characteristics. Solid Liquid Gas

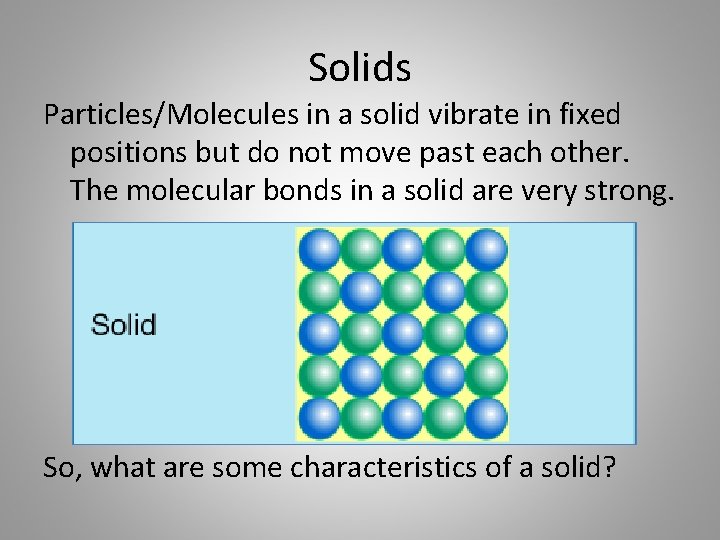
Solids Particles/Molecules in a solid vibrate in fixed positions but do not move past each other. The molecular bonds in a solid are very strong. So, what are some characteristics of a solid?

Liquids Particles/Molecules in a liquid are not tightly bound to each other as it is in a solid, molecules in liquid slide past each other. So, what are the characteristics of liquids?

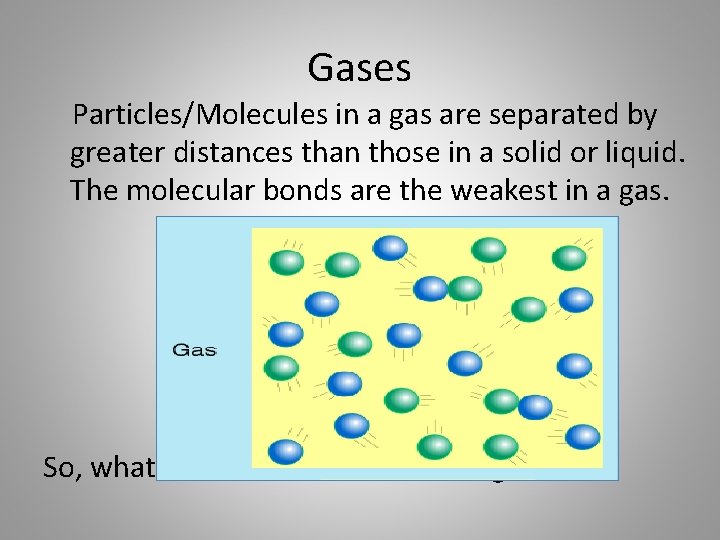
Gases Particles/Molecules in a gas are separated by greater distances than those in a solid or liquid. The molecular bonds are the weakest in a gas. So, what are the characteristics of gas?

FYI: There is a 4 th state of matter called plasma… Plasma is the most commonly seen state of matter in the universe. Plasma is similar to gas but portions are ionized. In an ordinary gas each atom contains an equal number of positive and negative charges; the positive charges in the nucleus are surrounded by an equal number of negatively charged electrons, and each atom is electrically "neutral. " A gas becomes a plasma when the addition of heat or other energy causes a significant number of atoms to release some or all of their electrons. The remaining parts of those atoms are left with a positive charge, and the detached negative electrons are free to move about. Those atoms and the resulting electrically charged gas are said to be "ionized. " When enough atoms are ionized to significantly affect the electrical characteristics of the gas, it is a plasma.

Recognize that all of these elements that you see can combine in a multitude of ways to make up all the things in the universe.

Temperature and Heat Unit B Chapter 2

Particles in a substance are not moving at the same speed and can change speeds.

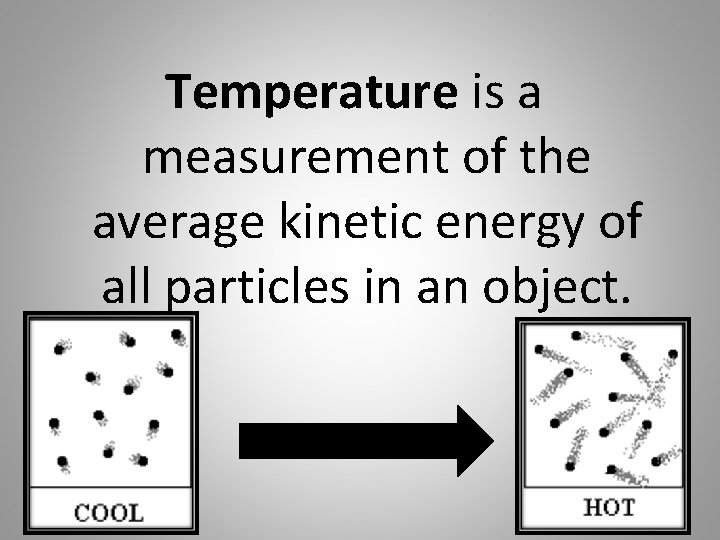
Temperature is a measurement of the average kinetic energy of all particles in an object.

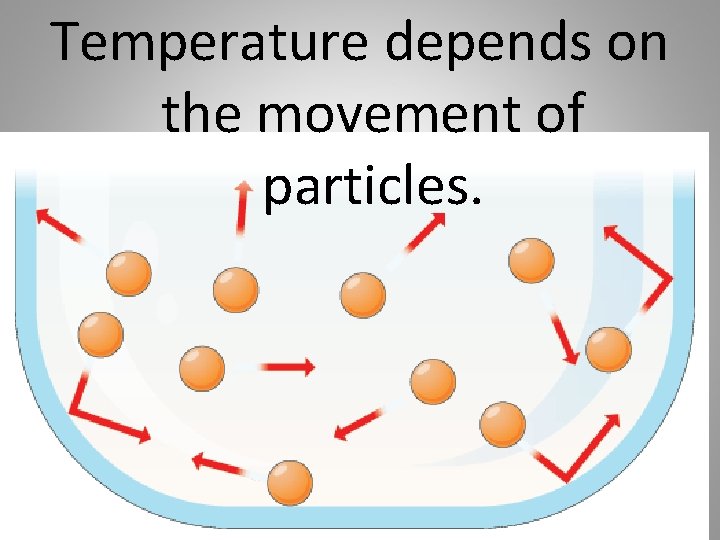
Temperature depends on the movement of particles.

Heat is a flow of energy due to temperature differences. And flows from higher temperatures to lower temperatures.

Now that’s HOT Ha ^ _ ^ ha

Heat and Temperature are closely related but not the same… • Temperature is the average kinetic energy of particles in a substance or location. • Heat is a flow of energy from an object with a higher temperature to an object with a lower temperature.

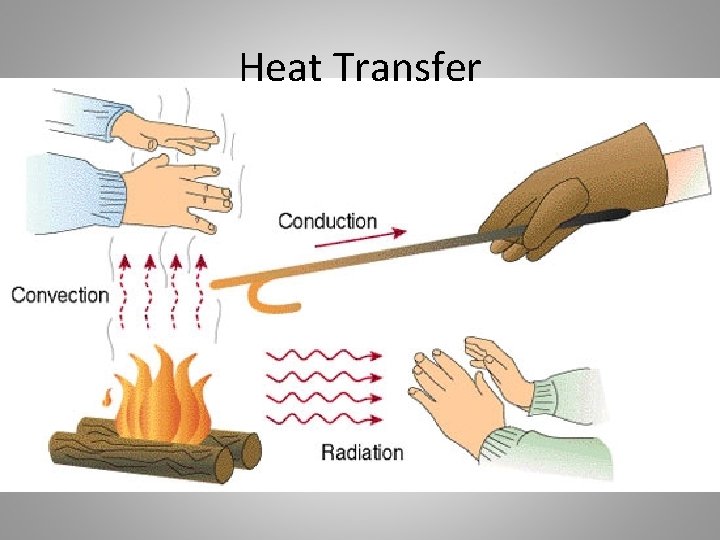
Kinds of Heat transfer • Heat Energy transfers in three ways. • Heat Energy can be transferred by conduction, convection, or radiation. • Heat Energy is transferred between objects or locations when there are temperature differences between them.

Conductors & Insulators • Materials that easily transfer energy are conductors; those that are poor conductors are called insulators. • Think about this question… • Why are pot and pan handles made out of plastic or wood? • Why is the pan made of metal? • Why are coolers made out of types of plastics? Good Conductors Metals Poor Conductors Wood Paper Plastic foam

Heat Transfer

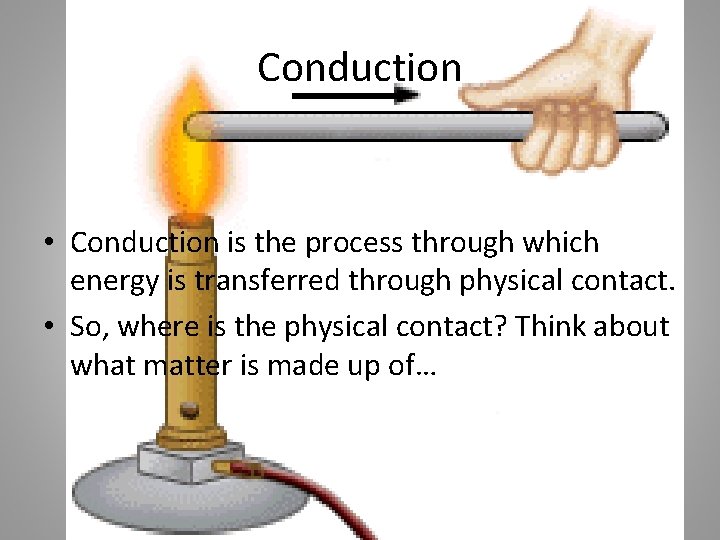
Conduction • Conduction is the process through which energy is transferred through physical contact. • So, where is the physical contact? Think about what matter is made up of…

Convection • Convection is the process that transfers energy in gases and liquids. • Hot air ____ and cold or cooler are _______. • Why do hot liquids and gases rise and cooler liquids and gases sink? Think about density… • When liquids and gases are heated, they become less dense and therefore will rise above any material that is more dense.

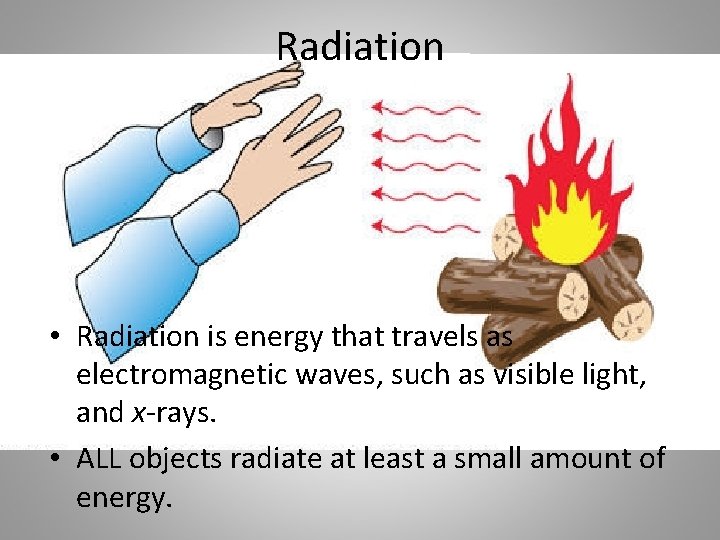
Radiation • Radiation is energy that travels as electromagnetic waves, such as visible light, and x-rays. • ALL objects radiate at least a small amount of energy.

Thermal Expansion - is a result of changes in temperature, matter changes in volume. Solids expand as the particles move ever so slightly farther apart.