Matter and Change Honors Chemistry Chemistry is a











- Slides: 13

Matter and Change Honors Chemistry

Chemistry is a Physical Science • Chemistry is the study of the composition, structure, and properties of matter and the changes it undergoes. • Branches of Chemistry – – – Organic Chemistry Inorganic Chemistry Physical Chemistry Analytical Chemistry Biochemistry Theoretical Chemistry

Matter and Its Properties • A chemical is any substance that has definite composition. • Matter is anything that has mass and volume. – Mass is the amount of matter in an object. – Volume is how much space the object occupies.

Building Blocks of Matter • An atom is the smallest unit of matter that maintains the properties of the element. • An element is a pure substance made of only one kind of atom. • A compound is a substance made of the atoms of two or more elements that are chemically bonded.

Properties of Matter • Extensive properties depend on how much matter is present. – i. e. mass, volume, etc. • Intensive properties do not depend upon the amount of matter. – Boiling point, melting point, density, etc.

Physical Properties and Physical Changes • Physical properties can be observed without changing the identity of the substance. – i. e. color, mass, odor, boiling point, density. • Physical changes do not change the identity of the substance. – i. e. phase changes, solutes dissolving, cutting, tearing, etc.

Chemical Properties and Chemical Changes • Chemical properties can only be observed when a chemical reaction has occurred. • Chemical changes occur when matter undergoes a chemical reaction and new substances are formed.

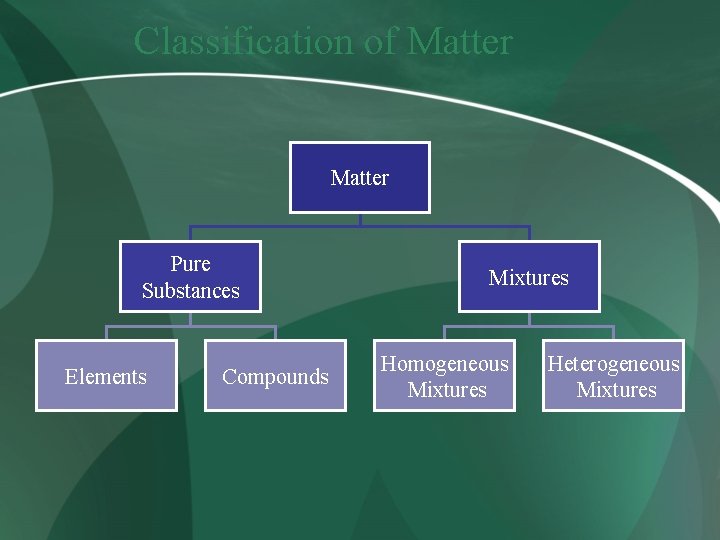
Classification of Matter Pure Substances Elements Compounds Mixtures Homogeneous Mixtures Heterogeneous Mixtures

Mixtures • Homogeneous mixtures include solutions and alloys. • In a homogeneous mixture, the particles are not observable. • Heterogeneous mixtures occur whenever we can see the particles that have been mixed together.

Pure Substances • Elements • Compounds

Introduction to the Periodic Table • The vertical columns are called groups or families. There are 18 of them. • The horizontal rows are called periods, there are 7 of them.

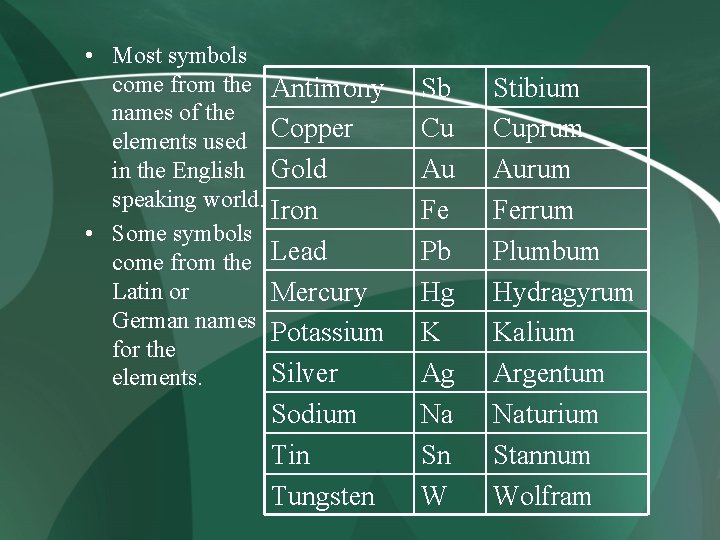
• Most symbols come from the Antimony names of the elements used Copper in the English Gold speaking world. Iron • Some symbols come from the Lead Latin or Mercury German names Potassium for the Silver elements. Sodium Tin Tungsten Sb Cu Au Fe Pb Hg K Ag Na Sn W Stibium Cuprum Aurum Ferrum Plumbum Hydragyrum Kalium Argentum Naturium Stannum Wolfram

Types of Elements • Metals • Non-Metals • Semi-Metals