CHEMISTRY UNIT 4 REVIEW PART 1 Significant Digits


















































- Slides: 52

CHEMISTRY UNIT 4 REVIEW PART 1 Significant Digits and Metric Conversions!

How Many? Conversions Add/Subtract Multiply/Divide Mix It Up! 10 10 10 20 20 20 30 30 30 40 40 40 50 50 50

Question 1 - 10 • How many significant Digits are in this number: 1422

Answer 1 – 10 • 4! • All numbers present are between 1 and 9.

Question 1 - 20 • How many significant digits are in the following number? 200

Answer 1 – 20 • There is only 1! • The zeros don’t count because the number doesn’t have a decimal place in it!

Question 1 - 30 • How many significant ZEROs are in the following number? 0. 0030050

Answer 1 – 30 • 3! • The captures zeros and the trailing zero are the only ones that are significant!

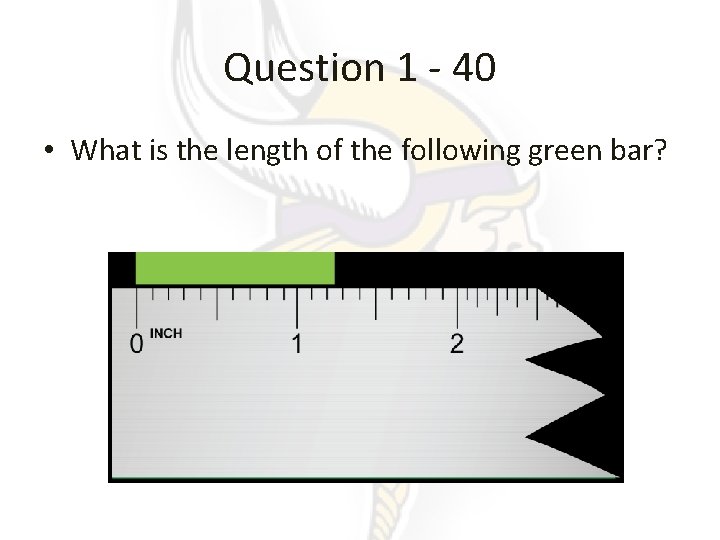
Question 1 - 40 • What is the length of the following green bar?

Answer 1 – 40 • 1. 25! • The 1 and the 2 are in concrete, the 5 can be a different number that is reasonable.

Question 1 - 50 • How many zeros in the following number are NOT significant? . 0005050090

Answer 1 – 50 • 3! • Only the zeros in the beginning of the number do not count!

Question 2 - 10 • Convert the following: 254. 3 grams to Kilograms

Answer 2 – 10 • . 2543 Kg • The number will get smaller because the unit gets bigger!

Question 2 - 20 • How many milliliters are in the following number? 0. 0367 Liters

Answer 2 – 20 • 36. 7 m. L • The number gets bigger because the unit gets smaller!

Question 2 - 30 • Which weighs more? 4. 25 grams of Au or 425 mg of Au

Answer 2 – 30 • 4. 25 grams does! • Convert milligrams to grams and you get. 425 mg! That is less than 4. 25 grams!

Question 2 - 40 • What is the metric prefix that correspond to the following quantity? -2 10

Answer 2 – 40 • Centi!

Question 2 - 50 • How many m. L are in the following number? 87 DL

Answer 2 – 50 • There are 870, 000 m. L! • KHD*dcm

Question 3 - 10 • What is the answer to the following addition problem? 212. 2 + 26. 7 + 402. 09

Answer 3 – 10 • Addition, so look for least # of decimal places! • 641. 0

Question 3 - 20 • What is the answer to the following addition problem? 2 2. 01 x 10 + 3 3. 014 x 10

Answer 3 – 20 • 3215 • You have to take the numbers out of scientific notation to look at decimal places!

Question 3 - 30 • What is the answer to the following problem? 52. 331 + 26. 01 – 0. 9981

Answer 3 – 30 • 77. 34 • Watch number of decimal places here! Add and subtract carry least number of decimal places to the answer.

Question 3 - 40 • How many digits after the decimal point should be reported when the following calculation is performed? 199. 0354 + 43. 09 + 121. 2

Answer 3 – 40 • There should only be 1 decimal place in this answer!

Question 3 - 50 • How many digits after the decimal place should be reported when the following calculation is performed? 10, 434 – 9. 3344

Answer 3 – 50 • There will not be any decimal places in this number!

Question 4 - 10 • What is the answer to the following division problem? 12. 011 / 23 6. 022 x 10

Answer 4 – 10 • 1. 995 x 10 -23 • We need to look at number of significant digits in the numbers of the problem—least number determines answers sig figs!

Question 4 - 20 • What is the answer to the following problem? (4. 031)(0. 082068)(373. 1) /. 995

Answer 4 – 20 • 124 • Remember least # of sig figs will be reported in the answer!

Question 4 - 30 • The answer to the following question should be written with how many significant digits? 2 2. 3733 x 10 / 343

Answer 4 – 30 • 3!

Question 4 - 40 • When the following calculation is performed, the answer should be reported with how many significant digits? . 0043*. 0821*298

Answer 4 – 40 • 2! • Remember least goes!

Question 4 - 50 • What is the answer to the following problem? -4 2 (6. 1982 x 10 )

Answer 4 – 50 • 3. 8418 x 10 -7 • Or 3. 8000 x 10 -7

Question 5 - 10 • What is the answer to the following problem? (2. 0944+. 0003233*12. 22) /7. 001

Answer 5 – 10 • . 2997 • Remember your order of operations! • Please Excuse My Dear Aunt Sally!

Question 5 - 20 • What is the answer to the following problem? 2 3 [(1. 42 x 10 )+(1. 021 x 10 )]/ -1 3. 1 x 10

Answer 5 – 20 • 3. 8 x 103

Question 5 - 30 • Consider the following numbers 23. 68 and 4. 12. • The sum of these numbers has how any significant digits? • The product of these numbers has how many significant digits?

Answer 5 – 30 • Sum has 4 • Product has 3

Question 5 - 40 • You pour the water from these beakers into a separate container. What is the volume with the correct number of significant digits.

Answer 5 – 40 • Your answer should range from 81 to 82 m. L without any decimals!

Question 5 - 50 • Report the volume of the water in each beaker with the proper # of significant digits.

Answer 5 – 50 • I has 26. 4(ish) m. L • II has 28(ish) m. L • III has 26. 42(ish) m. L • The –ish comes from your interpretation. Remember the first digits are known and the last digit is an educated guess (is it uncertain).