Disorder in crystals Disorder in crystals All lattice


















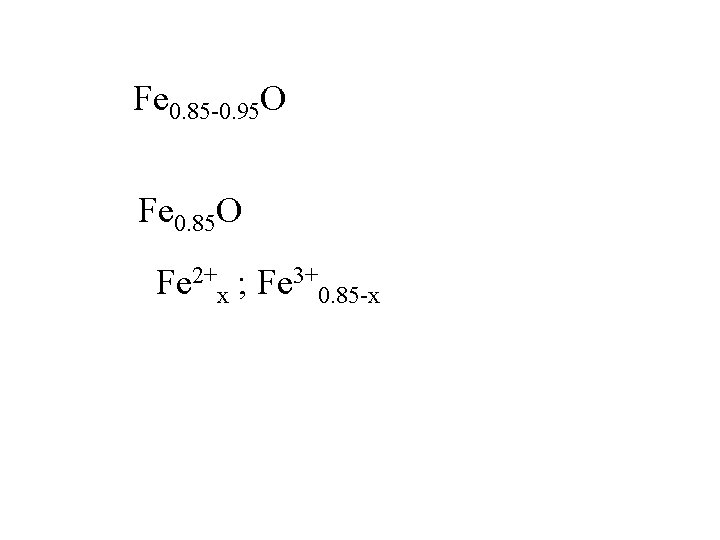

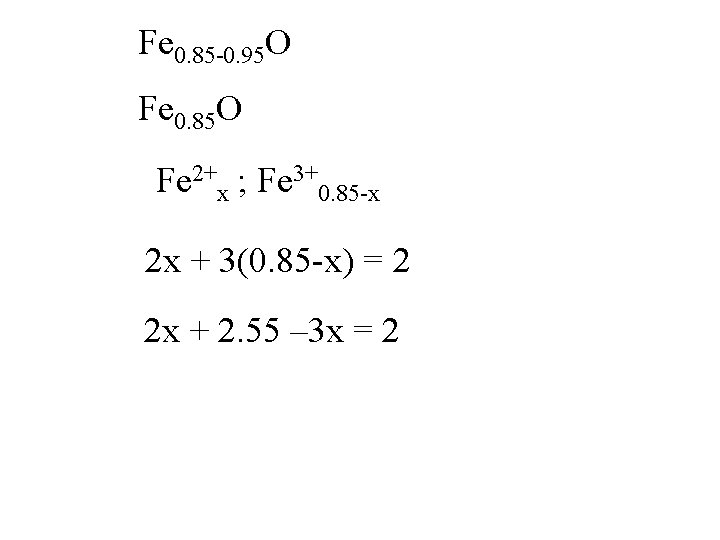
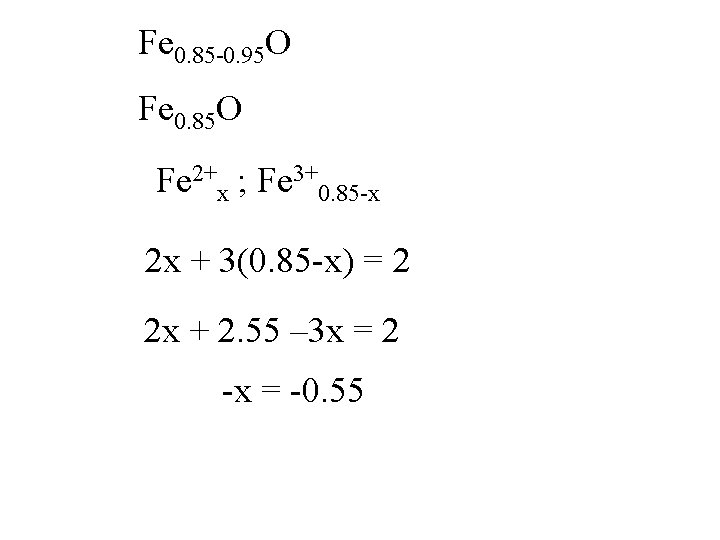
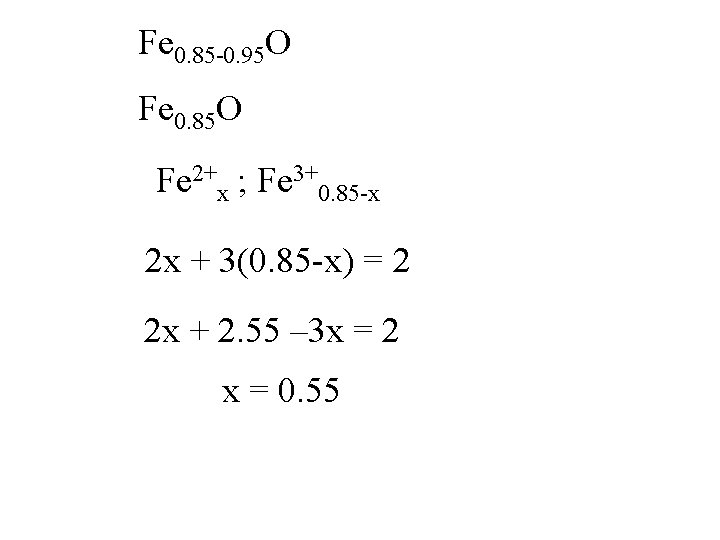
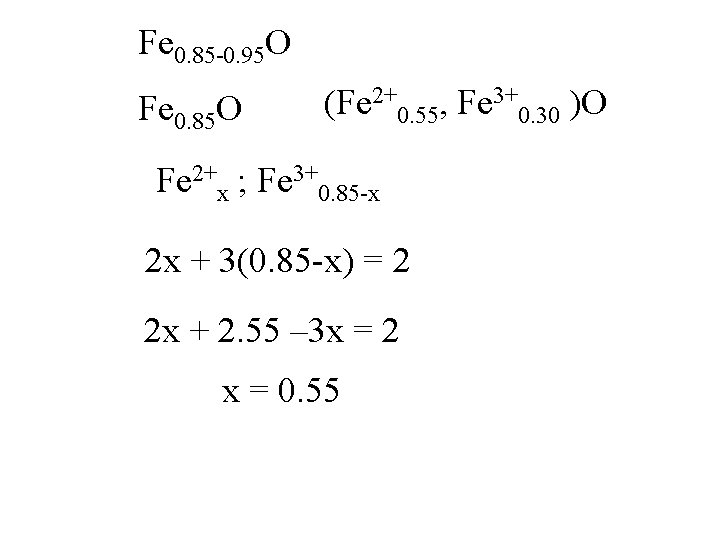











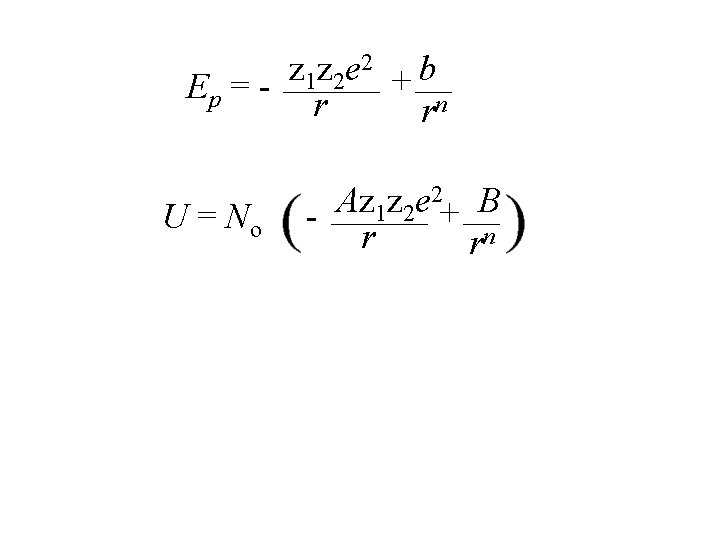





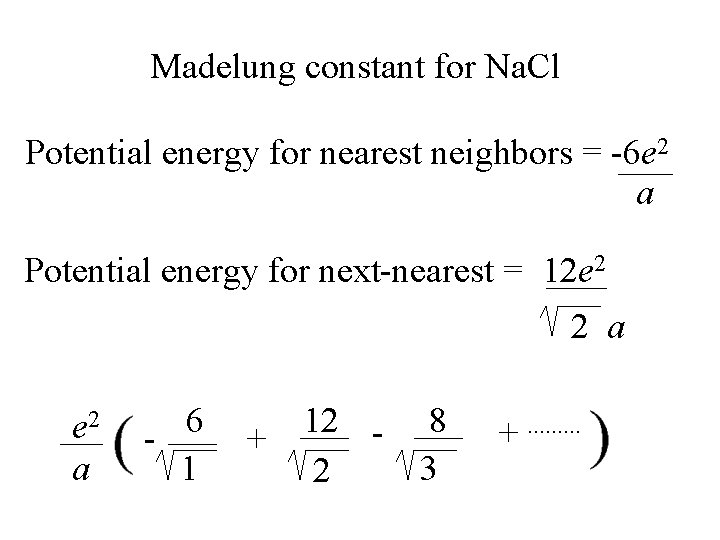
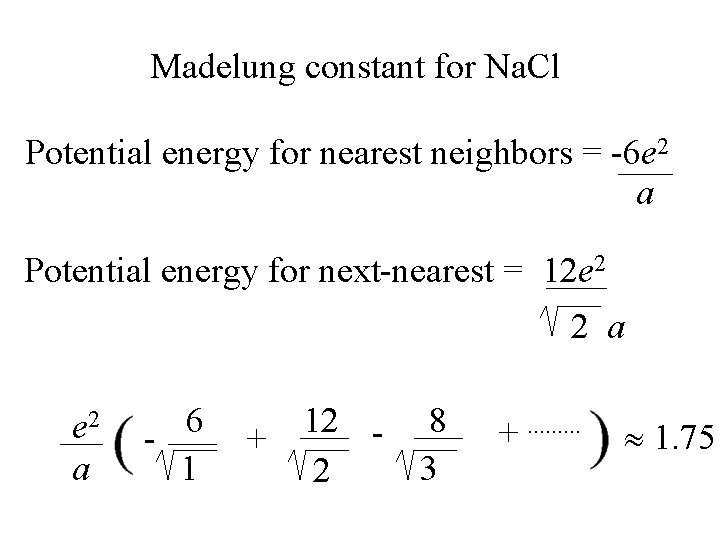
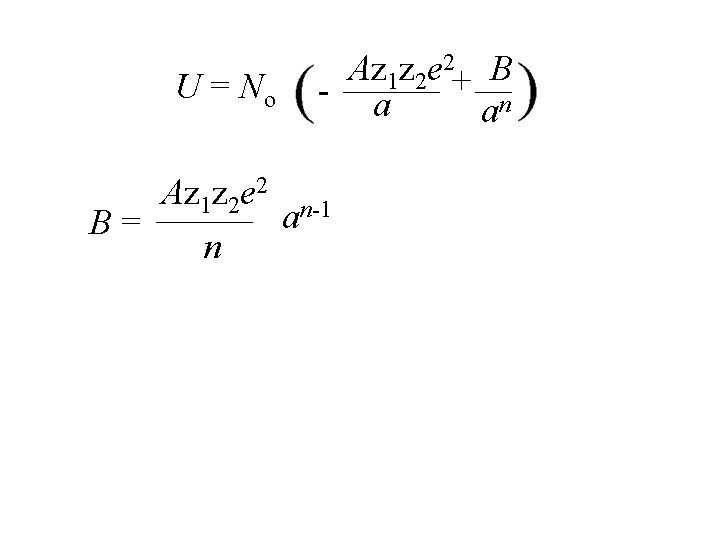
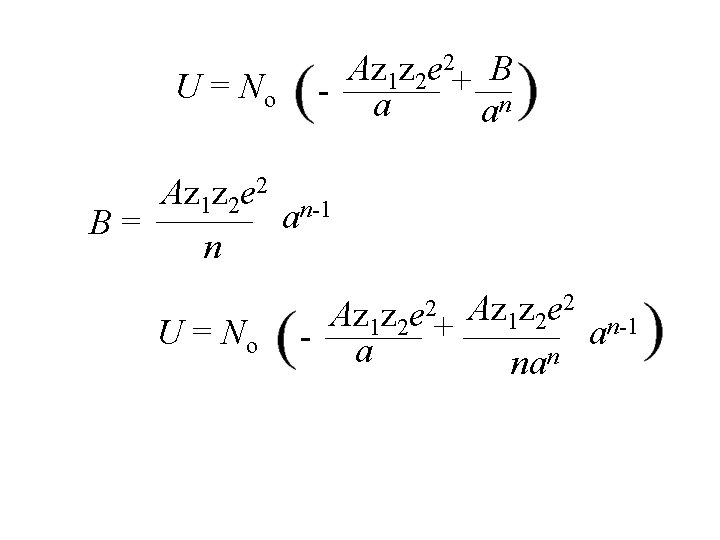
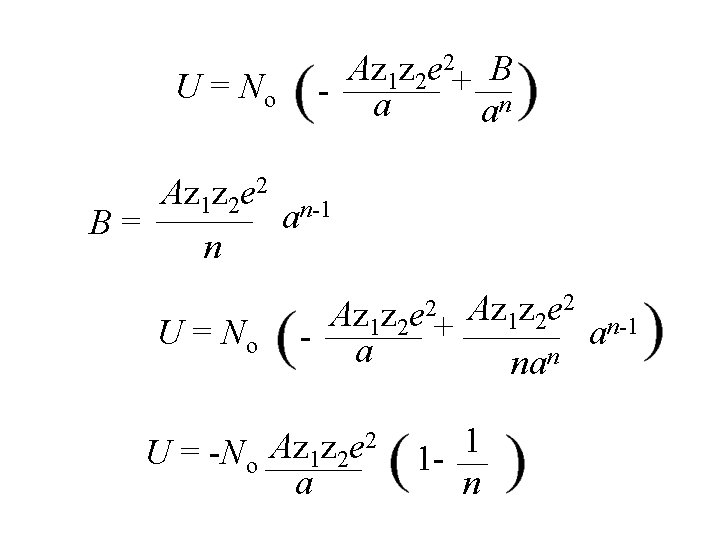

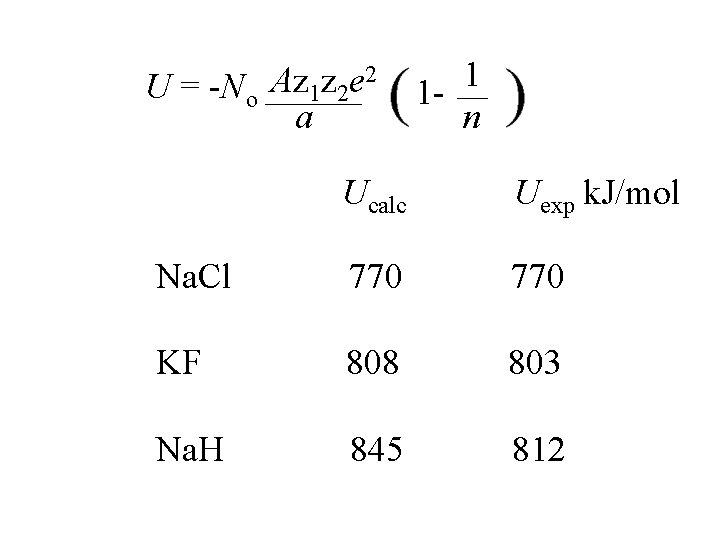








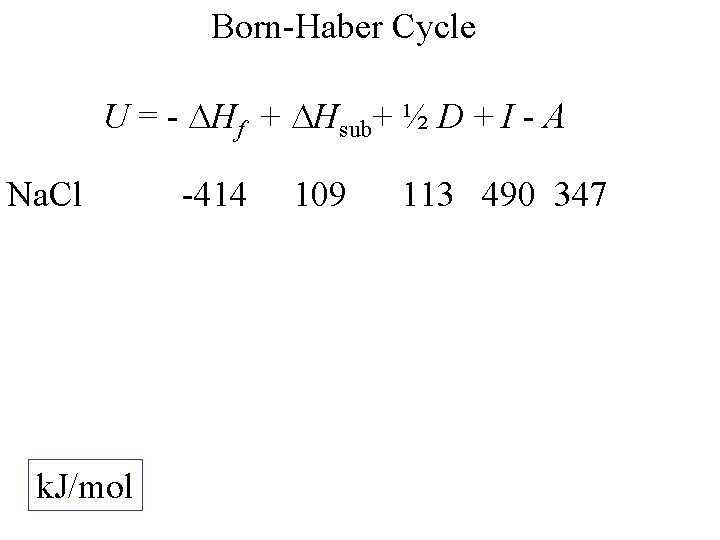
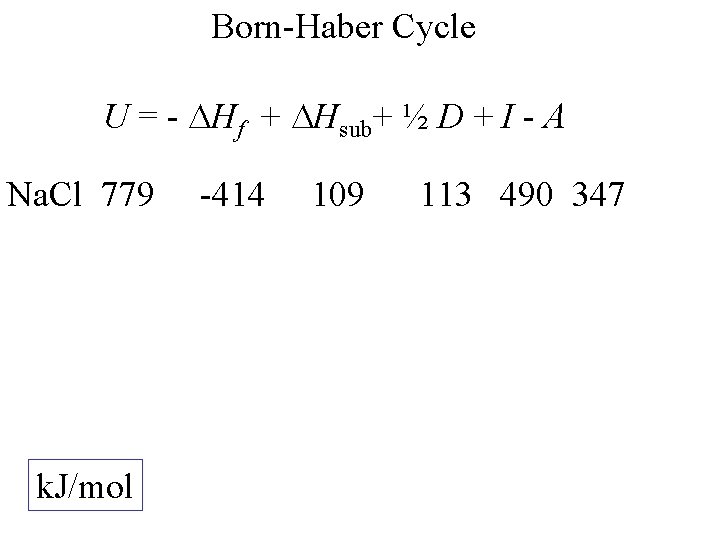
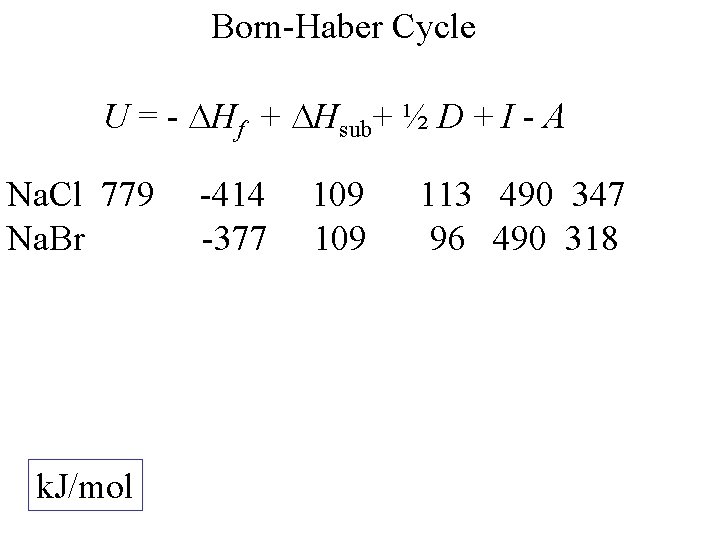
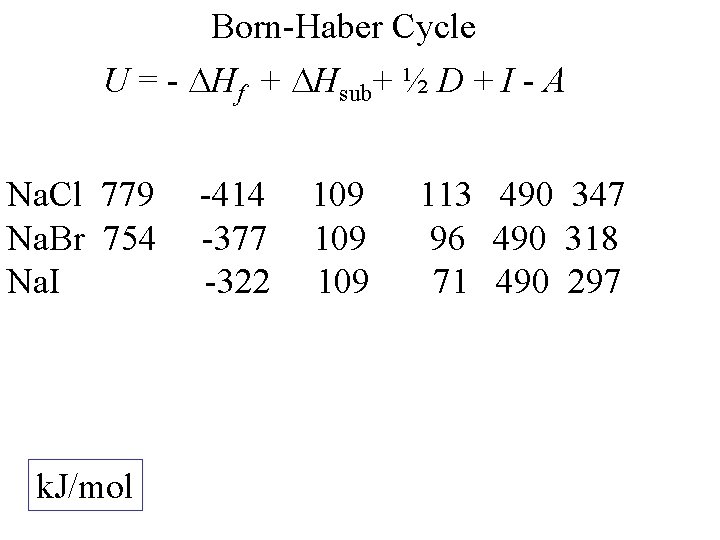
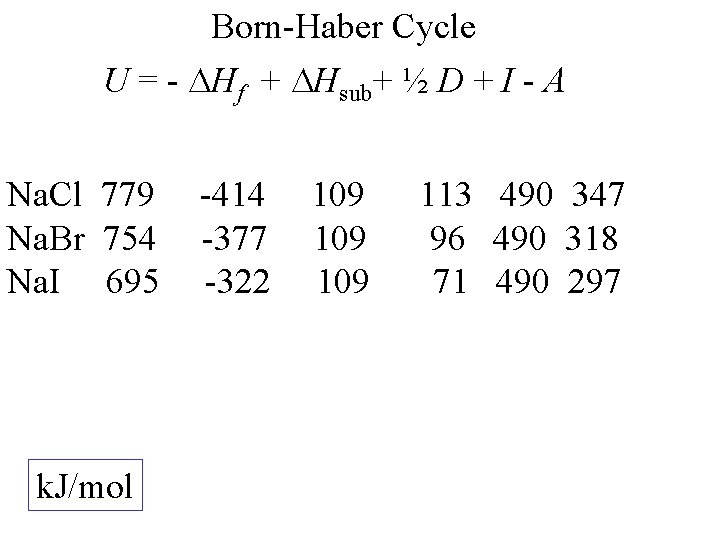
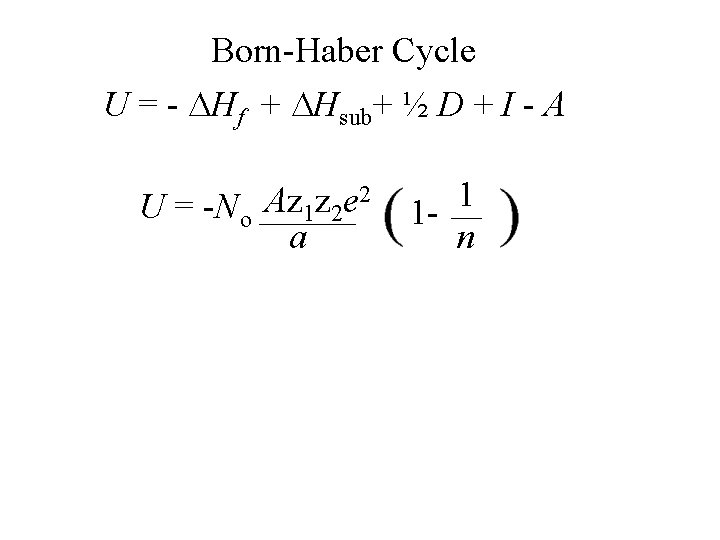
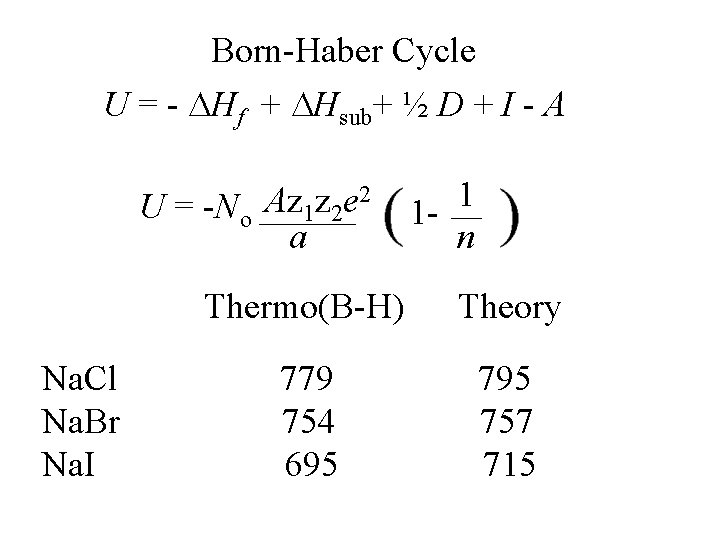


- Slides: 130

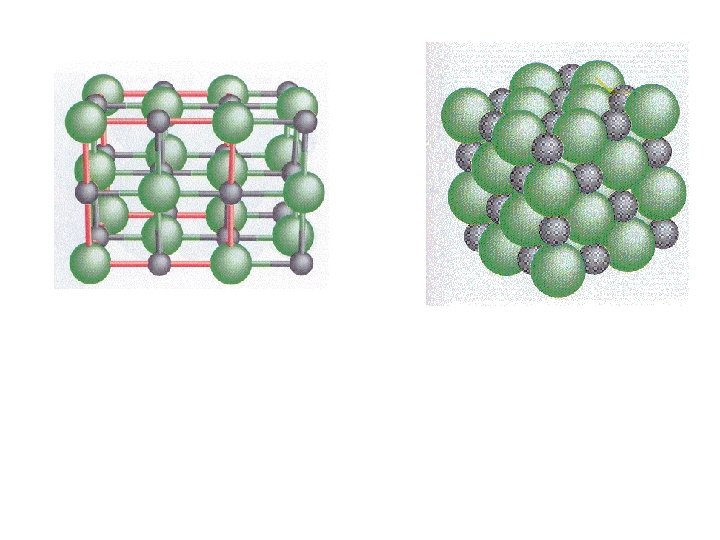
Disorder in crystals

Disorder in crystals All lattice points are not always the same.

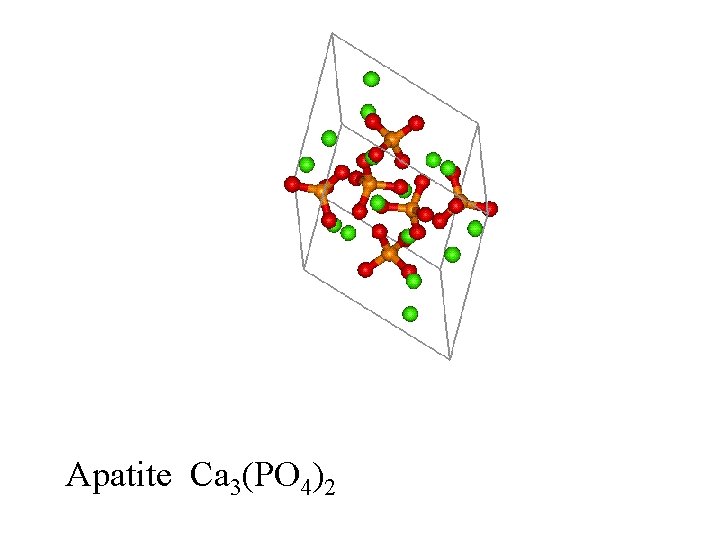
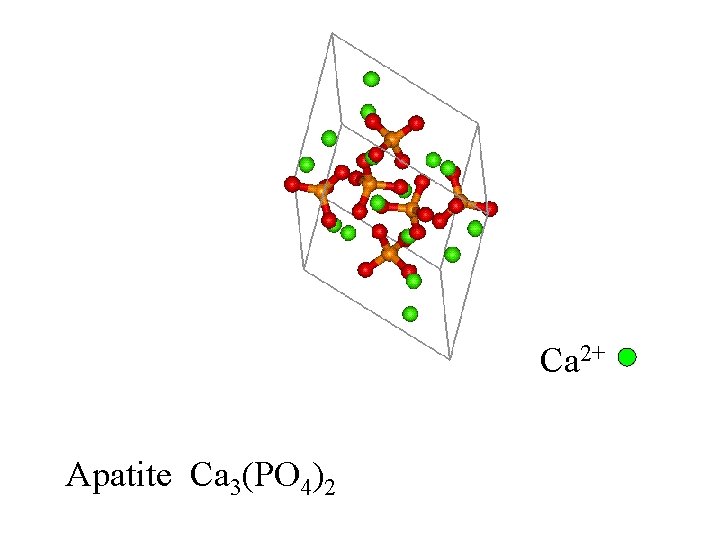
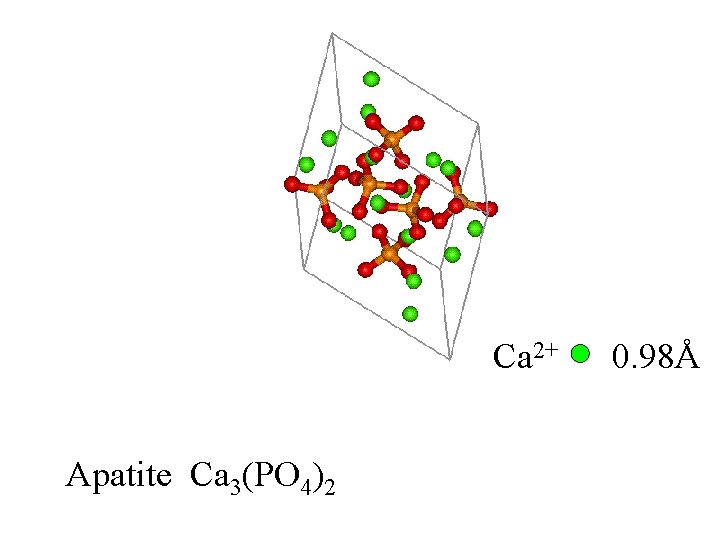
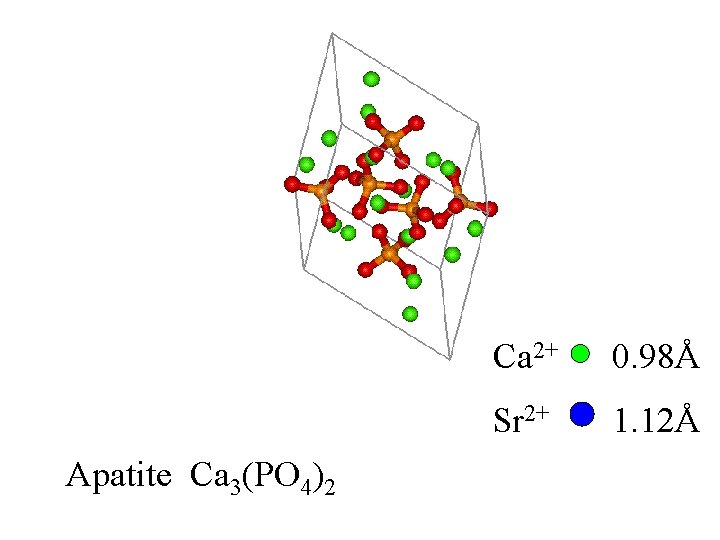
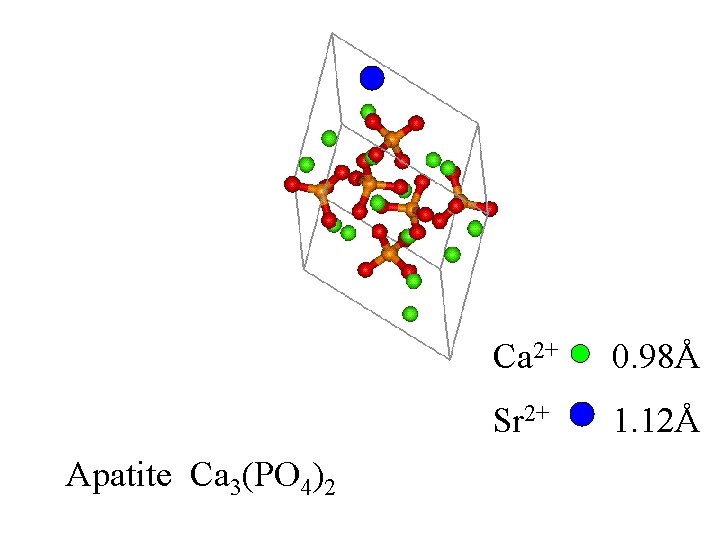
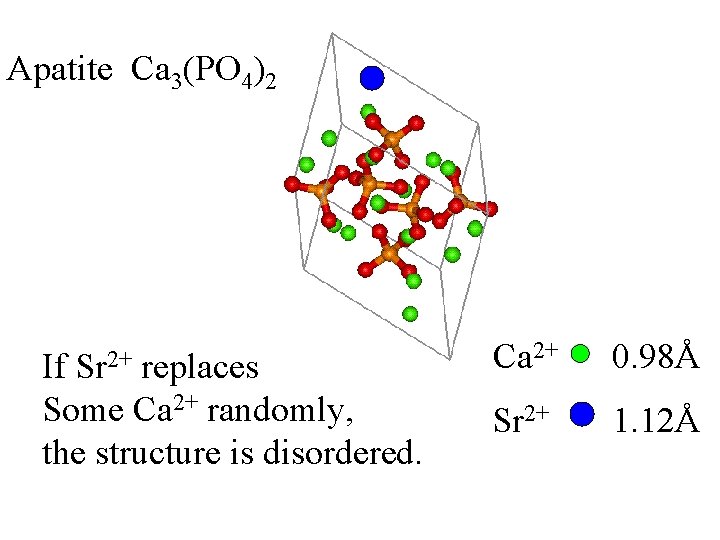
Apatite Ca 3(PO 4)2

Ca 2+ Apatite Ca 3(PO 4)2

Ca 2+ Apatite Ca 3(PO 4)2 0. 98Å

Apatite Ca 3(PO 4)2 Ca 2+ 0. 98Å Sr 2+ 1. 12Å

Apatite Ca 3(PO 4)2 Ca 2+ 0. 98Å Sr 2+ 1. 12Å

Group II Be Mg Ca Sr Ba Ra

Group II Be Mg Ca Sr Ba Ra 2+ in ionic compounds

Group II Be Mg Ca Sr Ba Ra 2+ in ionic compounds 88 Sr 38 – 86% of naturally occuring

Group II Be Mg Ca Sr Ba Ra 2+ in ionic compounds 88 Sr 38 – 86% of naturally occuring 90 Sr 38 – radioactive isotope product of nuclear weapons testing

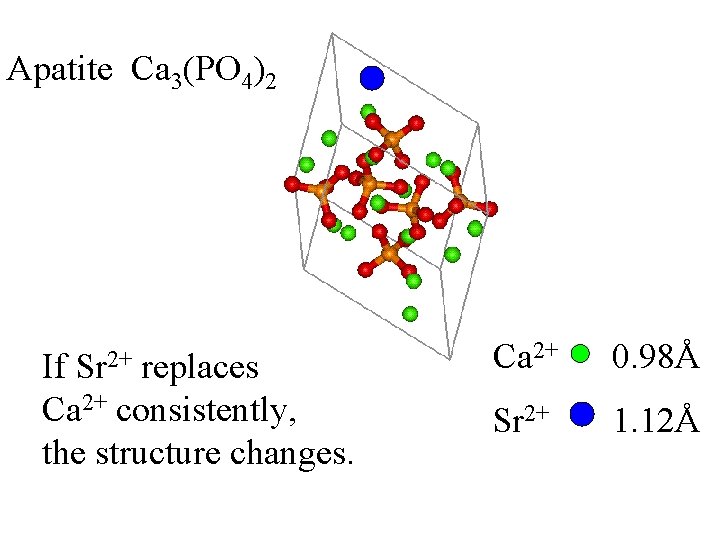
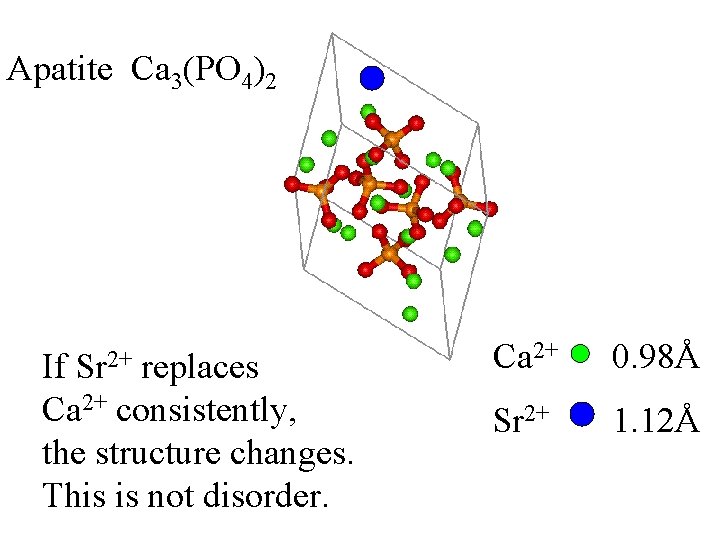
Apatite Ca 3(PO 4)2 Sr 2+ If replaces Ca 2+ consistently, the structure changes. Ca 2+ 0. 98Å Sr 2+ 1. 12Å

Apatite Ca 3(PO 4)2 Sr 2+ If replaces Ca 2+ consistently, the structure changes. This is not disorder. Ca 2+ 0. 98Å Sr 2+ 1. 12Å

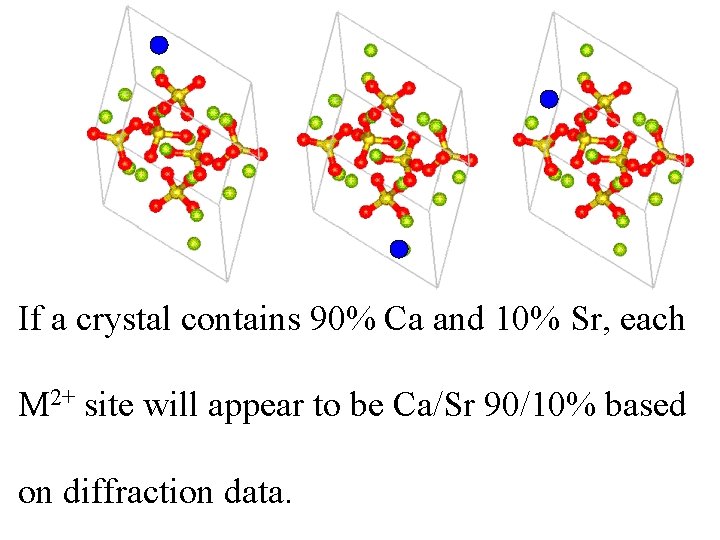
Apatite Ca 3(PO 4)2 Sr 2+ If replaces Some Ca 2+ randomly, the structure is disordered. Ca 2+ 0. 98Å Sr 2+ 1. 12Å


If a crystal contains 90% Ca and 10% Sr, each M 2+ site will appear to be Ca/Sr 90/10% based on diffraction data.





Defects in Crystals

Defects in Crystals Disorder implies that all positions are occupied, but the occupation of some sites may not be consistent.

Defects in Crystals A defect is a break in the infinite lattice.

Defects in Crystals A defect is a break in the infinite lattice. Some sites that would normally be occupied in a perfect lattice, are open.

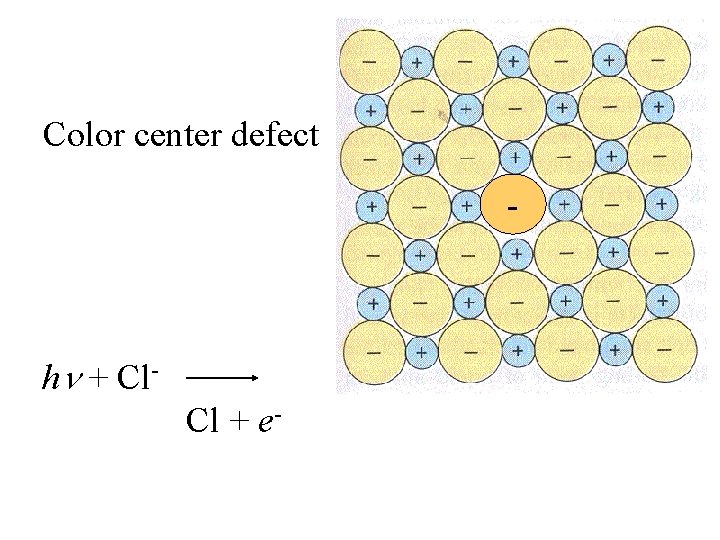
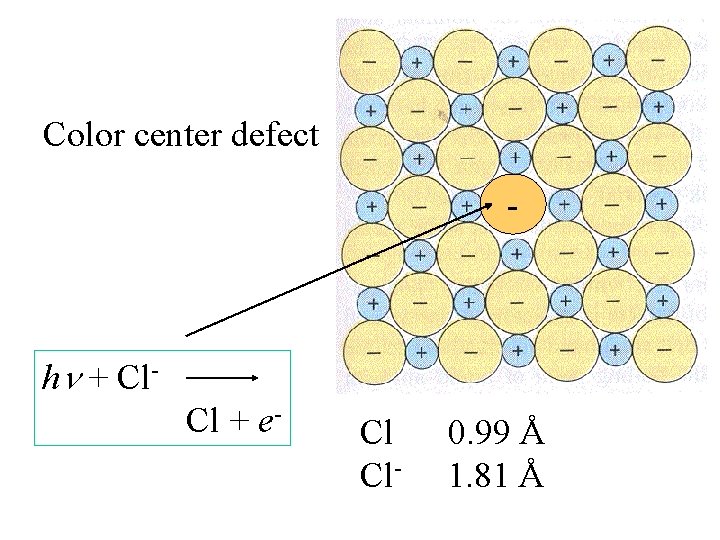
Color center defect - h + Cl. Cl + e-

Color center defect - h + Cl. Cl + e- Cl Cl- 0. 99 Å 1. 81 Å

The uncharged Cl is not affected by the + charges and is considerably smaller than the Cl-.

The uncharged Cl is not affected by the + charges and is considerably smaller than the Cl-. The Cl can move through, and leave, the lattice.

The uncharged Cl is not affected by the + charges and is considerably smaller than the Cl-. The Cl can move through, and leave, the lattice. The electron can be trapped in the octahedral vacancy left by the Cl-.

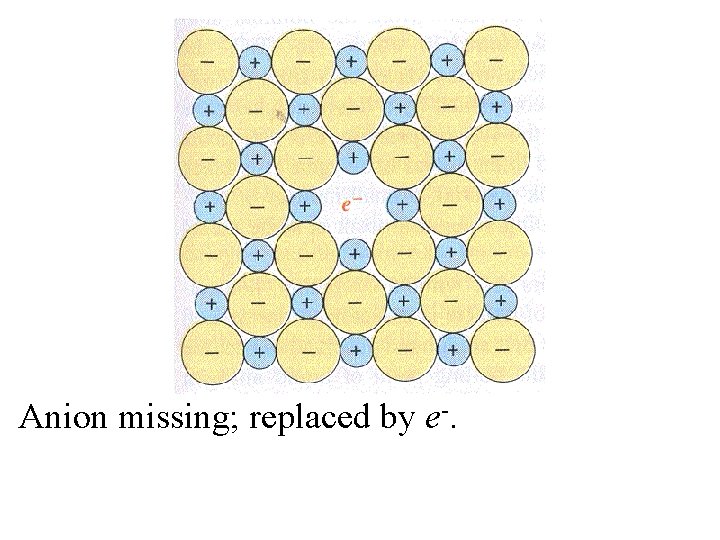
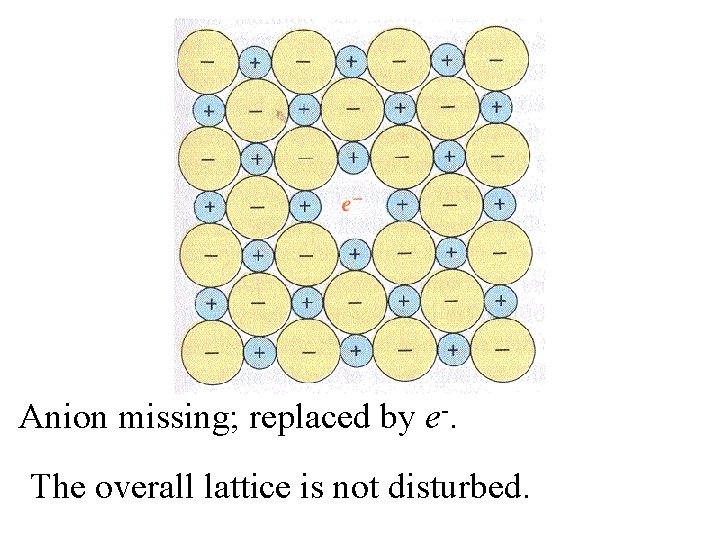
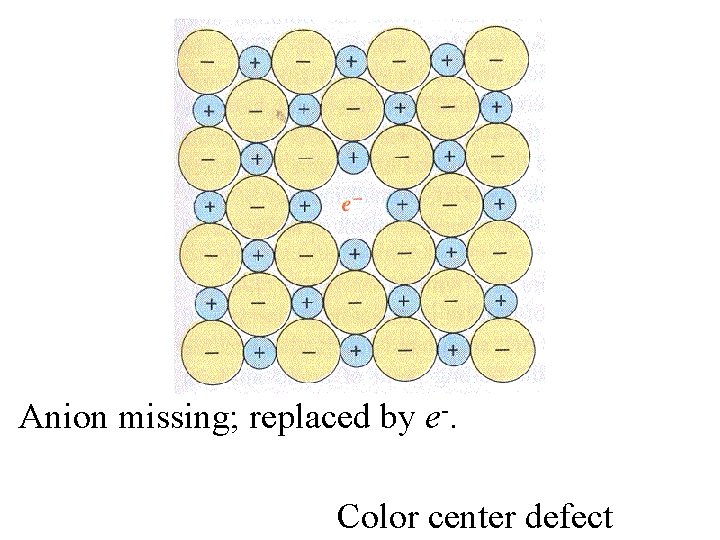
Anion missing; replaced by e-.

Anion missing; replaced by e-. The overall lattice is not disturbed.

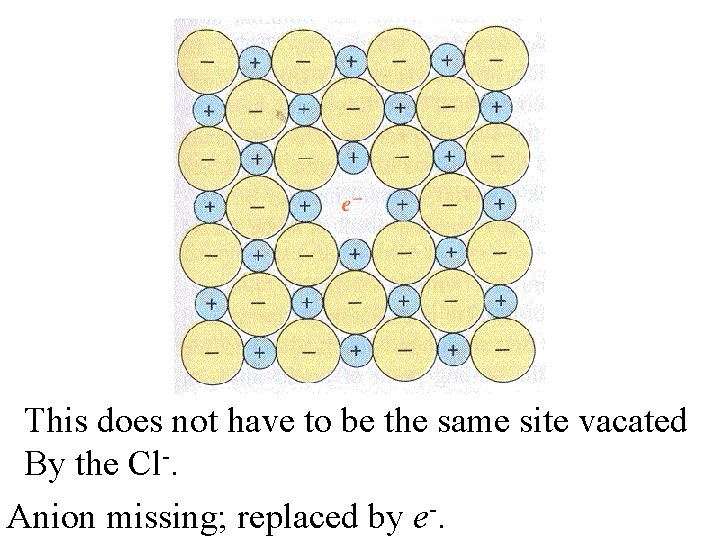
This does not have to be the same site vacated By the Cl-. Anion missing; replaced by e-.

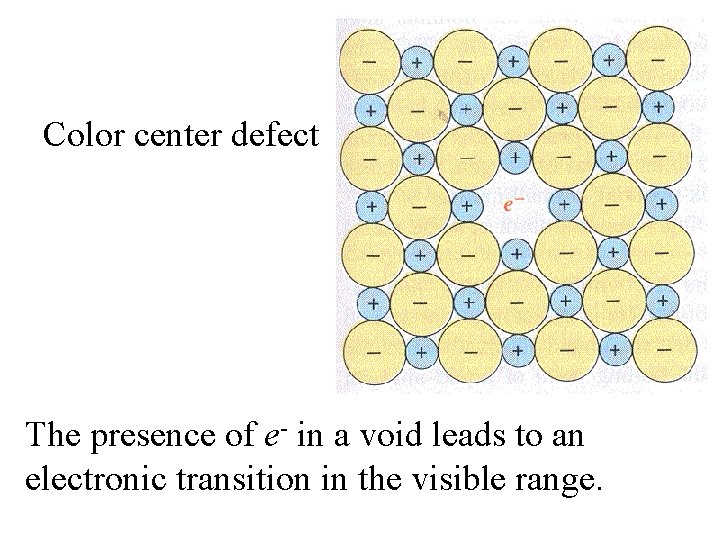
Anion missing; replaced by e-. Color center defect

Color center defect The presence of e- in a void leads to an electronic transition in the visible range.

In a real (as opposed to a ‘perfect’) Crystal, a small portion of the sites will be unoccupied.

In a real (as opposed to a ‘perfect’) Crystal, a small portion of the sites will be unoccupied. This is called a Shottky defect.

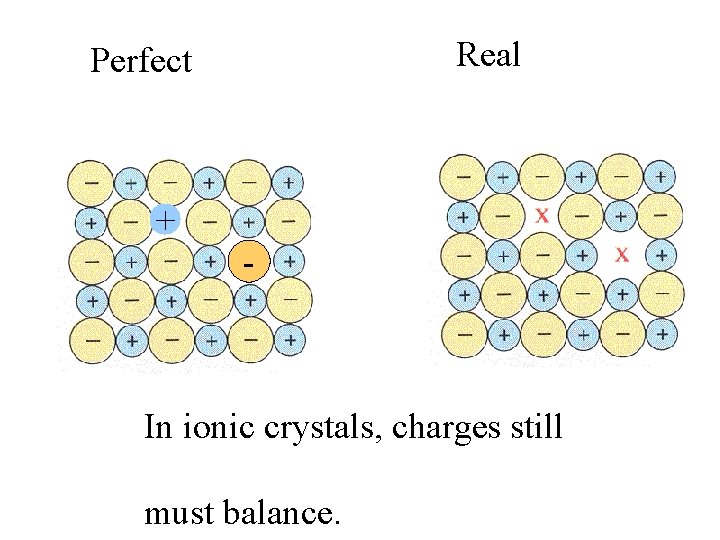
Perfect + -

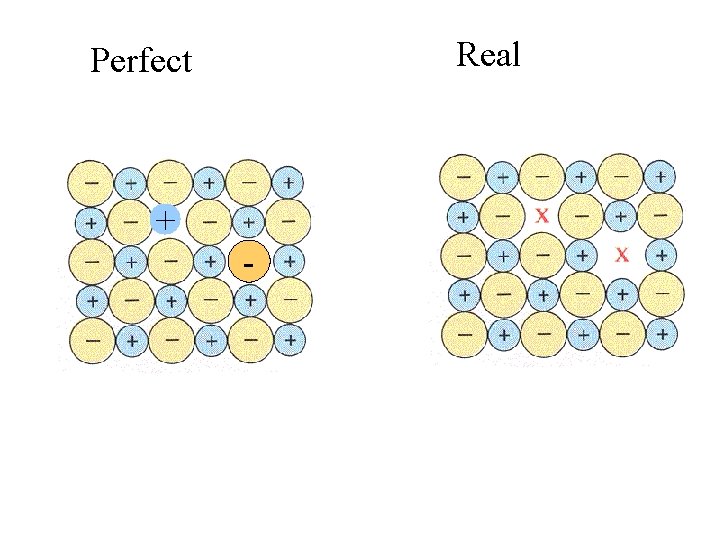
Real Perfect + -

Real Perfect + - In ionic crystals, charges still must balance.

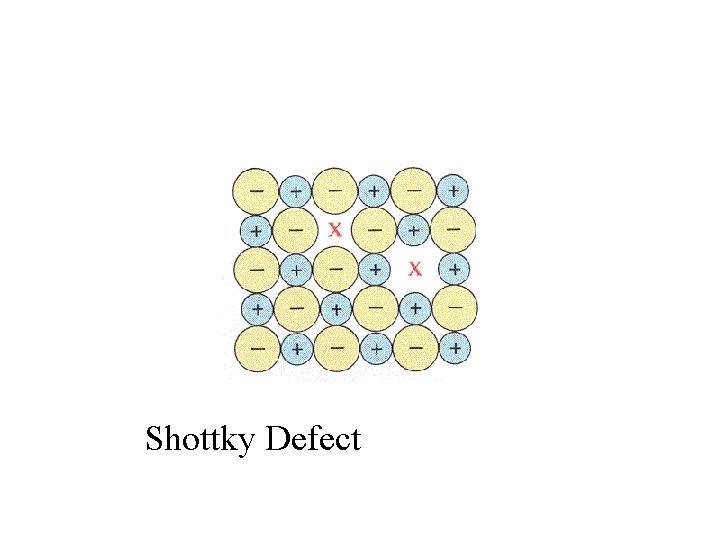
Shottky Defect

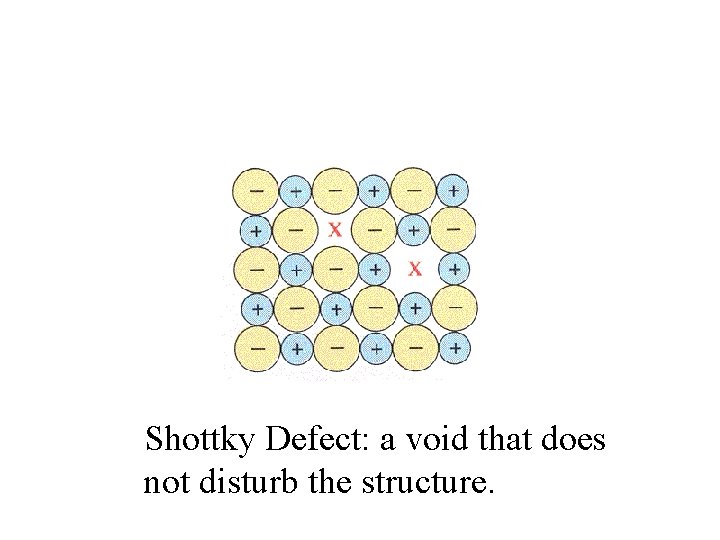
Shottky Defect: a void that does not disturb the structure.

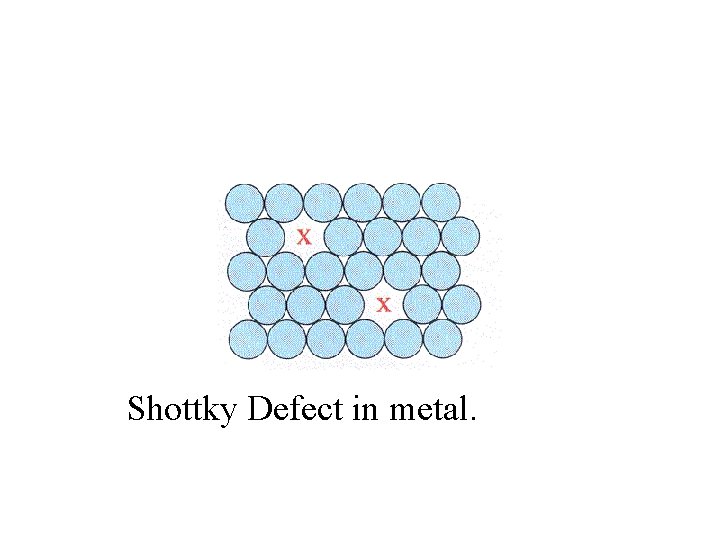
Shottky Defect in metal.

Other defects may alter the lattice.

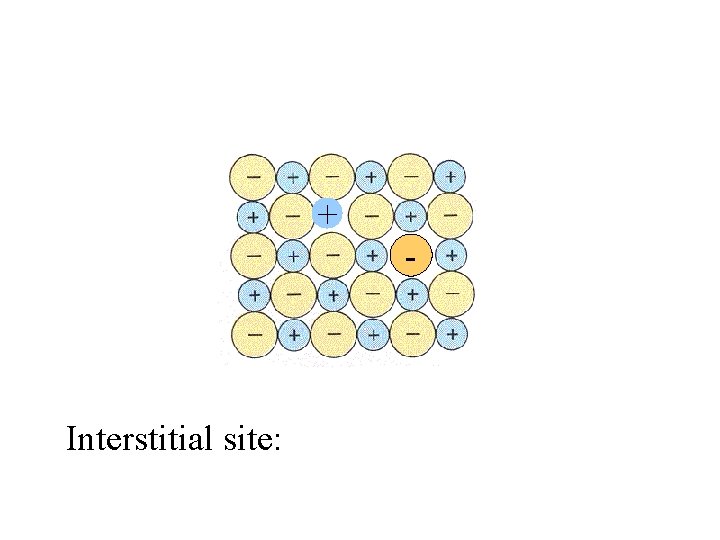
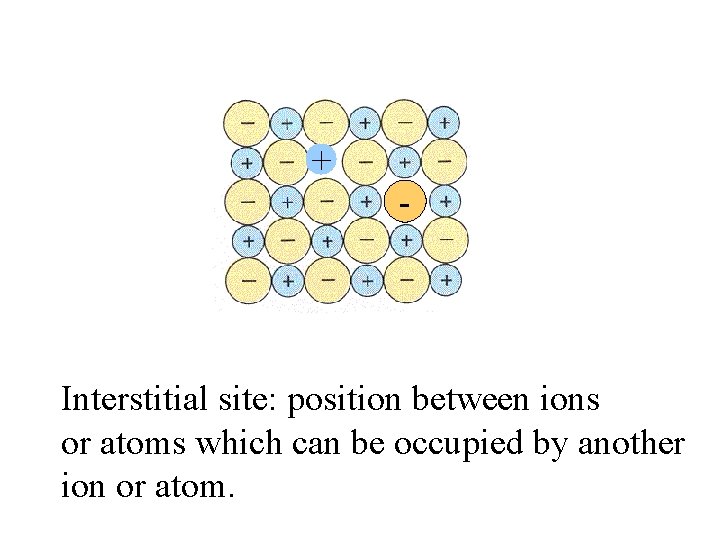
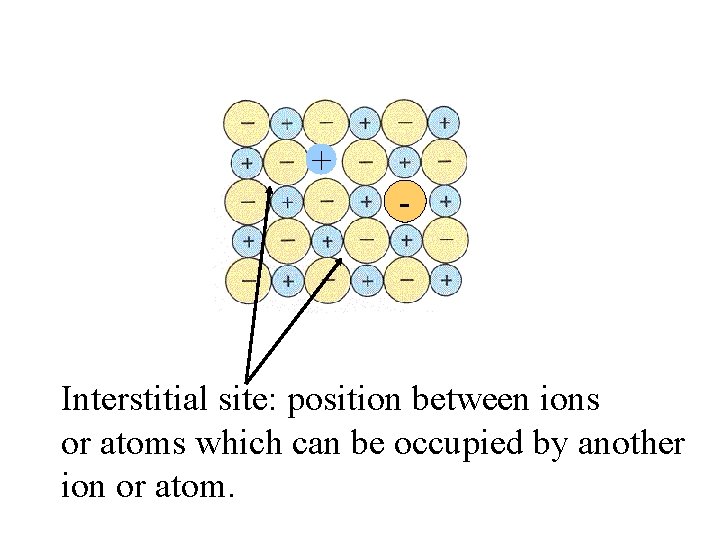
+ - Interstitial site:

+ - Interstitial site: position between ions or atoms which can be occupied by another ion or atom.

+ - Interstitial site: position between ions or atoms which can be occupied by another ion or atom.

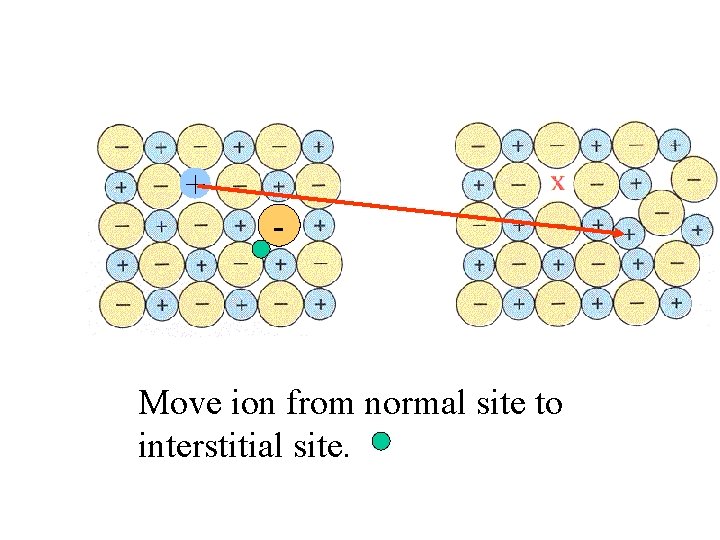
+ - Move ion from normal site to interstitial site.

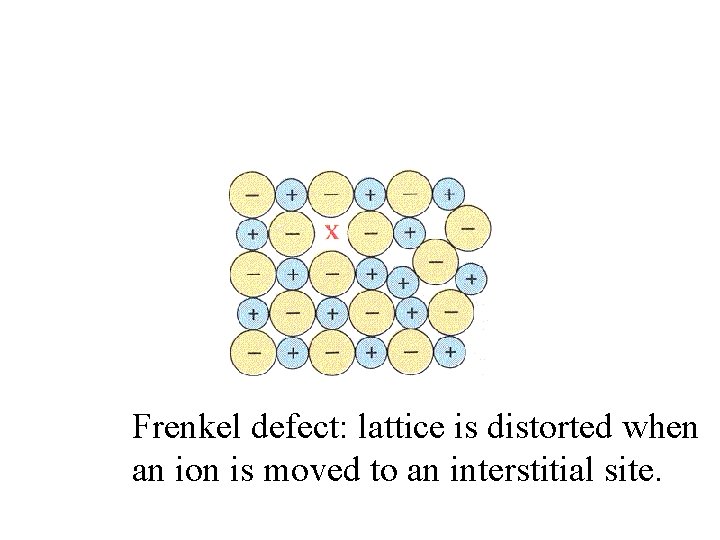
Frenkel defect: lattice is distorted when an ion is moved to an interstitial site.

Defects tend to be dynamic.


Nonstoichiometric Compounds


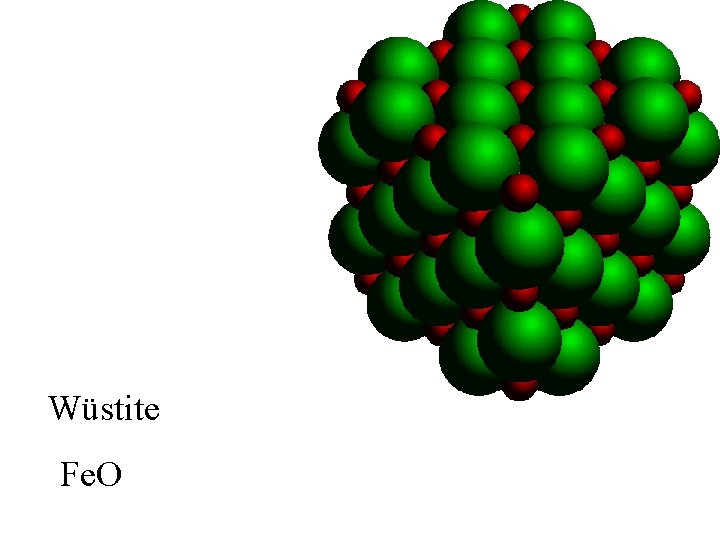
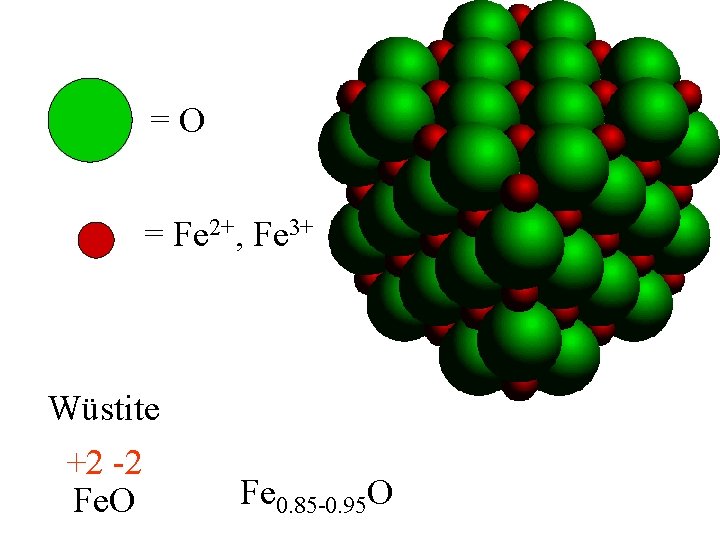
Wüstite

Wüstite Fe. O

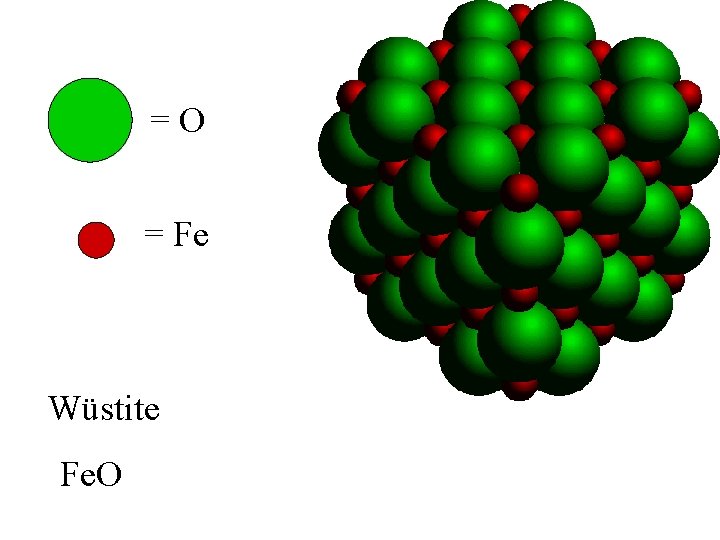
=O = Fe Wüstite Fe. O

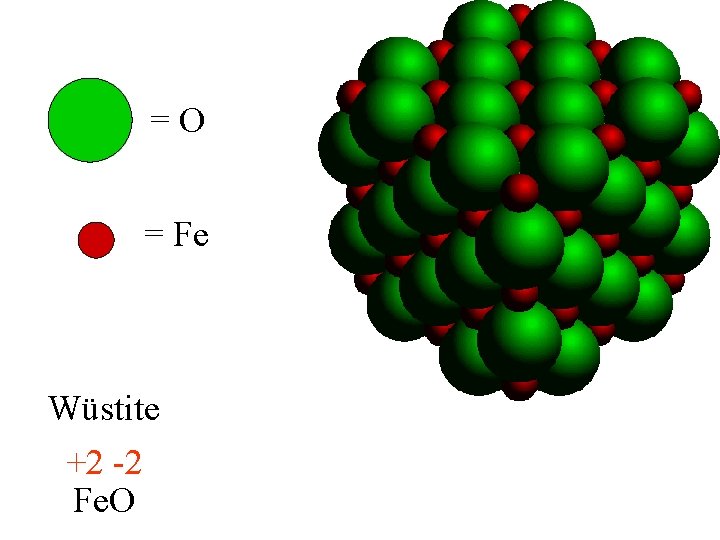
=O = Fe Wüstite +2 -2 Fe. O

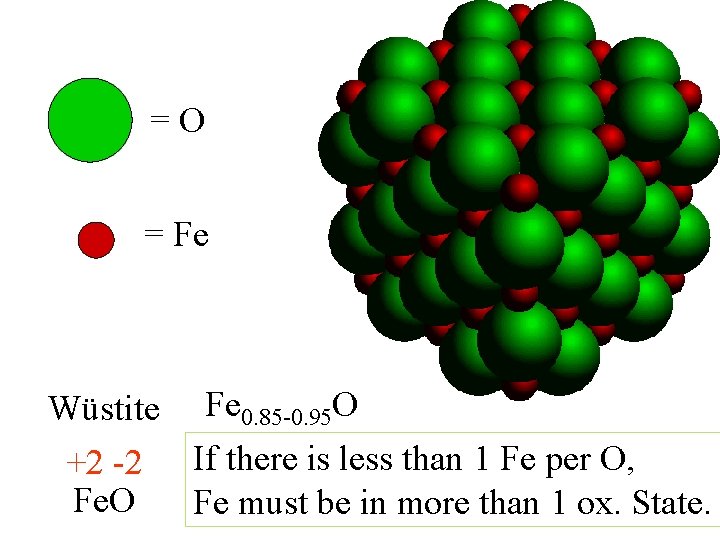
=O = Fe Wüstite Fe 0. 85 -0. 95 O +2 -2 If there is less than 1 Fe per O, Fe. O Fe must be in more than 1 ox. State.

=O = Fe 2+, Fe 3+ Wüstite +2 -2 Fe. O Fe 0. 85 -0. 95 O

Fe 0. 85 -0. 95 O Fe 0. 85 O
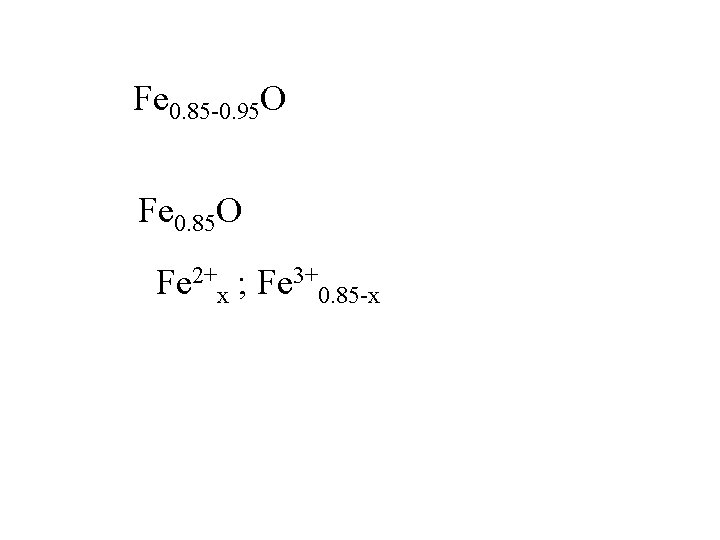
Fe 0. 85 -0. 95 O Fe 0. 85 O Fe 2+x ; Fe 3+0. 85 -x

Fe 0. 85 -0. 95 O Fe 0. 85 O Fe 2+x ; Fe 3+0. 85 -x 2 x + 3(0. 85 -x) = 2
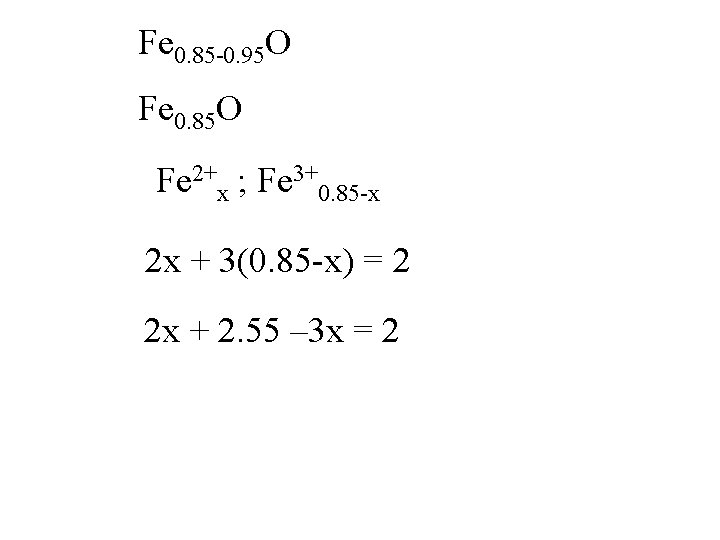
Fe 0. 85 -0. 95 O Fe 0. 85 O Fe 2+x ; Fe 3+0. 85 -x 2 x + 3(0. 85 -x) = 2 2 x + 2. 55 – 3 x = 2
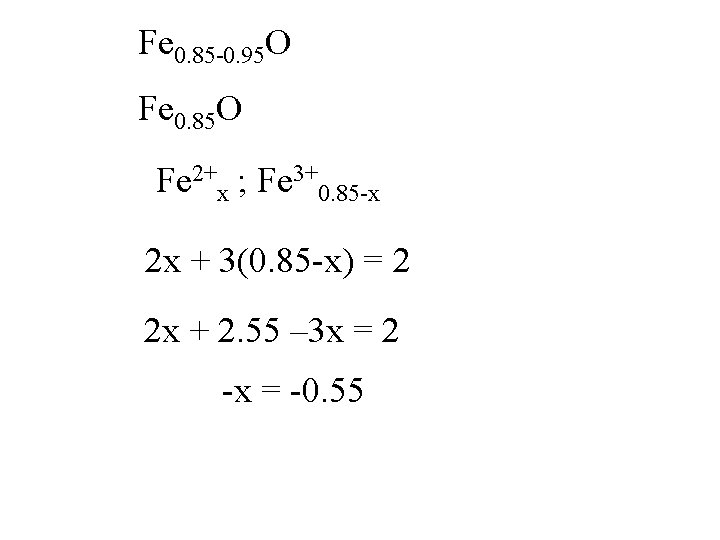
Fe 0. 85 -0. 95 O Fe 0. 85 O Fe 2+x ; Fe 3+0. 85 -x 2 x + 3(0. 85 -x) = 2 2 x + 2. 55 – 3 x = 2 -x = -0. 55
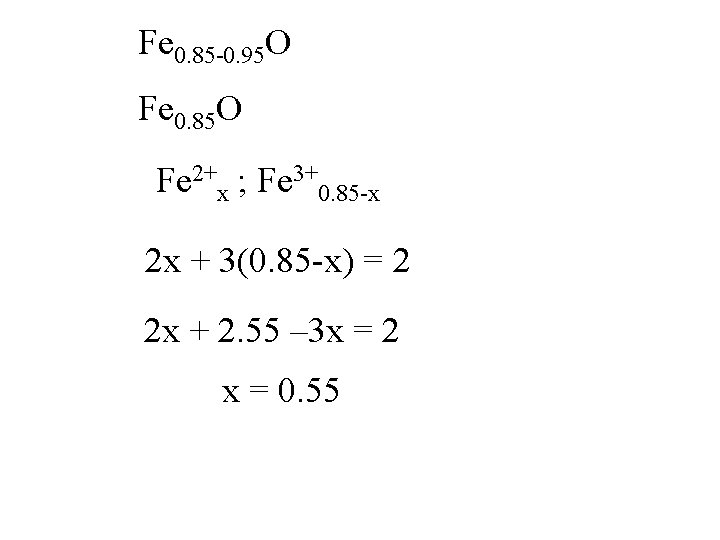
Fe 0. 85 -0. 95 O Fe 0. 85 O Fe 2+x ; Fe 3+0. 85 -x 2 x + 3(0. 85 -x) = 2 2 x + 2. 55 – 3 x = 2 x = 0. 55
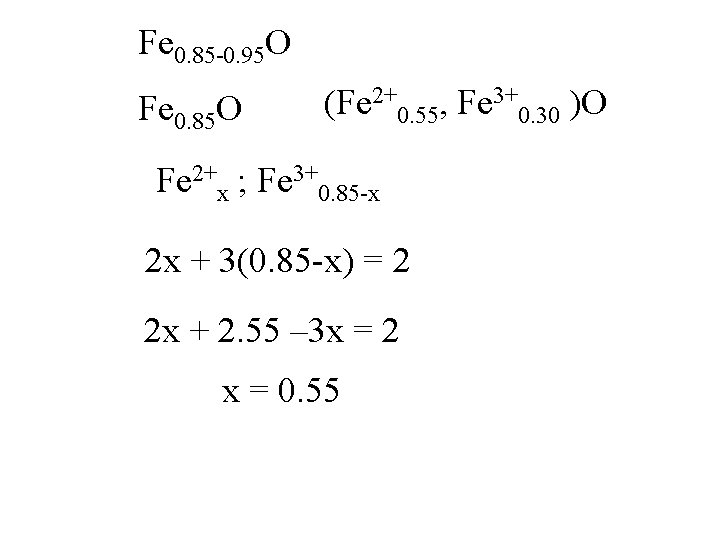
Fe 0. 85 -0. 95 O Fe 0. 85 O (Fe 2+0. 55, Fe 3+0. 30 )O Fe 2+x ; Fe 3+0. 85 -x 2 x + 3(0. 85 -x) = 2 2 x + 2. 55 – 3 x = 2 x = 0. 55

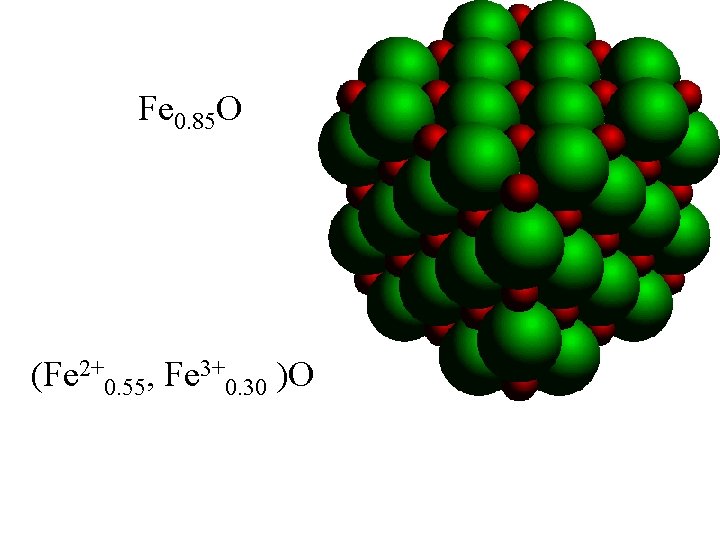
Fe 0. 85 O (Fe 2+0. 55, Fe 3+0. 30 )O

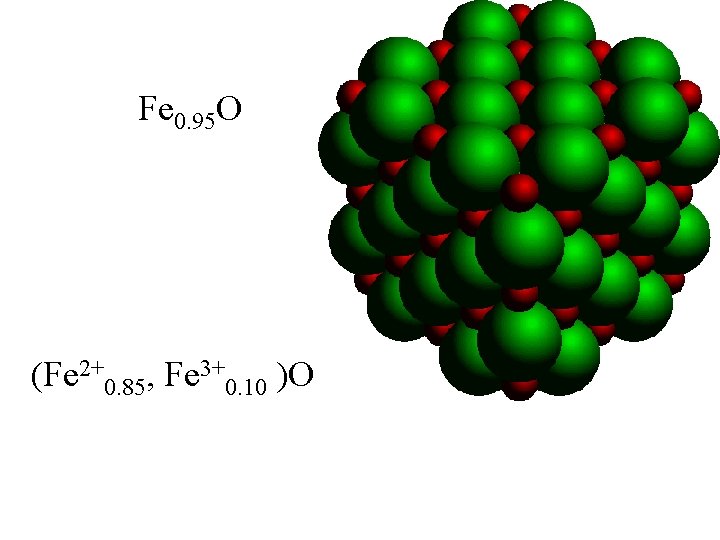
Fe 0. 95 O (Fe 2+0. 85, Fe 3+0. 10 )O

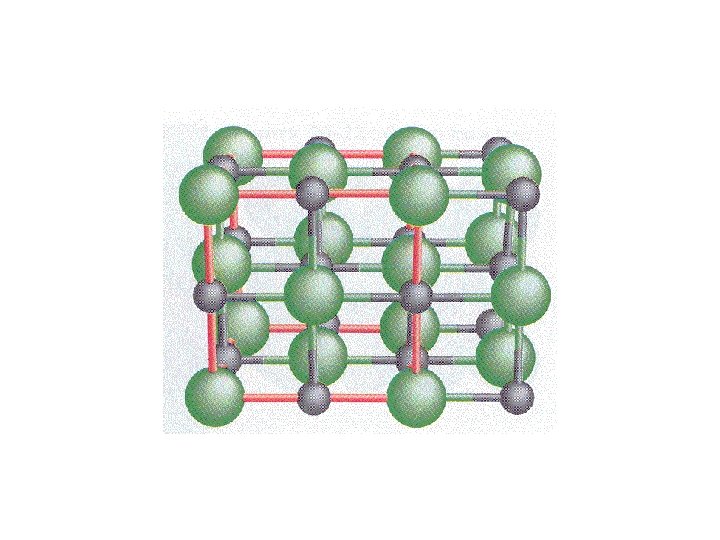
Thermodynamics of Crystals



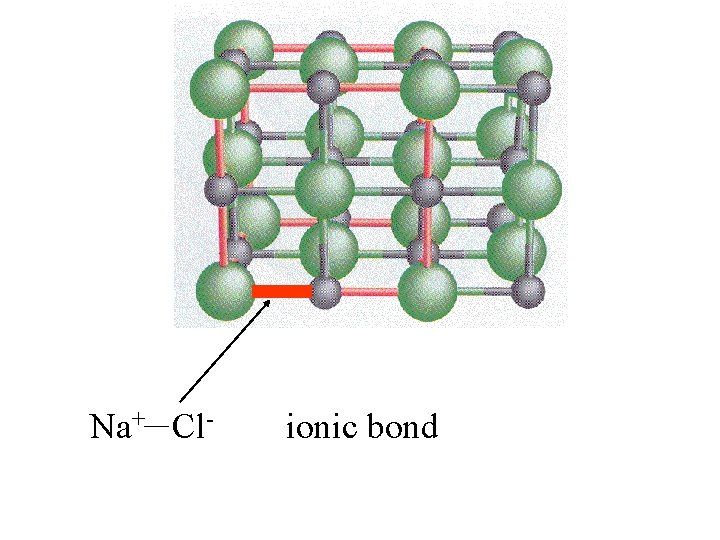
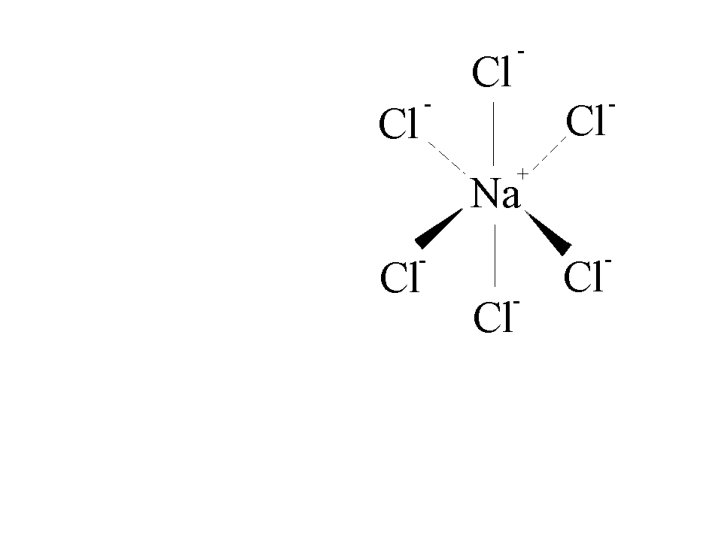
Na+ Cl- ionic bond

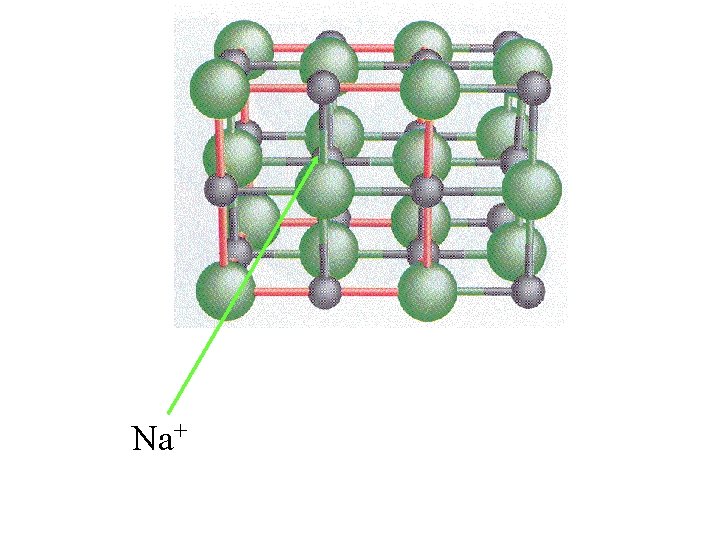
Na+



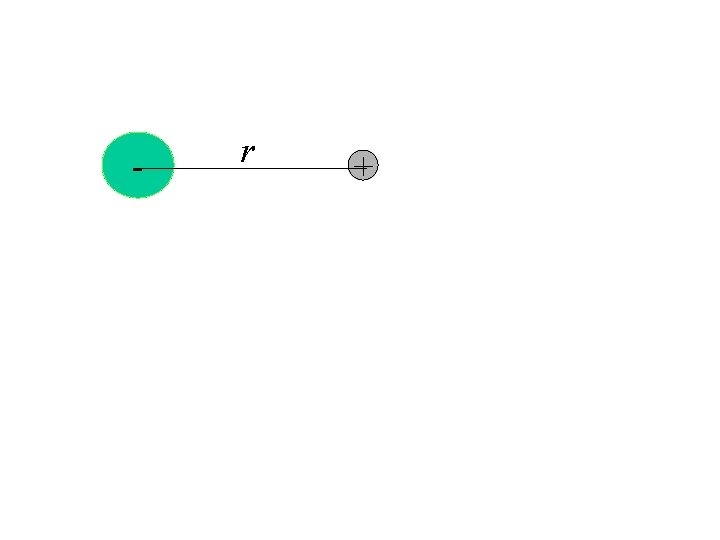
Account for ionic attractions and repulsions based on the distance of the ions and their charges.


- r + The energy of this pair depends on coulombic attraction and repulsion. 2 z z e b 1 2 + Ep = - r rn

- r + The energy of this pair depends on coulombic attraction and repulsion. 2 z z e b 1 2 + Ep = - r rn attraction term (decreases energy)

- r + The energy of this pair depends on coulombic attraction and repulsion. 2 z z e b 1 2 + Ep = - r rn attraction term Repulsive term (decreases energy) (increases energy)

- r + 2 z z e b 1 2 + Ep = - r rn z = charge number

- r + 2 z z e b 1 2 + Ep = - r rn z = charge number e = electron charge

- r + 2 z z e b 1 2 + Ep = - r rn z = charge number e = electron charge r = internuclear separation

- r + 2 z z e b 1 2 + Ep = - r rn z = charge number e = electron charge r = internuclear separation b, n are repulsion constants

- r + 2 z z e b 1 2 + Ep = - r rn z = charge number e = electron charge r = internuclear separation b, n are repulsion* constants * repulsion due to physical contact, not coulombic repulsion

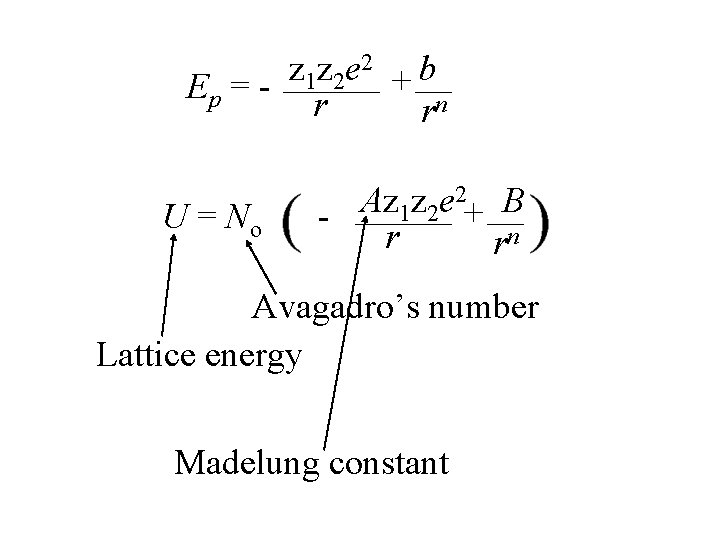
2 z z e b 1 2 + Ep = - r rn The lattice energy for a mole of Na. Cl can be evaluated by multiplying the energy by No and including a factor that accounts for all ion-ion interactions.
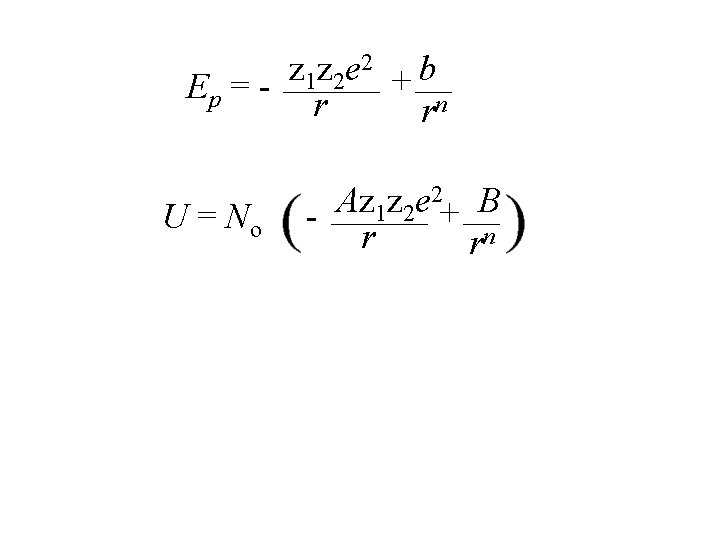
2 z z e b 1 2 + Ep = - r rn U = No 2 Az z e 1 2 + B r rn

2 z z e b 1 2 + Ep = - r rn U = No Lattice energy 2 Az z e 1 2 + B r rn

2 z z e b 1 2 + Ep = - r rn U = No Az 1 z 2 e 2+ B - r rn Avagadro’s number Lattice energy

2 z z e b 1 2 + Ep = - r rn U = No 2 Az z e 1 2 + B r rn Avagadro’s number Lattice energy Madelung constant

Repeat S&P pg 80


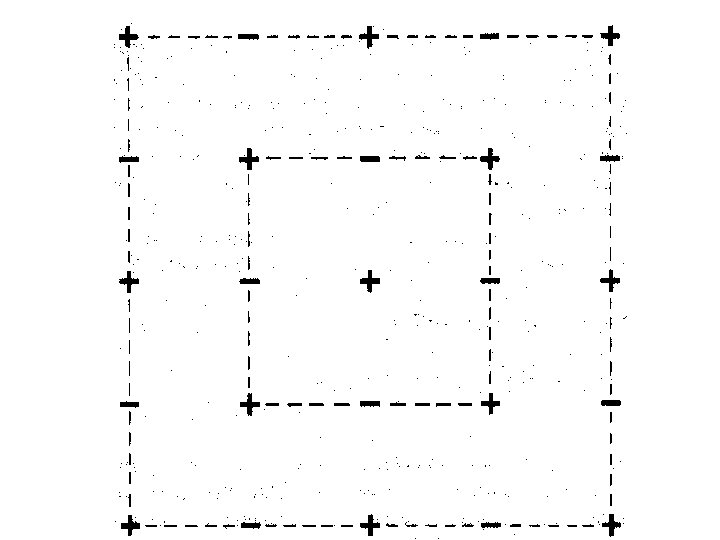
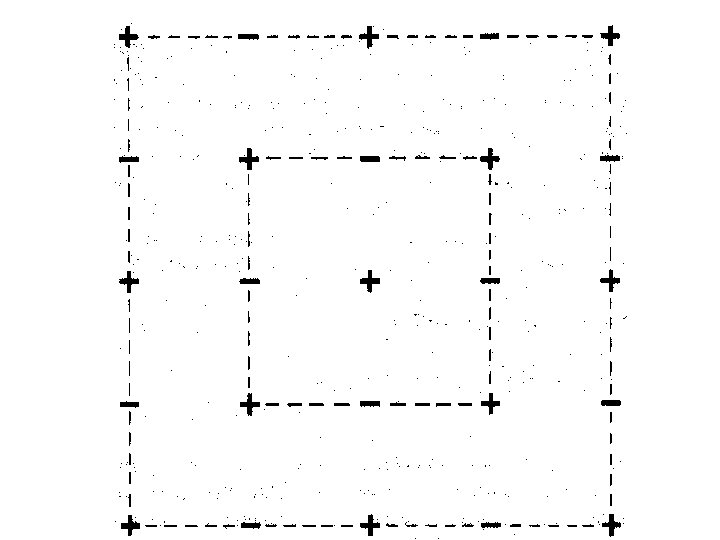
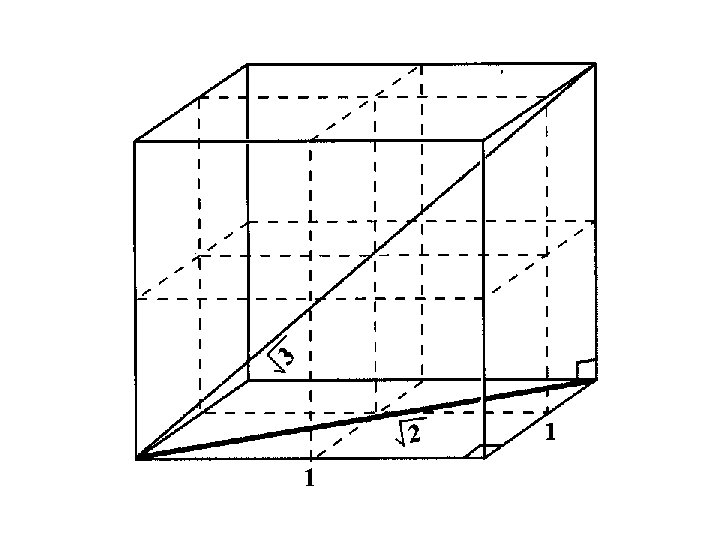
When an individual ion is considered in a cubic lattice, there is a group of oppositely charged ions at a given distance followed by a group of like charged ions at a longer distance.

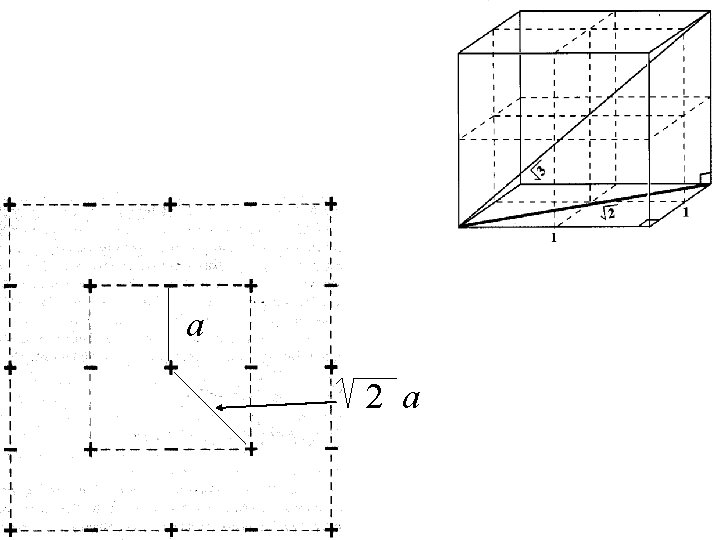
If r = a in Na. Cl then there are 6 Cl- at distance a from Na+.

If r = a in Na. Cl then there are 6 Cl- at distance a from Na+. There are 12 Na+ at a distance of 2 a from the initial Na+.

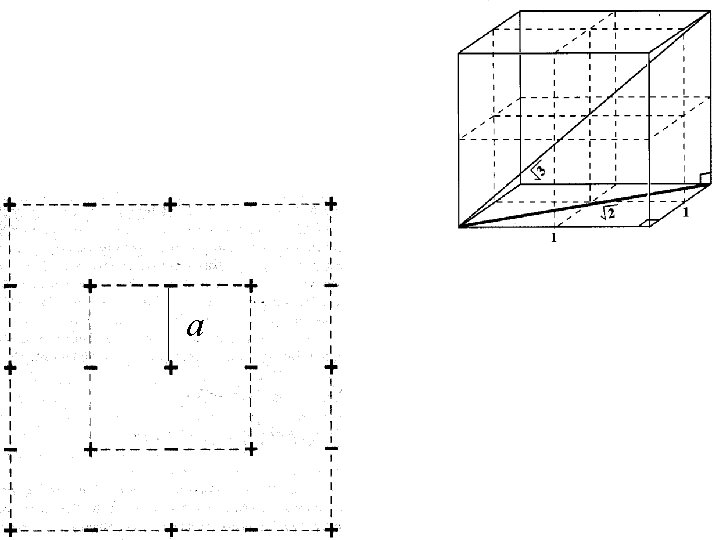
a

a 2 a
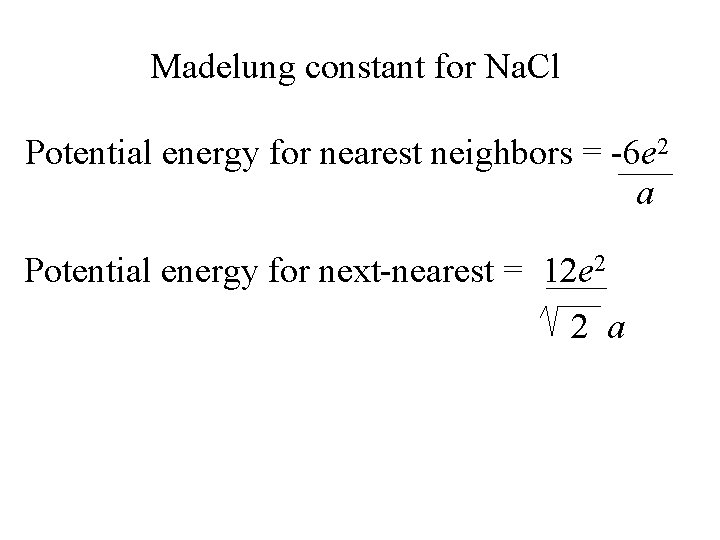
Madelung constant for Na. Cl Potential energy for nearest neighbors = -6 e 2 a Potential energy for next-nearest = 12 e 2 2 a
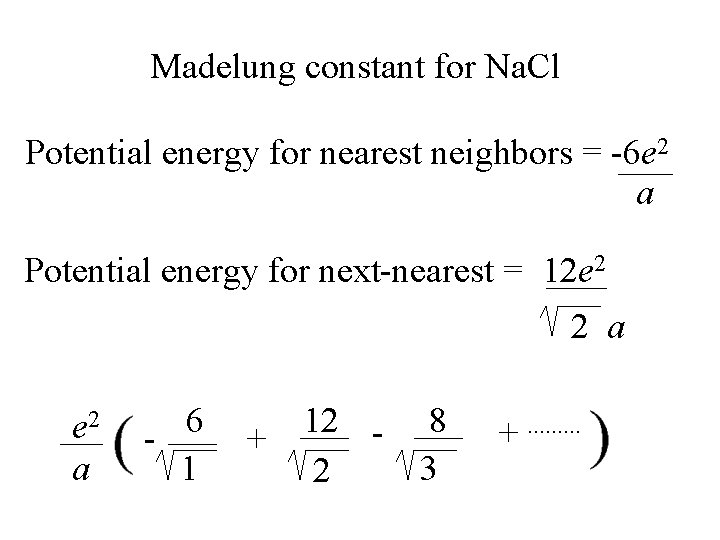
Madelung constant for Na. Cl Potential energy for nearest neighbors = -6 e 2 a Potential energy for next-nearest = 12 e 2 2 a e 2 a 6 1 + 12 2 8 3 +. .
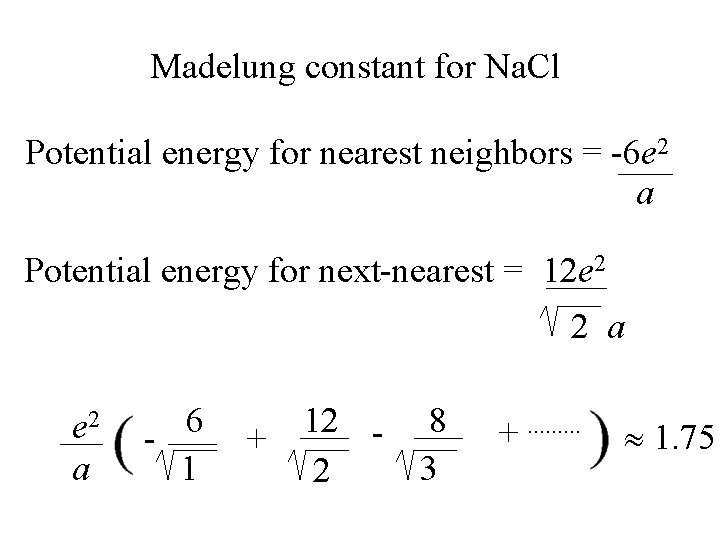
Madelung constant for Na. Cl Potential energy for nearest neighbors = -6 e 2 a Potential energy for next-nearest = 12 e 2 2 a e 2 a 6 1 + 12 2 8 3 +. . 1. 75
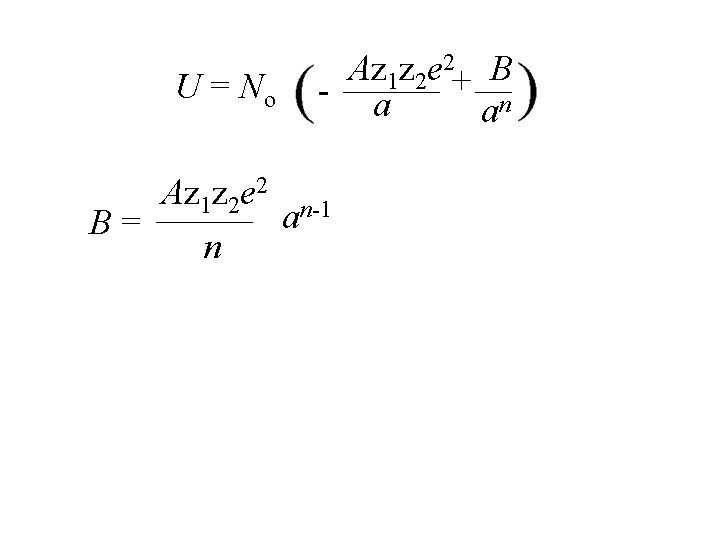
U = No Az 1 z 2 e 2+ B - a an Az 1 z 2 e 2 n-1 a B= n
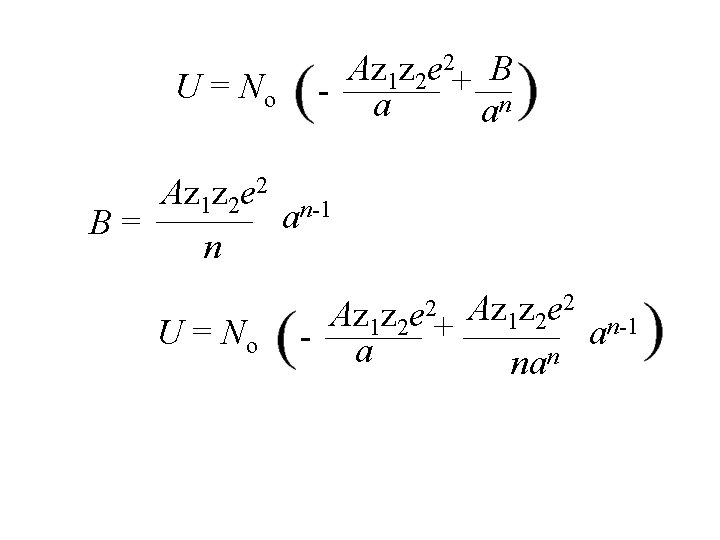
Az 1 z 2 e 2+ B - a an U = No Az 1 z 2 e 2 n-1 a B= n U = No - Az 1 z 2 e 2+ a Az 1 z 2 e 2 n-1 a nan
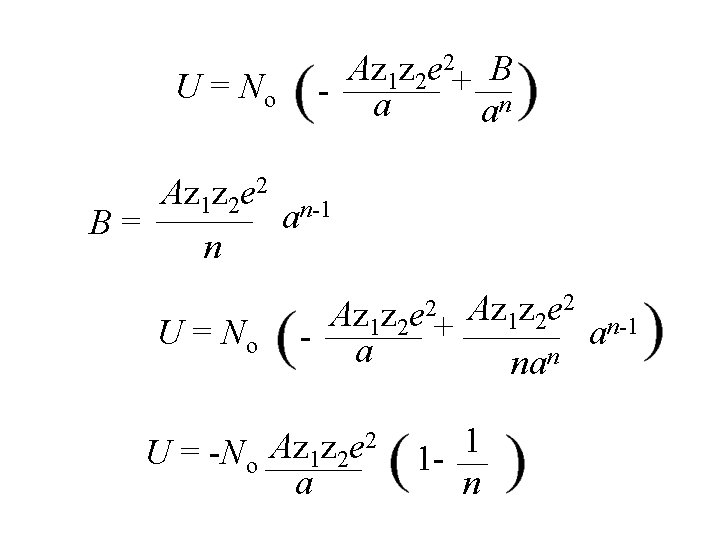
Az 1 z 2 e 2+ B - a an U = No Az 1 z 2 e 2 n-1 a B= n U = No - Az 1 z 2 e 2+ a 2 Az z e U = -No 1 2 a Az 1 z 2 e 2 n-1 a nan 1 1 n

2 Az z e U = -No 1 2 a 1 1 n n varies from 9 to 12; it is determined from the compressibility of the material
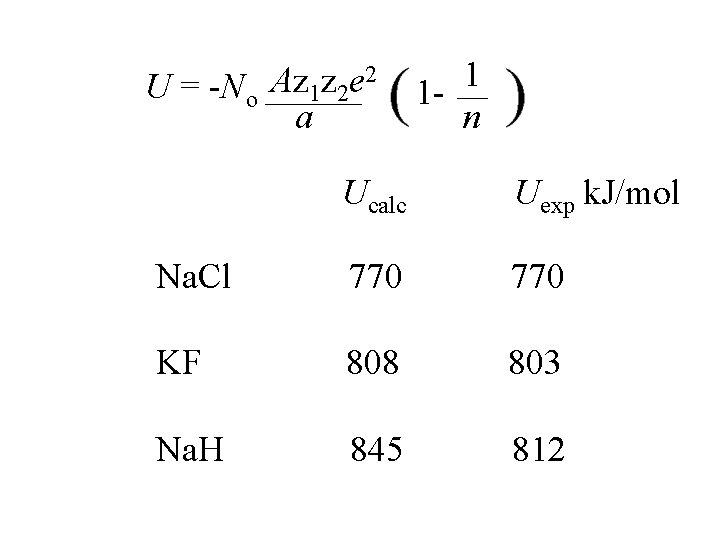
2 Az z e U = -No 1 2 a 1 1 n Ucalc Uexp k. J/mol Na. Cl 770 KF 808 803 Na. H 845 812

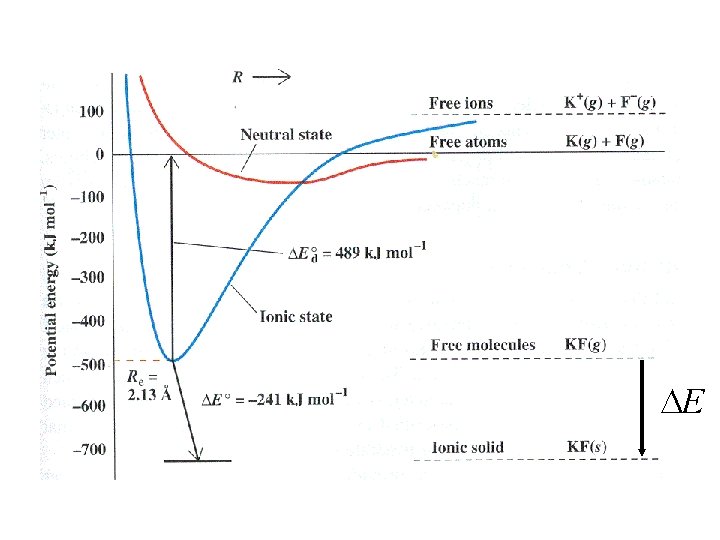
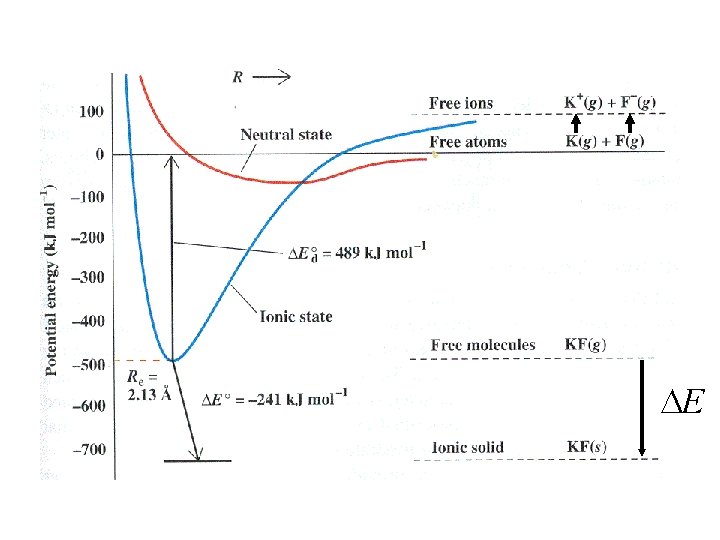
Where do experimental values for U come from?

E

E

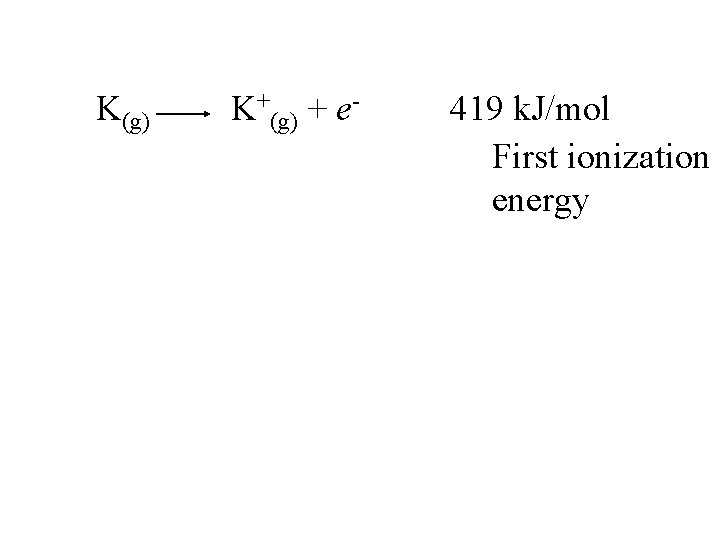
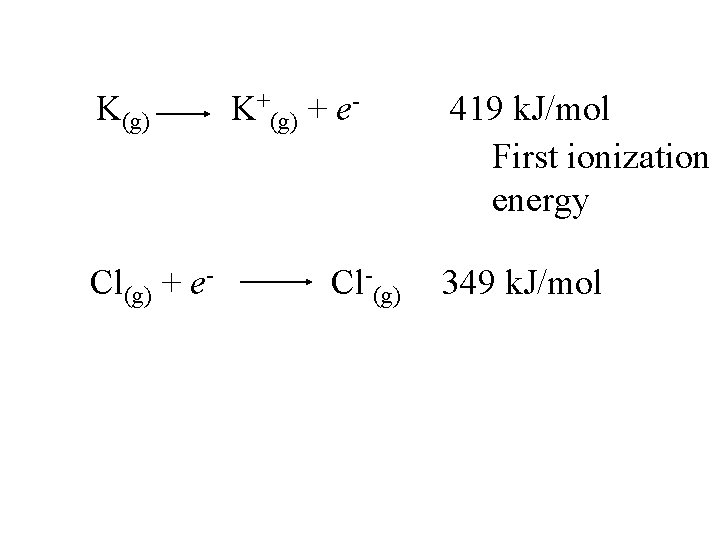
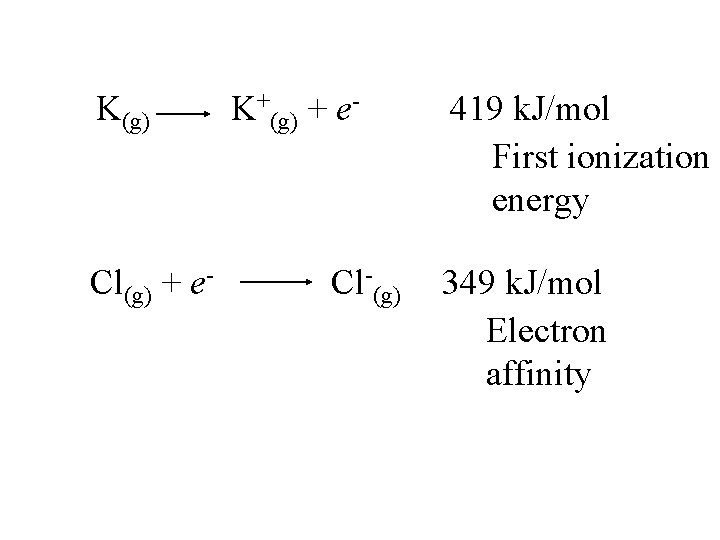
K(g) K+(g) + e- 419 k. J/mol

K(g) K+(g) + e- 419 k. J/mol First ionization energy

K(g) Cl(g) + e- K+(g) + e- Cl-(g) 419 k. J/mol First ionization energy 349 k. J/mol

K(g) Cl(g) + e- K+(g) + e- Cl-(g) 419 k. J/mol First ionization energy 349 k. J/mol Electron affinity

K(s) K(g) Hsublimation

K(s) ½ Cl 2(g) K(g) Hsublimation Cl(g) Hdissociation

K(s) ½ Cl 2(g) K(g) Cl(g) + e- K(g) Hsublimation Cl(g) Hdissociation K+(g) + e. Cl-(g) 419 k. J/mol 349 k. J/mol

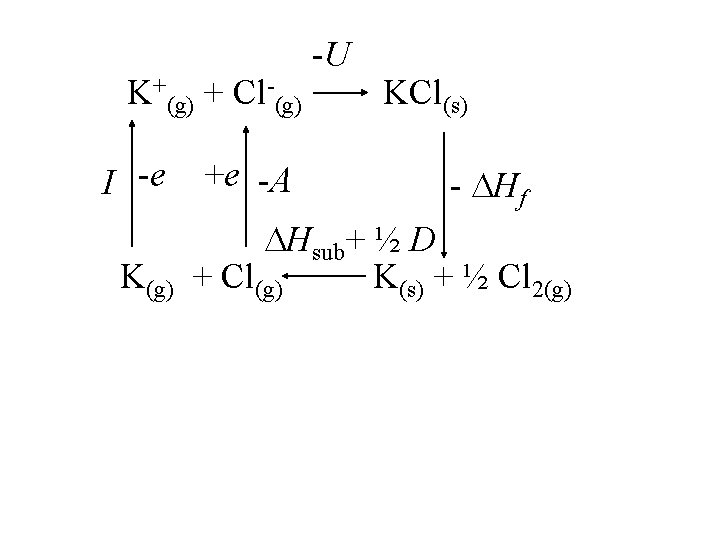
K(s) K(g) Cl(g) Hdissociation ½ Cl 2(g) K(g) Hsublimation K+(g) + e- Cl(g) + e. K+(g) + Cl-(g) 419 k. J/mol Cl-(g) 349 k. J/mol KCl(s) U

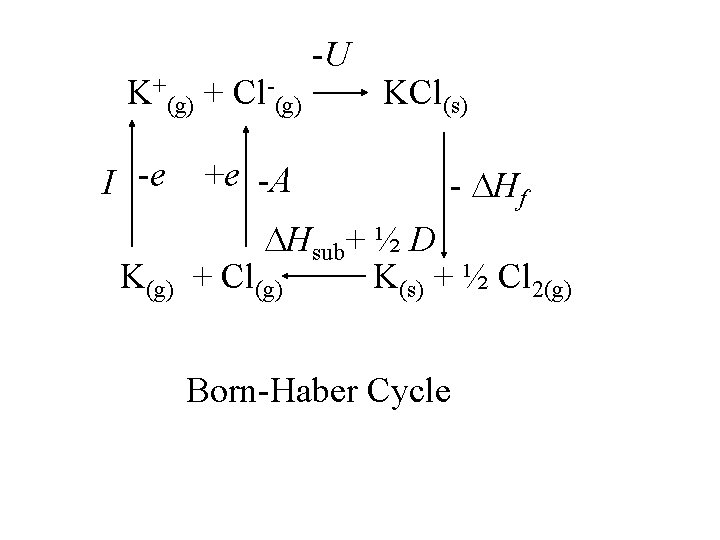
K+(g) + Cl-(g) I -e K(g) +e -A -U KCl(s) - Hf Hsub+ ½ D + Cl(g) K(s) + ½ Cl 2(g)

K+(g) + Cl-(g) I -e K(g) +e -A -U KCl(s) - Hf Hsub+ ½ D + Cl(g) K(s) + ½ Cl 2(g) Born-Haber Cycle

-U Only term not from K+(g) + Cl-(g) KCl(s) experiment I -e K(g) +e -A - Hf Hsub+ ½ D + Cl(g) K(s) + ½ Cl 2(g) Born-Haber Cycle

-U Only term not from K+(g) + Cl-(g) KCl(s) experiment I -e K(g) +e -A - Hf Hsub+ ½ D + Cl(g) K(s) + ½ Cl 2(g) Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A
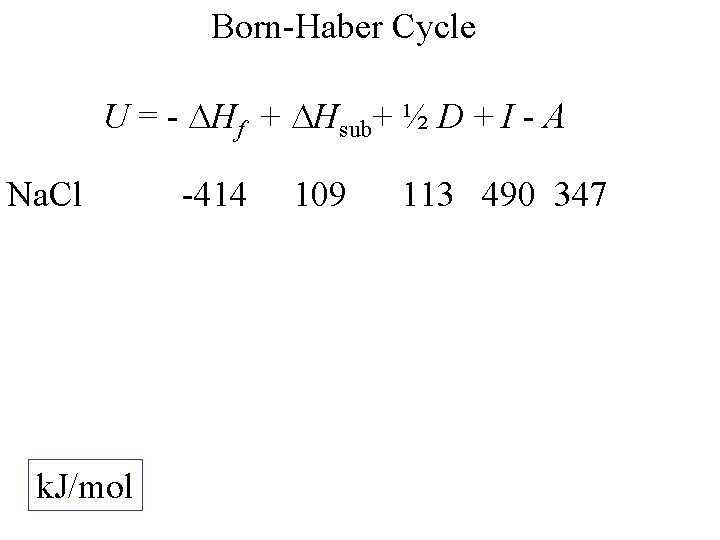
Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A Na. Cl k. J/mol -414 109 113 490 347
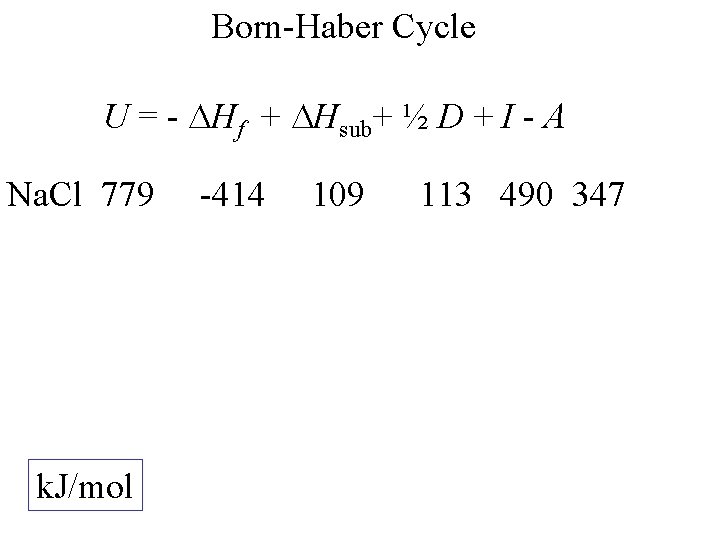
Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A Na. Cl 779 k. J/mol -414 109 113 490 347
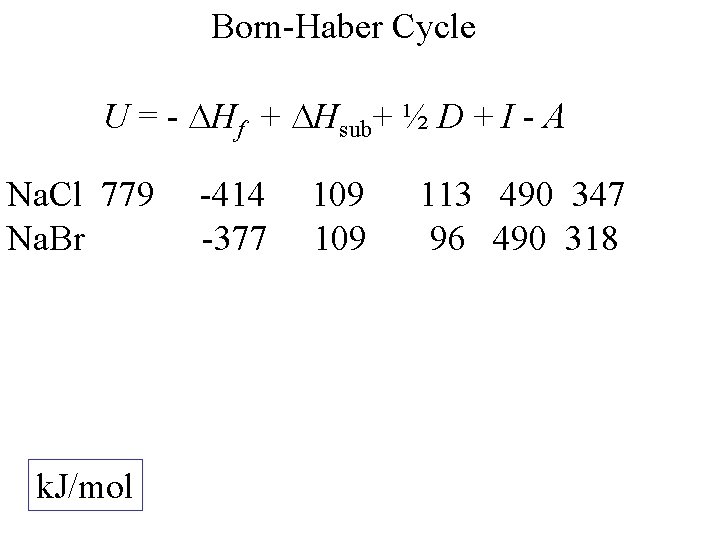
Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A Na. Cl 779 Na. Br k. J/mol -414 -377 109 113 490 347 96 490 318
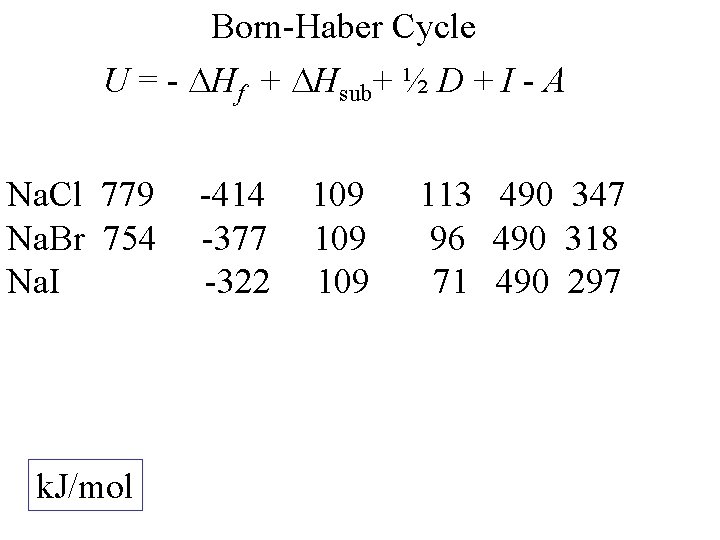
Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A Na. Cl 779 Na. Br 754 Na. I k. J/mol -414 -377 -322 109 109 113 490 347 96 490 318 71 490 297
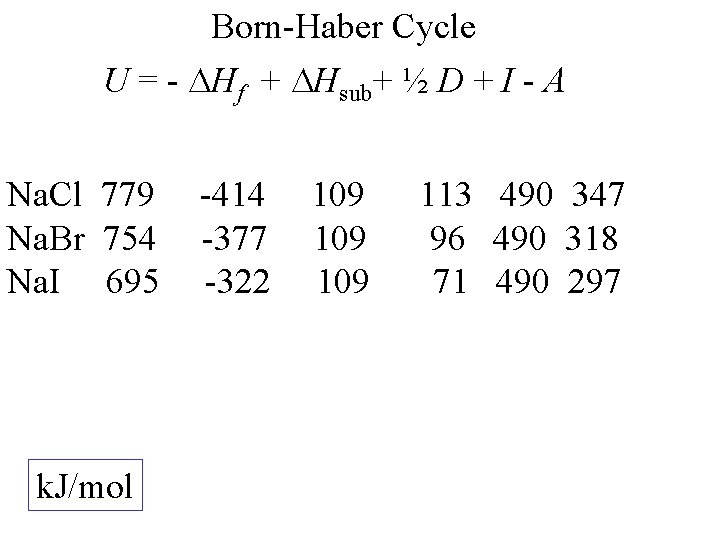
Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A Na. Cl 779 Na. Br 754 Na. I 695 k. J/mol -414 -377 -322 109 109 113 490 347 96 490 318 71 490 297
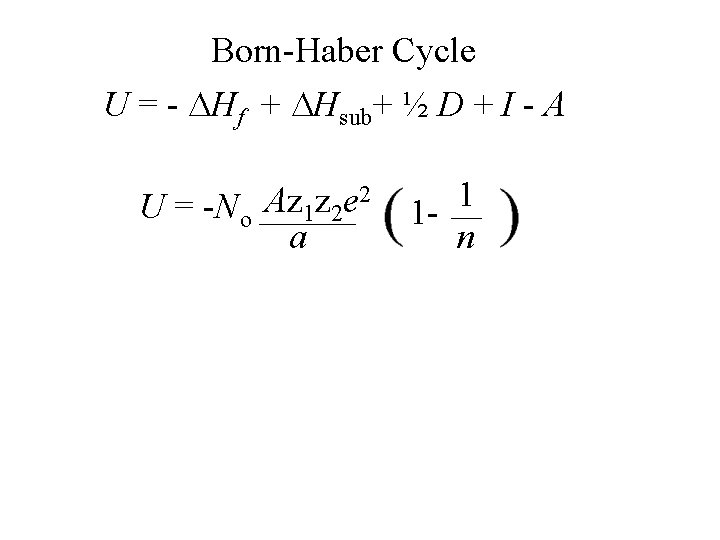
Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A 2 Az z e U = -No 1 2 a 1 1 n
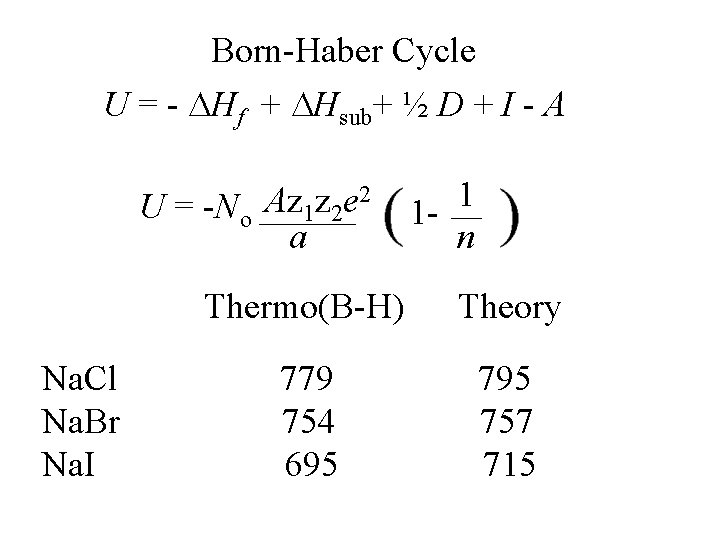
Born-Haber Cycle U = - Hf + Hsub+ ½ D + I - A 2 Az z e U = -No 1 2 a Na. Cl Na. Br Na. I 1 1 n Thermo(B-H) Theory 779 754 695 757 715

Homework problems for 10/3 1. Construct a diagram for the Born-Haber cycle for the various thermodynamic properties associated with the formation of magnesium chloride. continued

The important values are: Hsub Mg 147. 7 k. J/mol IE 1 Mg 737. 7 k. J/mol IE 2 Mg 1450. 7 k. J/mol D Cl 2 243 k. J/mol A Cl 348. 6 k. J/mol Hf Mg. Cl 2 -642 k. J/mol continued

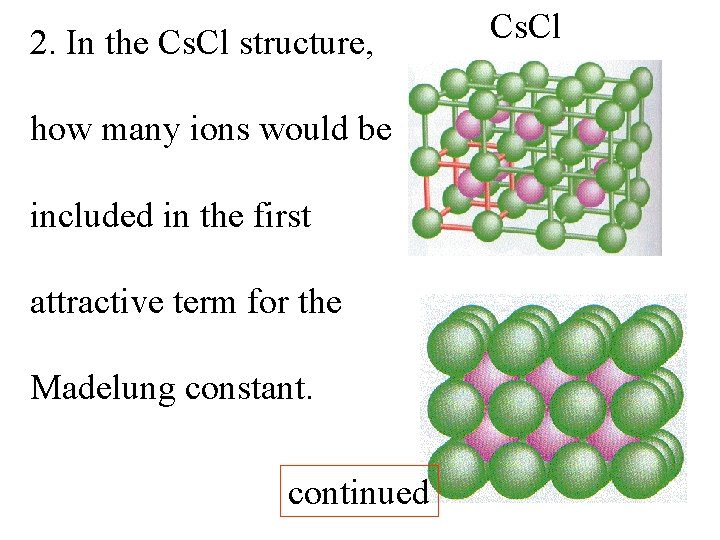
2. In the Cs. Cl structure, how many ions would be included in the first attractive term for the Madelung constant. continued Cs. Cl

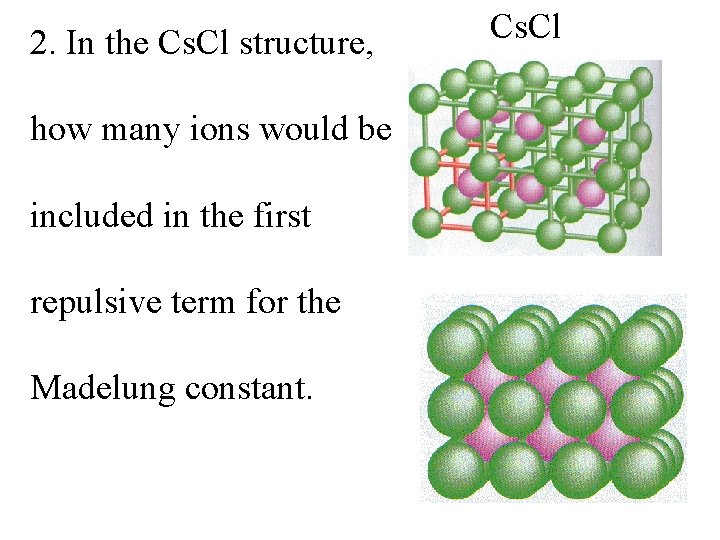
2. In the Cs. Cl structure, how many ions would be included in the first repulsive term for the Madelung constant. Cs. Cl