LAB ORATORY DIAGNOSIS OF PARASITIC DISEASES DEPARTEMENT OF

LAB ORATORY DIAGNOSIS OF PARASITIC DISEASES DEPARTEMENT OF PARASITOLOGY FACULTY OF MEDICINE UNIVERSITAS PADJADJARAN

AN INTRODUCTION DIAGNOSIS FOR PARASITIC INFECTION w Anamnesa – ( The history should include details of the presenting complaint ) w Physical examination w Laboratory examination espectially Parasitological diagnosis w Imunodiagnosis
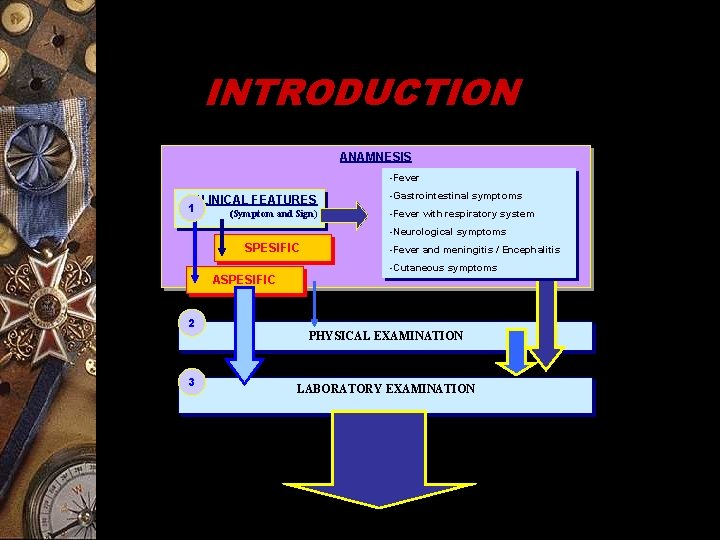
INTRODUCTION ANAMNESIS -Fever CLINICAL FEATURES 1 (Symptom and Sign) -Gastrointestinal symptoms -Fever with respiratory system -Neurological symptoms SPESIFIC ASPESIFIC 2 3 -Fever and meningitis / Encephalitis -Cutaneous symptoms PHYSICAL EXAMINATION LABORATORY EXAMINATION

INTRODUCTION 3 LABORATORY EXAMINATION DEPEND ON : v. Habitat Parasite v. Parasite distribution TYPE OF SPECIMEN : 1. Stool 2. Blood IMPORTANT 3 Urine 4 Sputum 5. Vaginal discharge, urethral discharge 6. Skin scrapings 7. Liquor Cerebro Spinal 8. Tissue biopsy 9. Nasal discharge 10. Corneal scrapings • Very important for examination Helminth parasite and Protozoa parasite • Repeating of Laboratory examination occasionally be needed *Should be understood about examination technique, morphology of parasite and life cycle of parasite 4 IMUNODIAGNOSIS

INTRODUCTION FECAL SPECIMENS Ø Ø Immediatelly have to examine : § Liquid specimens should be examined within 30 minute of passage § Soft (semiformed) specimens 1 hour § Formed specimens 24 hour after passage If this time is not possible, the specimen should be placed in one of the available fixatives : § Ø Formalin 10%, MIF (Merthiolate Iodine Formalin), PVA (Polyvinyl Alcohol) Generally Helminthic eggs more endure without preservation than intestinal protozoa

INTRODUCTION FECAL SPECIMENS Ø The specimens should not be contaminated with § Water – because water may contain freeliving organisms that can be mistaken for human parasites § Urine – may destroy motile organisms Ø Prior to examination , fecal specimens should never be incubated or frozen. Ø A chatartic with an oil base should not be used, and a stool softener (taken either orally or as a suppository) is usually inadequate for obtaining a purged specimen.

INTRODUCTION FECAL SPECIMENS Ø Repeating fecal examination after therapy : § Ascariasis, 2 -3 weeks after therapy § Protozoa infection, 3 -4 weeks after therapy § Taeniasis, 5 -6 weeks after therapy

EXAMINATION OF HELMINTH PARASITE SPECIMENS ( most important ) Ø Ø FECAL SPECIMENS BLOOD AND TISSUE SPECIMENS &

EXAMINATION OF HELMINTH PARASITE FECAL SPECIMENS ? Ø Ø For examination of helminth egg Most important for examination intestinal nematode including “Soil Transmitted Helminths” : § Ascaris lumbricoides § Trichuris trichiura § Hook worm : - Necator americanus and Ancylostoma duodenale § Strongyloides stercoralis

QUALITATIVE EXAMINATION OF HELMINTH PARASITE FECAL SPECIMENS DIRECT WET SMEAR ANOTHER QUALITATIVE EXAMINATION AND QUANTITATIVE EXAMINATION TO BE STUDIED IN FECAL EXAMINATION

LABORATORY TECHNIQUE FOR EXAMINATION OF HELMINTH PARASITE
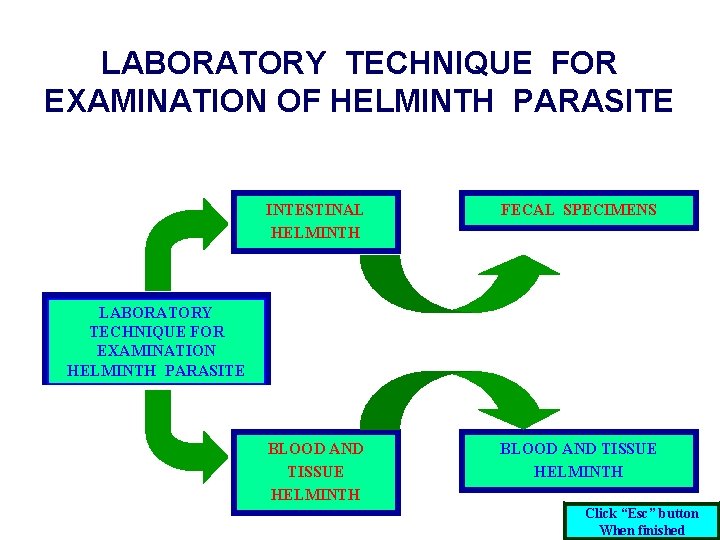
LABORATORY TECHNIQUE FOR EXAMINATION OF HELMINTH PARASITE INTESTINAL HELMINTH FECAL SPECIMENS BLOOD AND TISSUE HELMINTH LABORATORY TECHNIQUE FOR EXAMINATION HELMINTH PARASITE Click “Esc” button When finished
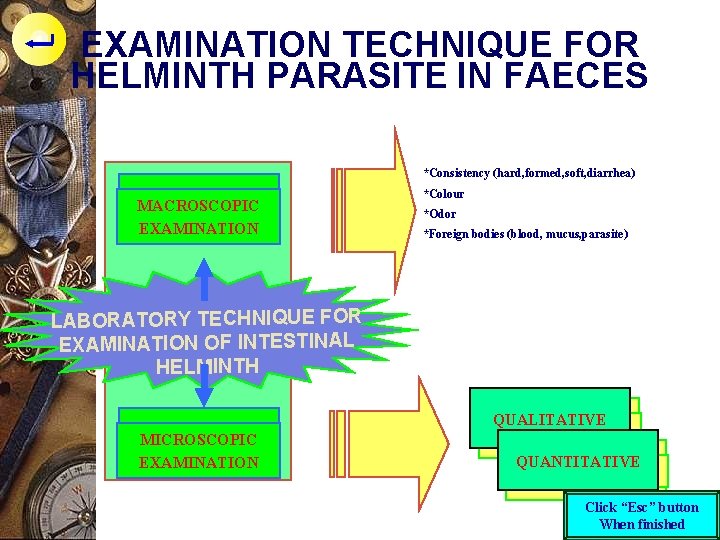
EXAMINATION TECHNIQUE FOR HELMINTH PARASITE IN FAECES *Consistency (hard, formed, soft, diarrhea) MACROSCOPIC EXAMINATION *Colour *Odor *Foreign bodies (blood, mucus, parasite) LABORATORY TECHNIQUE FOR EXAMINATION OF INTESTINAL HELMINTH QUALITATIVE MICROSCOPIC EXAMINATION QUANTITATIVE Click “Esc” button When finished
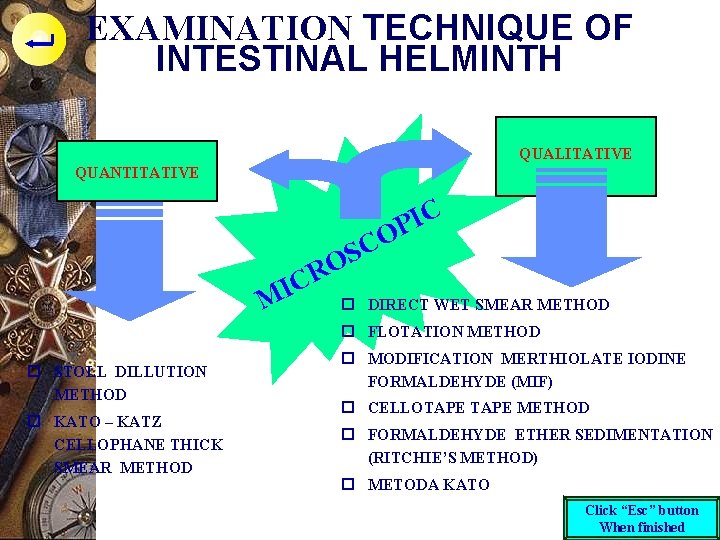
EXAMINATION TECHNIQUE OF INTESTINAL HELMINTH QUALITATIVE QUANTITATIVE C I M S O R C I P O C p DIRECT WET SMEAR METHOD p FLOTATION METHOD p STOLL DILLUTION METHOD p KATO – KATZ CELLOPHANE THICK SMEAR METHOD p MODIFICATION MERTHIOLATE IODINE FORMALDEHYDE (MIF) p CELLOTAPE METHOD p FORMALDEHYDE ETHER SEDIMENTATION (RITCHIE’S METHOD) p METODA KATO Click “Esc” button When finished

MICROSCOPIC QUALITATIVE DIRECT WET SMEAR METHOD p. Fast p. Severe infection p. Reagens - Na. Cl Physiologis (0, 9%), or - Eosin 2% Click “Esc” button When finished
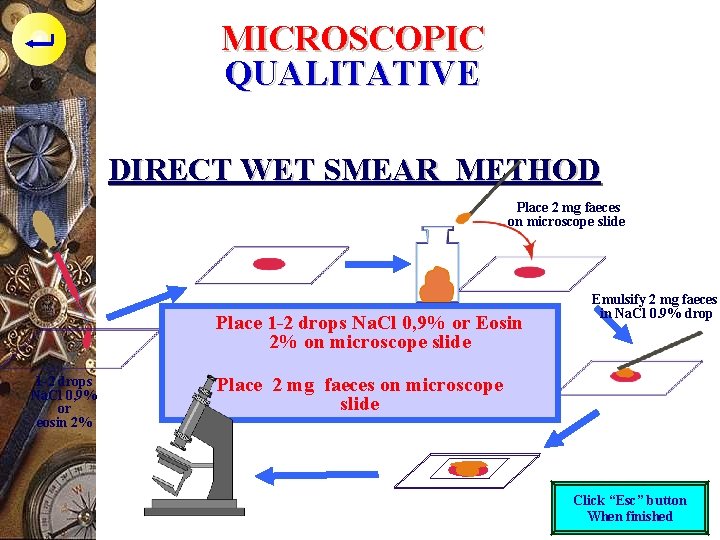
MICROSCOPIC QUALITATIVE DIRECT WET SMEAR METHOD Place 2 mg faeces on microscope slide Place 1 -2 drops Na. Cl 0, 9% or Eosin 2% on microscope slide 1 -2 drops Na. Cl 0, 9% or eosin 2% Emulsify 2 mg faeces in Na. Cl 0. 9% drop Place 2 mg faeces on microscope slide Click “Esc” button When finished
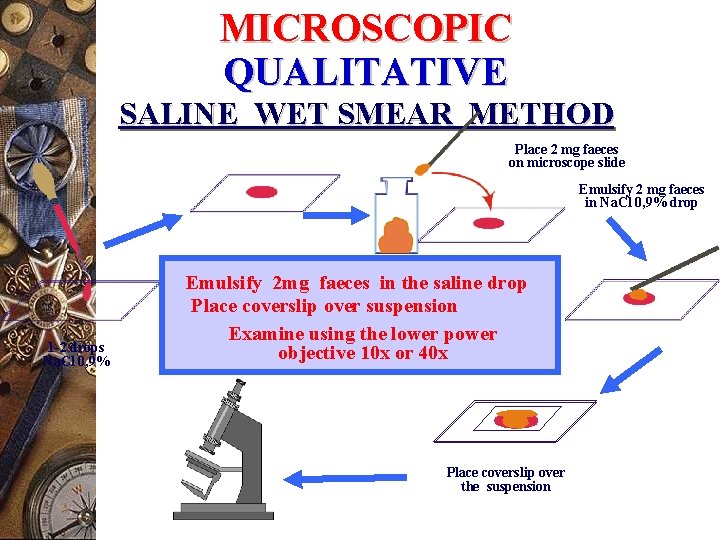
MICROSCOPIC QUALITATIVE SALINE WET SMEAR METHOD Place 2 mg faeces on microscope slide Emulsify 2 mg faeces in Na. Cl 0, 9% drop 1 -2 drops Na. Cl 0, 9% Emulsify 2 mg faeces in the saline drop Place coverslip over suspension Examine using the lower power objective 10 x or 40 x Place coverslip over the suspension

MICROSCOPIC QUALITATIVE FLOATATION METHOD p. Berdasarkan BJ telur < BJ larutan WITHOUT CENTRIFUGATION p. Berguna FLOTATION METHOD untuk infeksi ringan WITH CENTRIFUGATION p. Larutan yang dipergunakan : - Na. Cl jenuh, atau - Gula jenuh Click “Esc” button When finished

MICROSCOPIC QUALITATIVE FLOATATION METHOD WITHOUT CENTRIFUGATION Ose Place coverslip over the solution stirring glass 20 minutes Na. Cl saturated faeces 10 gr. faeces + 200 cc Na. Cl saturated Take down 1 -2 drops of Surface layer with Ose and Place on microscope slide 10 gr faeces mixed with 200 ml Na. Cl saturated until homogenous solution Take down 1 -2 drops the surface layer with Ose Place coverslip over microcope slide Examine under low power objective
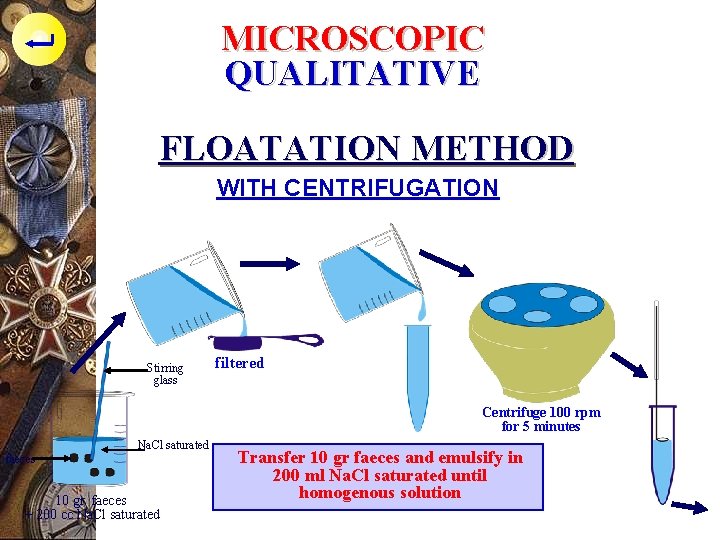
MICROSCOPIC QUALITATIVE FLOATATION METHOD WITH CENTRIFUGATION Stirring glass filtered Centrifuge 100 rpm for 5 minutes Na. Cl saturated faeces 10 gr. faeces + 200 cc Na. Cl saturated Transfer 10 gr faeces and emulsify in 200 ml Na. Cl saturated until homogenous solution
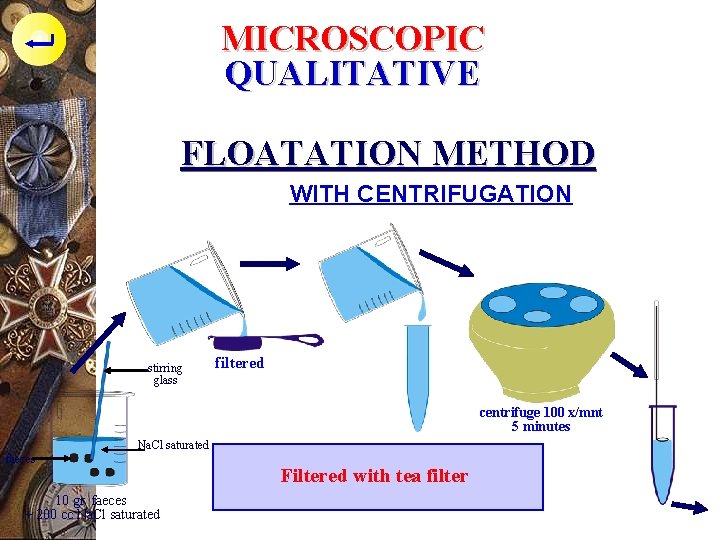
MICROSCOPIC QUALITATIVE FLOATATION METHOD WITH CENTRIFUGATION stirring glass filtered centrifuge 100 x/mnt 5 minutes Na. Cl saturated faeces 10 gr. faeces + 200 cc Na. Cl saturated Filtered with tea filter
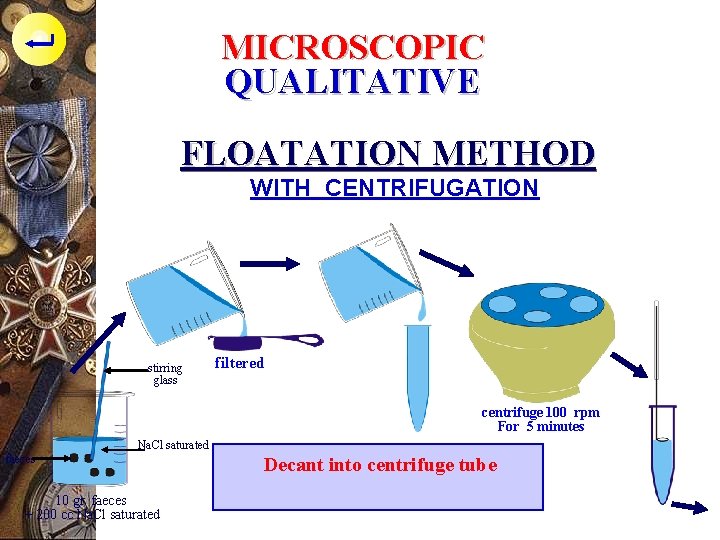
MICROSCOPIC QUALITATIVE FLOATATION METHOD WITH CENTRIFUGATION stirring glass filtered centrifuge 100 rpm For 5 minutes Na. Cl saturated faeces 10 gr. faeces + 200 cc Na. Cl saturated Decant into centrifuge tube

MICROSCOPIC QUALITATIVE FLOATATION METHOD WITH CENTRIFUGATION Stirring glass filtered centrifuge 100 rpm For 5 minutes Na. Cl saturated faeces 10 gr. faeces + 200 cc Na. Cl saturated Centrifuge 100 rpm for 5 minutes
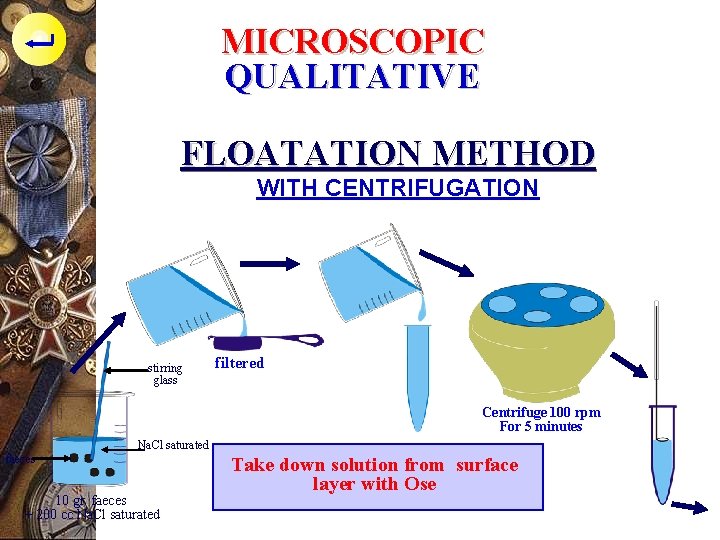
MICROSCOPIC QUALITATIVE FLOATATION METHOD WITH CENTRIFUGATION stirring glass filtered Centrifuge 100 rpm For 5 minutes Na. Cl saturated faeces 10 gr. faeces + 200 cc Na. Cl saturated Take down solution from surface layer with Ose

MICROSCOPIC QUALITATIVE FLOATATION METHOD WITH CENTRIFUGATION Place the solution on microscope slide Place coverslip over microscope slide Place solution on microscope slide and Place coverslip over the microscope slide. Examine under low power objective 10 x or 40 x.

MICROSCOPIC QUALITATIVE MERTIOLAT IODIN FORMALIN MODIFICATION (MIF MODIFICATION) p For identification eggs from intestinal helminth, Ameba and Giardia lamblia p Solution 1 : - 250 ml aquadest 200 ml thimerosal (1: 1. 000) 25 ml formalin 5 ml glycerin p Solution 2 : - Fresh lugol solution 5%

MICROSCOPIC QUALITATIVE MERTHIOLAT IODIN FORMALIN MODIFICATION (MIF MODIFICATION ) p. It’s good for staining and conservation of cyst of intestine protozoa and worm egg
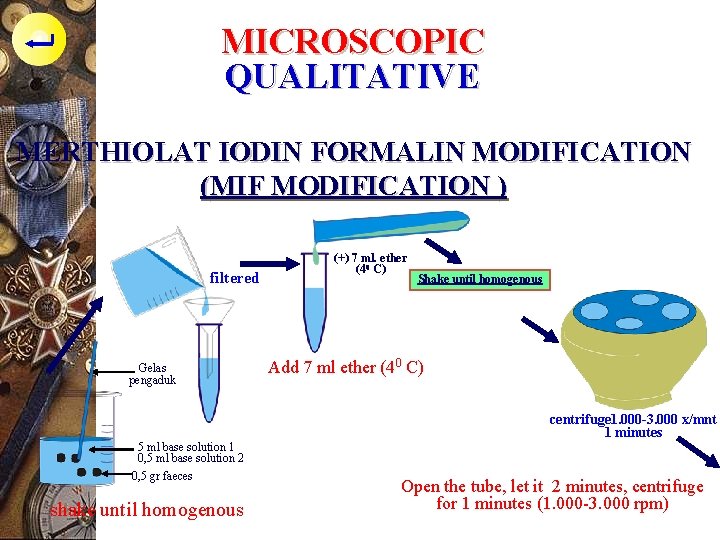
MICROSCOPIC QUALITATIVE MERTHIOLAT IODIN FORMALIN MODIFICATION (MIF MODIFICATION ) filtered Gelas pengaduk 5 ml base solution 1 0, 5 ml base solution 2 0, 5 gr faeces shake until homogenous (+) 7 ml. ether (40 C) Shake until homogenous Add 7 ml ether (40 C) centrifuge 1. 000 -3. 000 x/mnt 1 minutes Open the tube, let it 2 minutes, centrifuge for 1 minutes (1. 000 -3. 000 rpm)
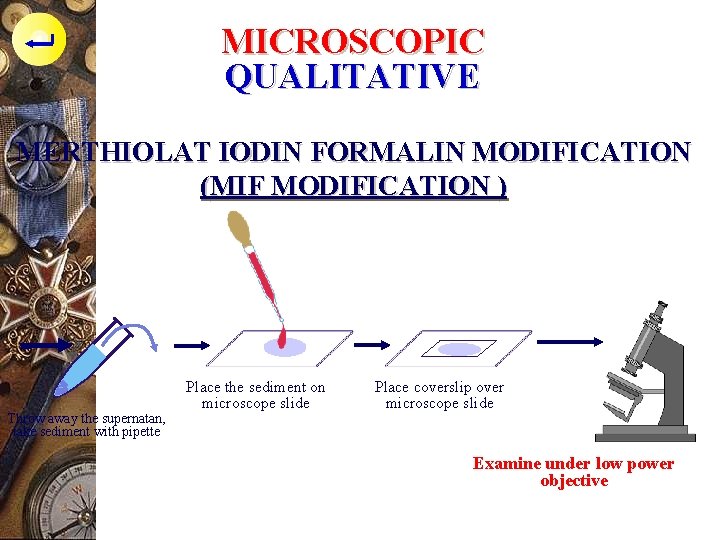
MICROSCOPIC QUALITATIVE MERTHIOLAT IODIN FORMALIN MODIFICATION (MIF MODIFICATION ) Throw away the supernatan, take sediment with pipette Place the sediment on microscope slide Place coverslip over microscope slide Examine under low power objective

MICROSCOPIC QUALITATIVE CELLOTAPE METHOD ý The egg adhere on perianal area, so rarely found in faeces (5 %). To find this parasite we need Scotch Adhesive tape Swab from Graham or Cellotape Method

MICROSCOPIC QUALITATIVE CELLOTAPE METHOD p To examine the egg of Enterobius vermicularis p Children 1 -10 years old p Doing in the morning before take a bath or wash the anus with water after defecating p Transparent plastic plaster (2 x 1, 5 cm)patched to skin around the anus p Press the plaster, then lift slowly p Patched to the object glass, examine under the microscope
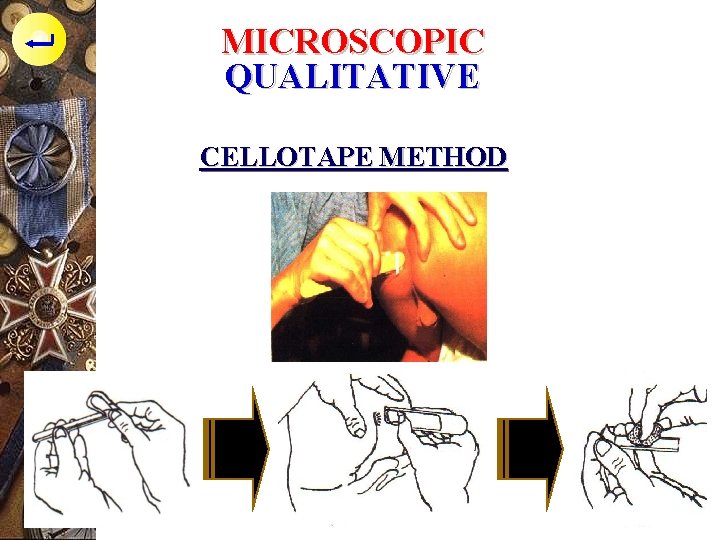
MICROSCOPIC QUALITATIVE CELLOTAPE METHOD

MICROSCOPIC QUALITATIVE Concentration Method p practical, simple, for ova examination in stool : - 1 gr faeces, put into the reaction tube, add aquadest, mixed until homogenous, put imyo the centrifuge tube - Centrifuge with velocity 3. 000 rpm for 1 mnt - Throw away the supernatan, take the sediment with pipette - Place the sediment on microscope slide, place coverslip on microscope slide
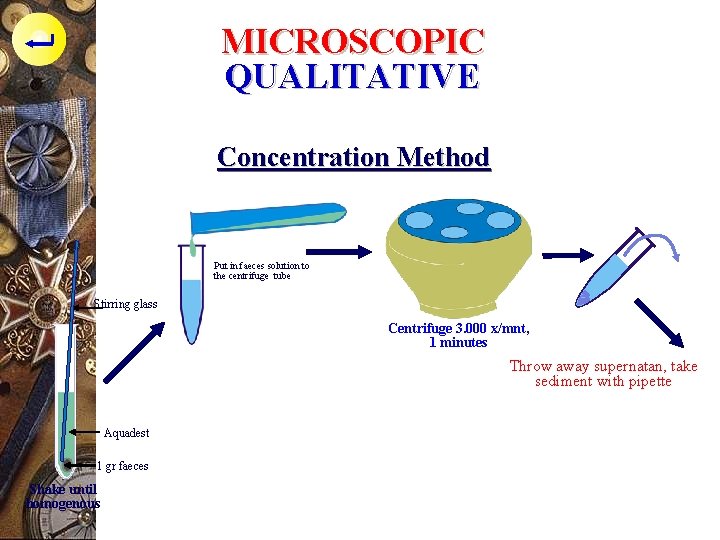
MICROSCOPIC QUALITATIVE Concentration Method Put in faeces solution to the centrifuge tube Stirring glass Centrifuge 3. 000 x/mnt, 1 minutes Throw away supernatan, take sediment with pipette Aquadest 1 gr faeces Shake until homogenous
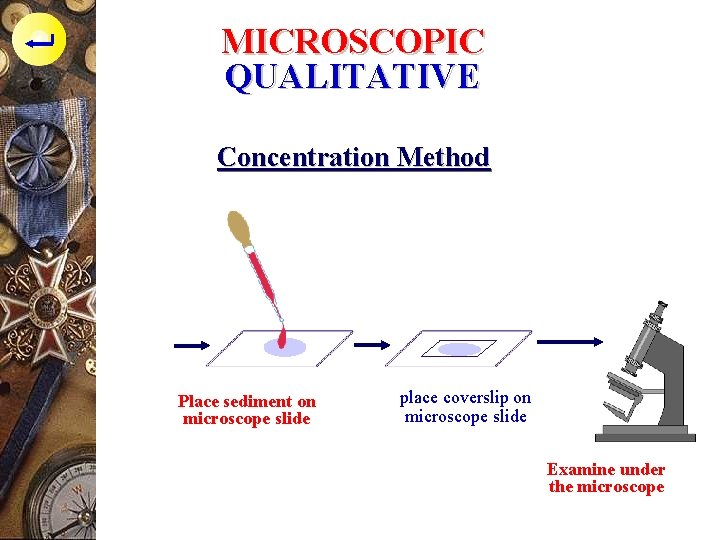
MICROSCOPIC QUALITATIVE Concentration Method Place sediment on microscope slide place coverslip on microscope slide Examine under the microscope

MICROSCOPIC QUALITATIVE CELLOPHANE THICK SMEAR METHOD (KATO METHOD) p Practical, simple, and cheap p Can be used in mass examination p Examination needs more stool, so the eggs can be found much more p Morphology of egg is clear

MICROSCOPIC QUALITATIVE CELLOPHANE THICK SMEAR METHOD (KATO METHOD) Substance : p Solution - 100 ml aquadest/fenol 6% - 100 ml glycerin (supaya kotoran tinja jadi jernih) - 1 ml malachit green solution (supaya mata tidak silau) p Cellophane tape, 2, 5 x 3 cm, soak in solution for >24 hours

MICROSCOPIC QUALITATIVE CELLOPHANE THICK SMEAR METHOD (KATO METHOD) p Technique : - Take 20 -50 mg faeces ( as large as red bean ) - Put on object glass, spread out - Cover with cellophane, pressing the faeces until flat and spread out under the cellophane - Drain the excessive fluid with filter paper - Let it 20 -30 minutes - Examine under the microscope
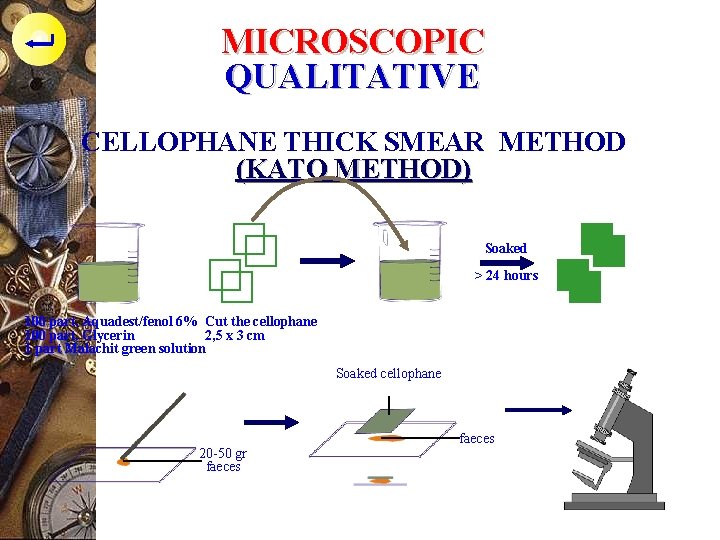
MICROSCOPIC QUALITATIVE CELLOPHANE THICK SMEAR METHOD (KATO METHOD) Soaked > 24 hours 100 part. Aquadest/fenol 6% Cut the cellophane 100 part. Glycerin 2, 5 x 3 cm 1 part Malachit green solution Soaked cellophane 20 -50 gr faeces

MICROSCOPIC QUANTITATIVE STOLL METHOD p Solution used : - Na. OH 0, 1 N (faeces solvent) or - KOH 10% p Good for severe and moderate infection p Less good for mild infection
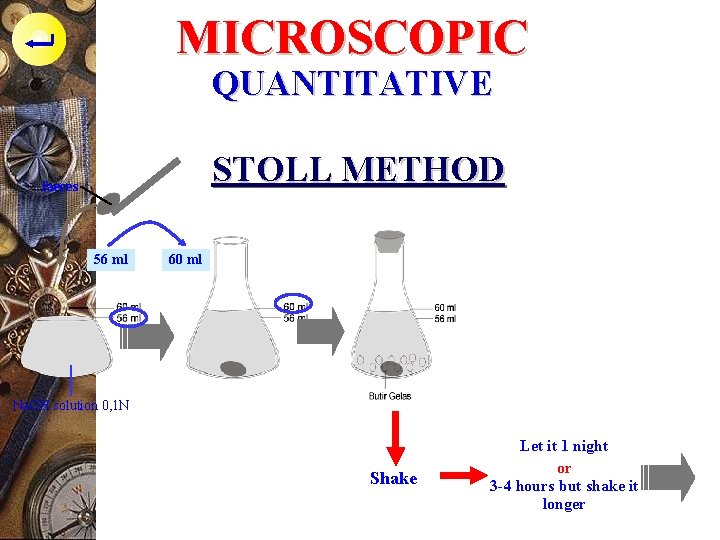
MICROSCOPIC QUANTITATIVE STOLL METHOD faeces 56 ml 60 ml Na. OH solution 0, 1 N Shake Let it 1 night or 3 -4 hours but shake it longer

MICROSCOPIC QUANTITATIVE STOLL METHOD Shake and take 0, 15 ml

MICROSCOPIC QUANTITATIVE STOLL METHOD p Formula : amount of egg in 1 gram feces = amount of seen egg (miroscopic) x 100

MICROSCOPIC QUANTITATIVE STOLL METHOD ENUMARATION p Na. OH = 56 ml, feces 4 ml ~ 4 gr p 4 gr feces in 60 ml p or 1 gr feces in 15 ml p In the 0. 15 ml of stool solution, can be found y eggs p So, in the 15 ml, is found y x 100 eggs(~ 1 gr stool)

MICROSCOPIC QUANTITATIVE STOLL METHOD Infection level based on amount of egg in each gram feces and amount of helminth Infectid by INFECTION LEVEL AMOUNT OF EGG PER-GRAM FAECES AMOUNT OF HELMINTH A. lumbricoides p p p MILD MODERATE SEVERE Ù < 7. 000 Ù 7. 000 -35. 000 Ù > 35. 000 Ù 5 or less Ù 6 -25 Ù > 25 A. duodenale p p p MILD MODERATE SEVERE Ù < 3. 000 Ù 3. 000 -10. 000 Ù > 10. 000 Ù 20 or less Ù 21 -100 Ù > 100 p p p MILD MODERATE SEVERE Ù < 2. 000 Ù 2. 000 -7. 000 Ù > 7. 000 Ù 50 or less Ù 51 -200 Ù > 200 N. americanus SOURCE : PARASITIC DISEASES PROGRAME. WHO. GENEVA, 1981

MICROSCOPIC QUANTITATIVE KATO-KATZ METHOD p Tolls : - Object glass - cellotape, thick 40 m, size 3 x 3 cm - Thick carton, make hole with fixed volume to print faeces as weight as 30 mg - Palm leaf rib, oily papper, wire netting

MICROSCOPIC QUANTITATIVE KATO-KATZ METHOD p Malachite-Green solution : - 100 ml aquadest - 100 ml glycerin (in order to make the feces clear) - 1 ml malachite green solution p Soak the cellotape in solution for > 24 hours

MICROSCOPIC QUANTITATIVE KATO-KATZ METHOD FILTERING FAECES - Put 5 gr faeces on the oily papper, put wire netting on it then press PRINTING FAECES - Put the holed carton on the object glass, print the filtered faeces on holed carton MAKING PREPARAT - Lift the carton - Cover the faeces with soaked cellotape - Press, spread out - Let it 30 mnt - Examine under the microscope
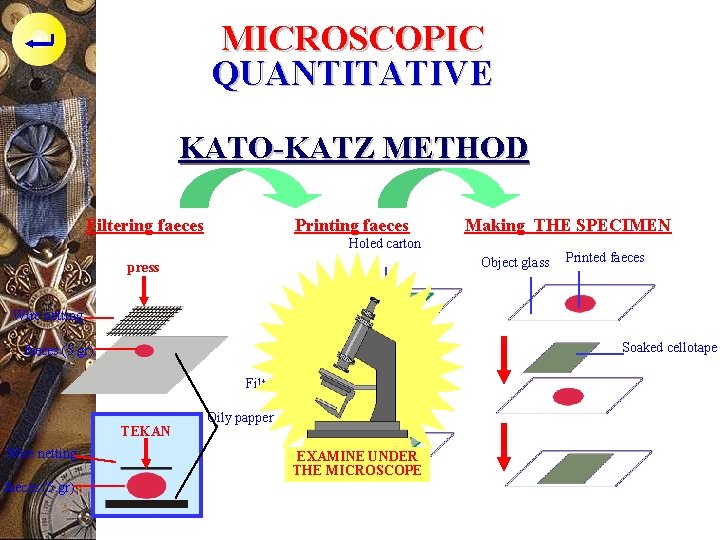
MICROSCOPIC QUANTITATIVE KATO-KATZ METHOD Filtering faeces Printing faeces Making THE SPECIMEN Holed carton Object glass press Printed faeces Wire netting Object glass faeces (5 gr) Soaked cellotape Filtered faeces TEKAN Wire netting faeces (5 gr) Oily papper printed EXAMINE UNDER THE MICROSCOPE

MICROSCOPIC QUANTITATIVE KATO-KATZ METHOD WHO (1981) p EGG PRODUCTION PER-DAY : û A. lumbricoides û A. doudenale û N. americanus : 200. 000 : 10. 000 -25. 000 : 5. 000 -10. 000 p WEIGHT OF FAECES PER-DAY û Children û Adult : 70 gram : 2 x children

MICROSCOPIC QUALITATIVE FORMOL ETHER SEDIMENTATION METHOD (RITCHIE) p (0, 5 ml faeces) + (1 -2 ml aquadest), shake, + (10 -12 ml aquadest), shake p Filter p Centrifuge 1. 000 rpm, 1 mnt p + (1 ml formalin 10%) + (formalin 10% sampai volume 8 ml), let it 10 mnt p + (3 ml ether), shake 15 -20 second p Centrifuge 2. 000 rpm, 1 -2 mnt p Throw away the supernatan, place the sediment on microscope slide, examine under the microscope

MICROSCOPIC QUALITATIVE FORMOL ETHER SEDIMENTATION METHOD (RITCHIE) 0, 5 ml. faeces 1 -2 ml. aquadest shake 10 -12 ml. aquadest shake filter centrifuge 1. 000 rpm, 1 minute p centrifuge 1. 000 rpm, 1 minute

MICROSCOPIC QUALITATIVE FORMOL ETHER SEDIMENTATION METHOD (RITCHIE) 1 ml. Formalin 10% + Formalin 10% until volume 8 ml LET IT (10 minutes) 3 ml. ether SHAKE (15 -20 seconds) CENTRIFUGE 2. 000 rpm, 1 -2 minutes + (1 ml formalin 10%) + (formalin 10% until volume 8 ml), let it 10 minutes Throw away the supernatan, place the sediment on microscope slide, examine under the microscope

CONSERVATION AND STORAGE METHOD EGG AND ADULT WORM IN FAECES USING FORMALIN SOLUTION 3, 5 % OR 4% p Making formalin solution 3, 5 % Or 4% : - 1 part of formalin solution 35% or 40% mixed with 9 part of running water, then put into closed bottle p Put the faeces into the bottle and closed tightly p For organ that attacked by parasite, wash cleanly before put into the bottle

BLOOD AND TISSUE NEMATODE (Blood specimens) Laboratory tecknique for examination of Filaria sp. ¶ · DIRECT METHOD F Blood taken of from finger tip F Thin blood smear - Qualitative - Determine microfilariae in peripheral blood vessels - Not for species identification F Thick blood smear - Quantitative - Measured blood releasefrom finger tip with mikropipet (160 m), make thick blood smear. CONCENTRATION METHOD F Take blood vein F More sensitive than direct method Click “esc” button When finished

SUBJECT MATERIAL � HOW TO EXAMINE AND INTERPRET INTESTINAL PROTOZOA - Direct wet mount - Modified Merthiolate-Iodine -Formalin (MIF) � HOW TO EXAMINE AND INTERPRET BLOOD PROTOZOA - Thin Blood smear with Giemsa staining - Thick Blood smear with Giemsa staining � EXAMINATION FOR Trichomonas vaginalis � HOW TO PREPARE PERMANENT (FIXED) MOUNT � - Blood parasites - Trichomonas vaginalis - Ameba, using Heidenhein method and staining � METHODS FOR PREPARING COLOR STAININGS

LAB METHODS FOR INTESTINAL PROTOZOA (1). Direct wet mount exam Purpose : For quick and simple examination - For trophozoite form of ameba, use 2% eosin solution - For cyst and nucleus of amoeba use lugol solution (2% of Iodine + 3% potasssium Iodine)

LAB METHODS FOR INTESTINAL PROTOZOA (1). Direct wet mount examination Preparation : w Using a small stick, add small amount of feces on top of an object glass w Add few drops of physiological saline solution Na. Cl/lugol/eosin 2% on top w Distribute/spread evenly and cover with coverglass w Examine under low power microscope

LAB METHOD FOR INTESTINAL PROTOZOA (2). Modified Method Merthiolate-Iodine-Formaline (MIF) Purpose : Good for diagnosis of Ameba and Giardia in feces Click “Esc”button When finished

LAB METHOD FOR INTESTINAL PROTOZOA Modified Method Merthiolate-Iodine-Formaline (MIF) Ingredients used : Basic solution (1) : – 250 ml aquadest – 200 ml tincture of merthiolate – 25 ml formaldehyde Basic solution (2) : –lugol 5 % (not to be kept > 3 weeks) Both basic solutions should be kept in brown colored bottle Click “Esc”button When finished

CHARACTERISTIC OF FECES WITH AMEBA MACROSCOPIC w Acidic in character w Foul smelling w Produce less mucus compared to bacillary dysentery, less sticky w With or without blood (blood may be found in solid feces) w In some cases mucosal wall may come out

CHARACTERISTIC OF FECES WITH AMEBA MACROSCOPIC Feces of amebic dysenteric patient, plenty of trophozoite stage Source : A Colour Atlas of Clinical Parasitology. Tomio Yamaguchi. Alih Bahasa : Lesmana Padmasutra, dkk.
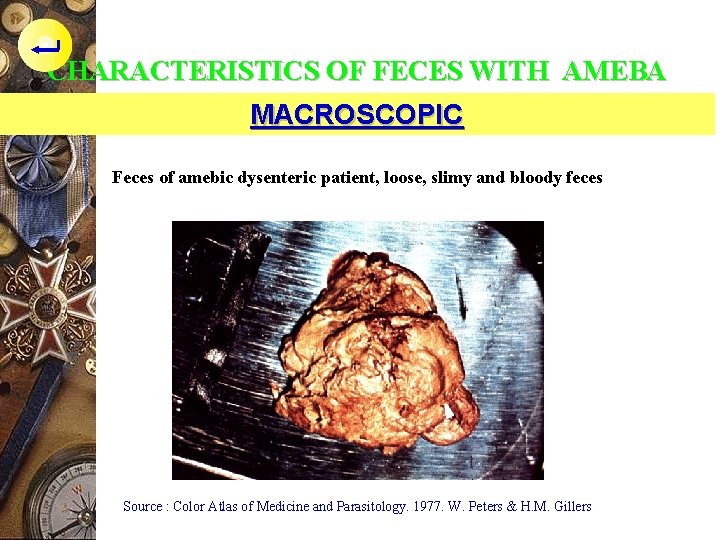
CHARACTERISTICS OF FECES WITH AMEBA MACROSCOPIC Feces of amebic dysenteric patient, loose, slimy and bloody feces Source : Color Atlas of Medicine and Parasitology. 1977. W. Peters & H. M. Gillers

CHARACTERISTICS OF FECES WITH AMEBA MICROSCOPIC w w Plenty of bacteria Entamoeba histolytica (+) containing erythrocytes Erythrocytes in reuleaux (chain) formation Charcot-Leyden crystals (unspecific for ameba dysentery, can be found also in other parasitic infection e. g. Strongyloides stercoralis) – from eosinophil desintegration w Pus less abundant compared to bacillary dysentery, if no secondary infection w Macrophages in bacillary dysentery mimics Entamoeba histolytica but nucleus and pseudopods differs w Cyst found in carrier patients and cases with light infection

CHARACTERISTIC OF FECES WITH AMEBA microscopic Charcot-Leyden crystals (From eosinophil desintegration) Source; Color Atlas of Medical Parasitology. Prayong Radomyos, dkk.

EXAMINATION METHODS FOR BLOOD PROTOZOA Blood slides with Giemsa staining Purpose : For the examination of blood protozoa, e. g. : Plasmodium, Trypanosoma, Babesia etc. Prepared in two stages : (1). Prepare the blood smear (2). Do the color staining

EXAMINATION METHOD FOR BLOOD PROTOZOA Blood slides with Giemsa staining Preparation of thin blood film w Place blood slide in upright position, allow to dry, keep away from dust or small insects.
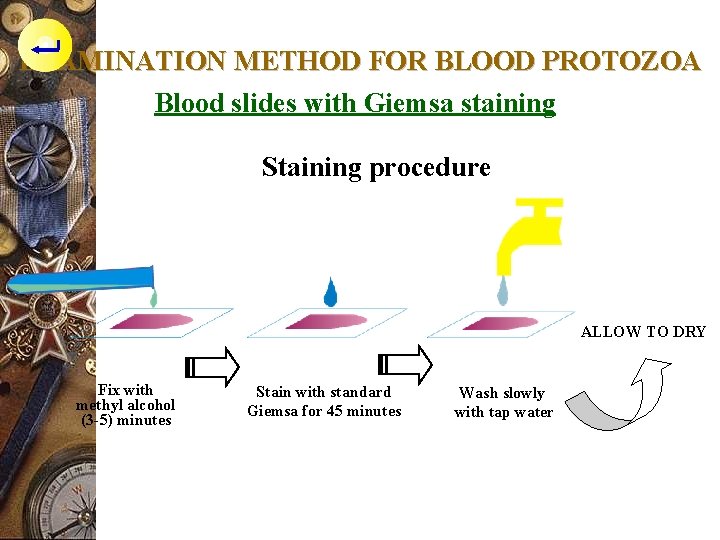
EXAMINATION METHOD FOR BLOOD PROTOZOA Blood slides with Giemsa staining Staining procedure ALLOW TO DRY Fix with methyl alcohol (3 -5) minutes Stain with standard Giemsa for 45 minutes Wash slowly with tap water

EXAMINATION METHOD FOR BLOOD PROTOZOA Thick blood smear with Giemsa stain Purpose : Rapid examination of blood protozoa (for mass survey and screening ) Conducted in two stages : (1). Prepare thick blood film (2). Staining the blood film
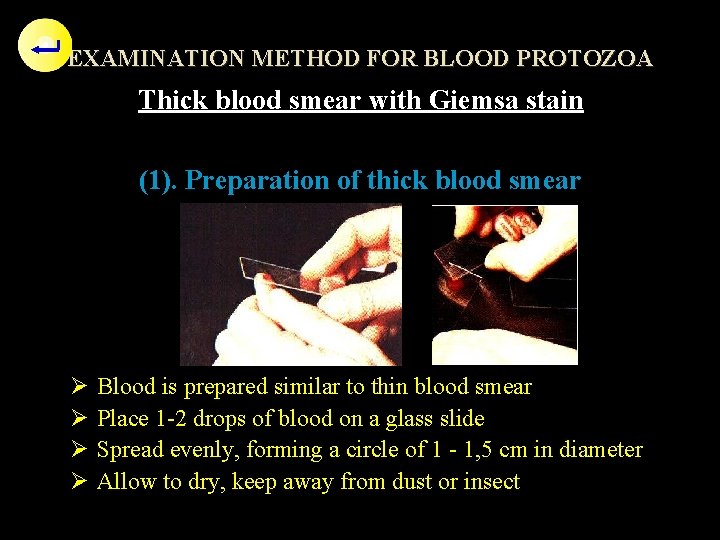
EXAMINATION METHOD FOR BLOOD PROTOZOA Thick blood smear with Giemsa stain (1). Preparation of thick blood smear Ø Ø Blood is prepared similar to thin blood smear Place 1 -2 drops of blood on a glass slide Spread evenly, forming a circle of 1 - 1, 5 cm in diameter Allow to dry, keep away from dust or insect
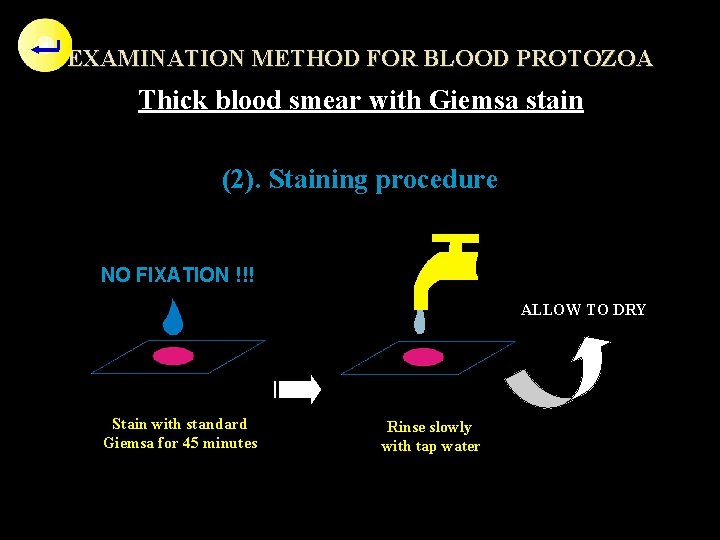
EXAMINATION METHOD FOR BLOOD PROTOZOA Thick blood smear with Giemsa stain (2). Staining procedure NO FIXATION !!! ALLOW TO DRY Stain with standard Giemsa for 45 minutes Rinse slowly with tap water

EXAMINATION METHOD FOR BLOOD PROTOZOA COMPARISON OF THIN AND THICK BLOOD SMEAR � THIN BLOOD SMEAR – – Morphology and stages of Plasmodium clearly defined Erythrocytes are intact (due to fixation) Slow to prepare Used for examination of moderate and heavy infection � THICK BLOOD SMEAR – Morphology and stages of Plasmodium not clearly defined – Erythrocytes are lysed, only stroma from erythrocytes are seen – Preparation and staining are faster (can be used for mass survey examination) – Commonly used for light infection

EXAMINATION OF Trichomonas Methods of examination : (1). Direct examination (2). Culture Direct method : (1). Direct wet mount (2). Staining vaginalis

EXAMINATION OF Trichomonas vaginalis Sample for examination : (1). Women : vaginal, or urethral discharge (using vaginal spiculum) (2). Men : discharge from urethra or prostate, and centrifugated urine sample

Examination for Trichomonas vaginalis DIRECT EXAMINATION Cotton bulb Glucose 5% Materials used : -Test tube containing 5% glucose (in physiological saline solution) - Cotton bulb

Terima kasih …………………. April 2005
- Slides: 76