Crystal Physics Prepared by S Sheik Fareed MOHAMED

Crystal Physics Prepared by S. Sheik Fareed MOHAMED SATHAK ENGINEERING COLLEGE 1

SOLIDS n Solids have “resistance” to changes in both shape and volume n Solids can be Crystalline or Amorphous n Crystals are solids that consist of a periodic array of atoms, ions, or molecules n If this periodicity is preserved over “large” (macroscopic) distances we call it “Long Range Order” n Amorphous solids do not have Long-Range Order n 2 Short Range Order
SOLIDS Crystals Solids: Short Range Order Long Range Order 3 n Amorphous solids: n Short Range Order n No Long-Range Order

Crystals The periodic array of atoms, ions, or molecules 4 that form the solids is called Crystal Structure = Space (Crystal) Lattice + Basis Space (Crystal) Lattice is a regular periodic arrangement of points in space, and is purely mathematical abstraction Crystal Structure is formed by “putting” the identical atoms (group of atoms) in the points of the space lattice This group of atoms is called Basis
Crystals Crystal Structure = Space Lattice + Basis 5
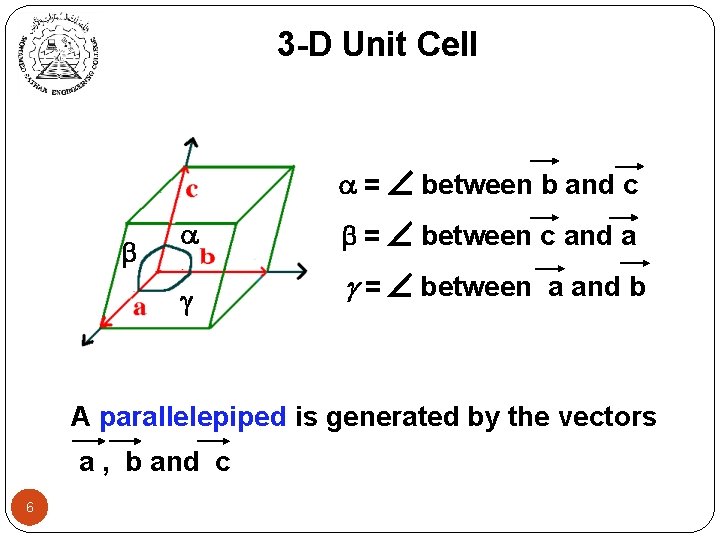
3 -D Unit Cell = between b and c = between c and a = between a and b A parallelepiped is generated by the vectors a , b and c 6

3 -D Unit Cell Different lattice systems are created by varying the lengths a, b and c and the angles , and . To examine the point symmetry, we look for: • rotation symmetry axes, • reflection planes and • inversion centre. SEVEN systems for 3 -D lattices. 7
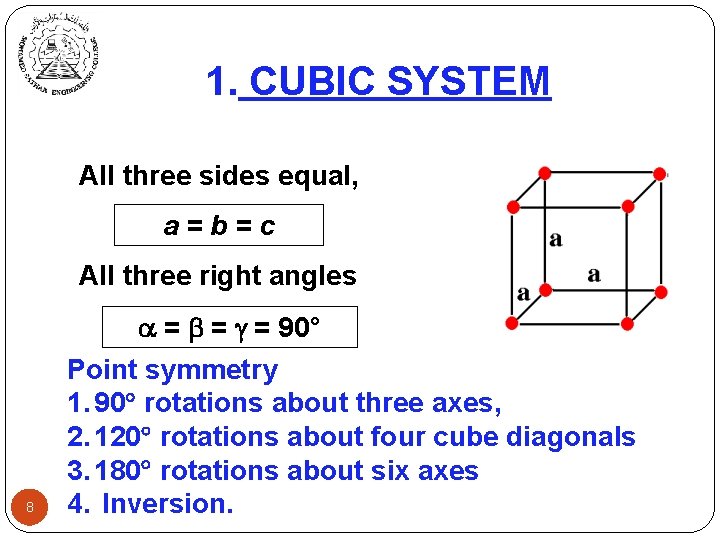
1. CUBIC SYSTEM All three sides equal, a=b=c All three right angles 8 = = = 90° Point symmetry 1. 90 rotations about three axes, 2. 120 rotations about four cube diagonals 3. 180 rotations about six axes 4. Inversion.
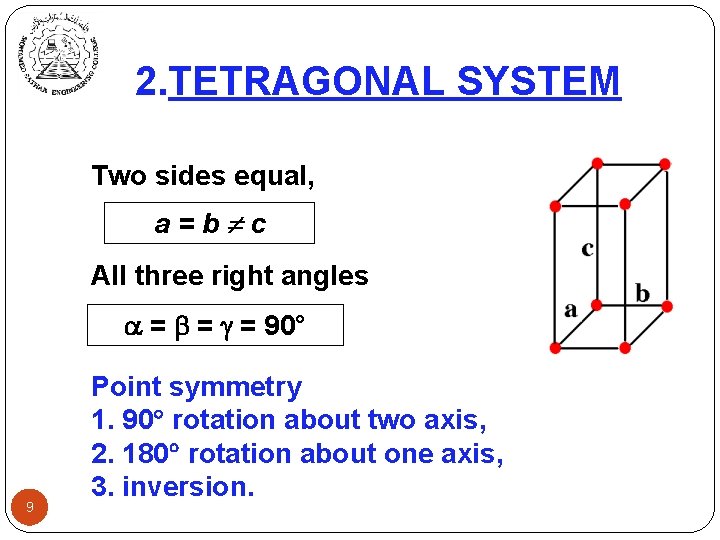
2. TETRAGONAL SYSTEM Two sides equal, a=b c All three right angles = = = 90° 9 Point symmetry 1. 90 rotation about two axis, 2. 180 rotation about one axis, 3. inversion.
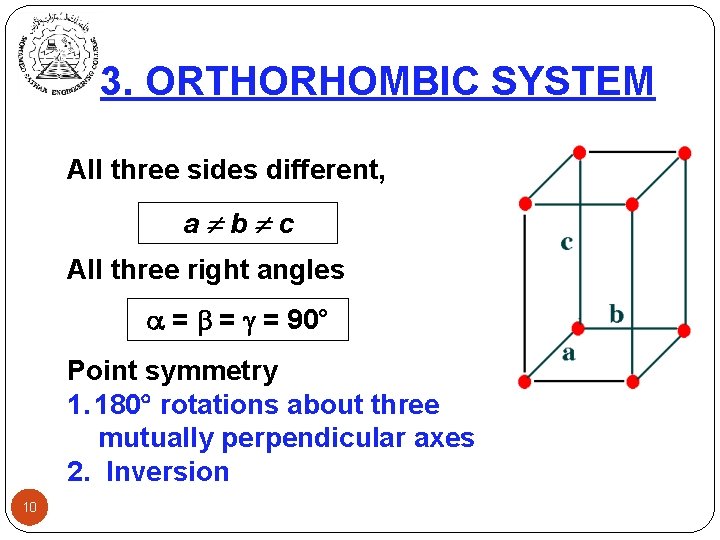
3. ORTHORHOMBIC SYSTEM All three sides different, a b c All three right angles = = = 90° Point symmetry 1. 180 rotations about three mutually perpendicular axes 2. Inversion 10
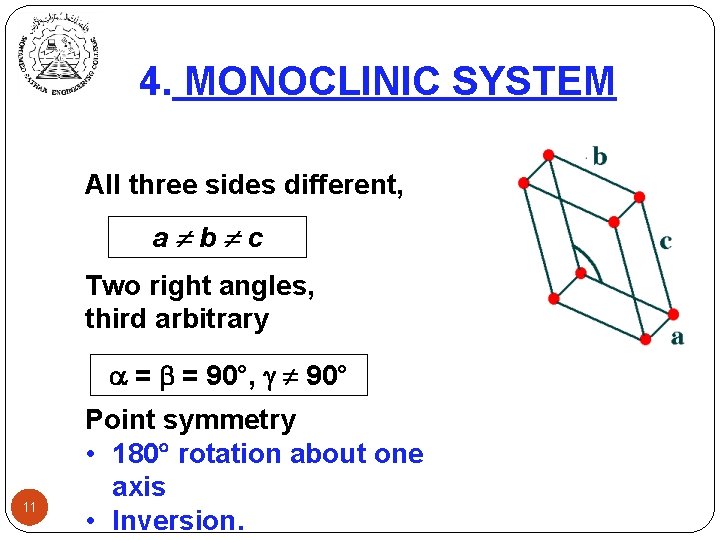
4. MONOCLINIC SYSTEM All three sides different, a b c Two right angles, third arbitrary = = 90°, 90° 11 Point symmetry • 180 rotation about one axis • Inversion.
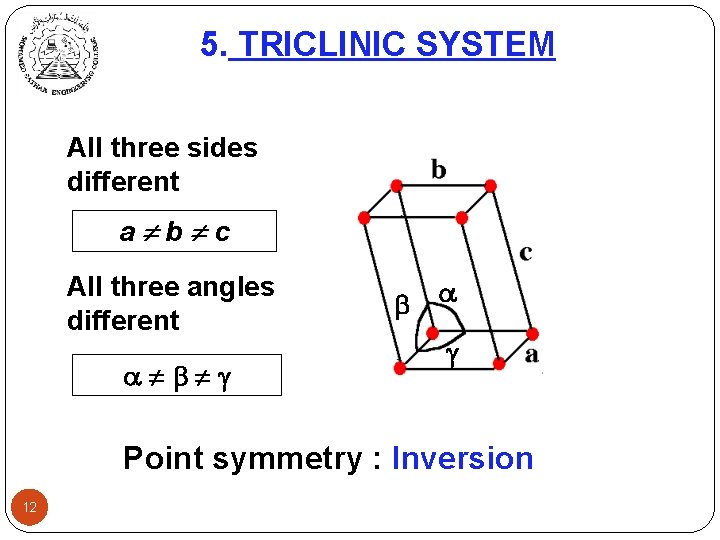
5. TRICLINIC SYSTEM All three sides different a b c All three angles different Point symmetry : Inversion 12
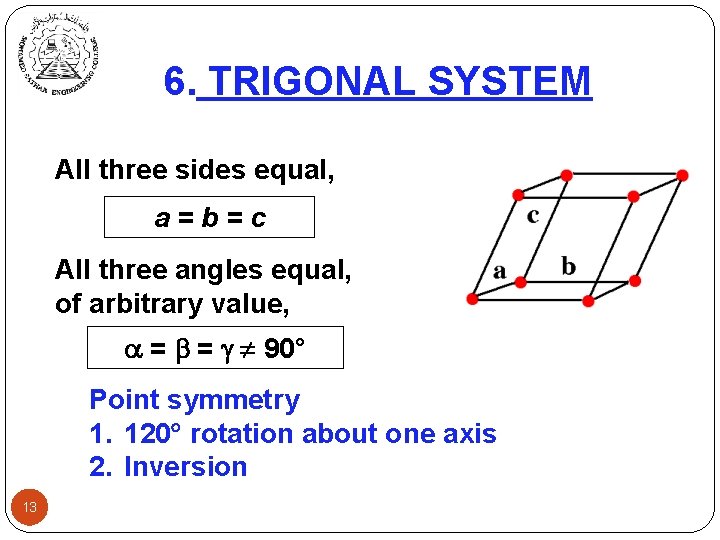
6. TRIGONAL SYSTEM All three sides equal, a=b=c All three angles equal, of arbitrary value, = = 90° Point symmetry 1. 120 rotation about one axis 2. Inversion 13
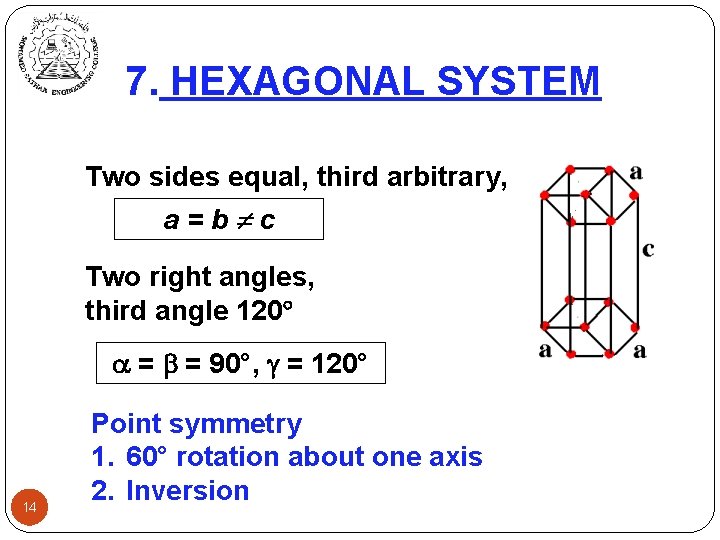
7. HEXAGONAL SYSTEM Two sides equal, third arbitrary, a=b c Two right angles, third angle 120 = = 90°, = 120° 14 Point symmetry 1. 60° rotation about one axis 2. Inversion

BRAVAIS LATTICES. A complete analysis based on mathematics and geometry has shown that a single lattice system can have at the most FOUR types. In all the seven lattice systems have a total of 14 types. 15 These are called the BRAVAIS LATTICES.
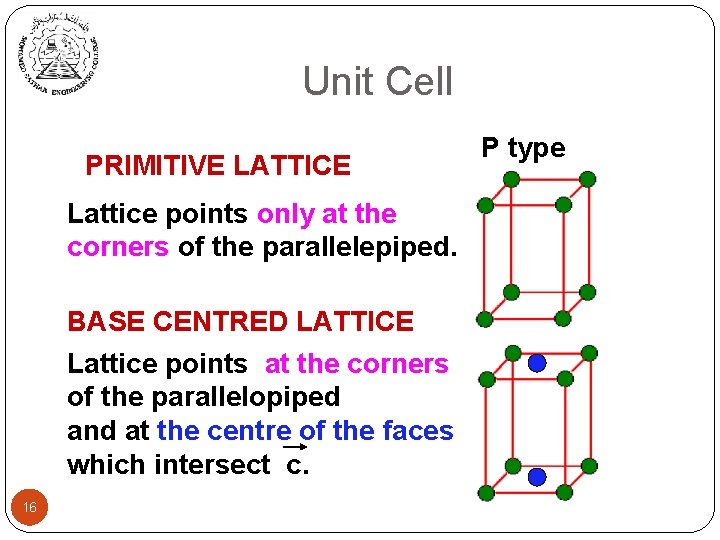
Unit Cell PRIMITIVE LATTICE Lattice points only at the corners of the parallelepiped. BASE CENTRED LATTICE Lattice points at the corners of the parallelopiped and at the centre of the faces which intersect c. 16 P type
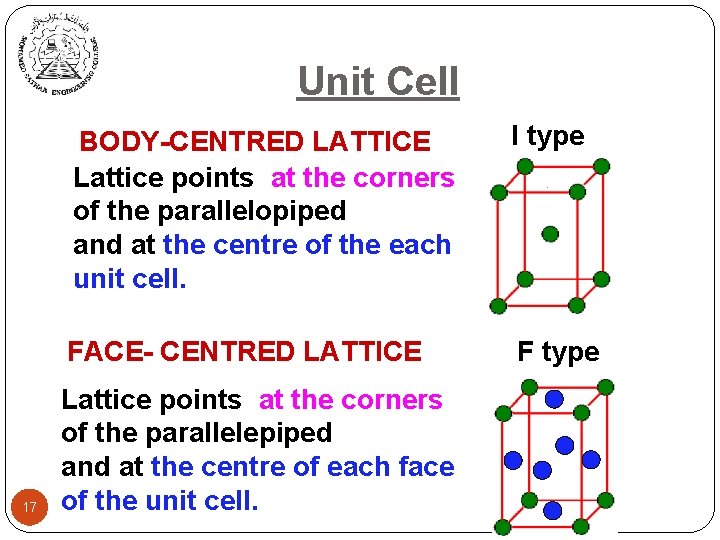
Unit Cell 17 BODY-CENTRED LATTICE Lattice points at the corners of the parallelopiped and at the centre of the each unit cell. I type FACE- CENTRED LATTICE F type Lattice points at the corners of the parallelepiped and at the centre of each face of the unit cell.

Seven systems divide into 14 Bravais lattices : 1. Triclinic P 2. Monoclinic P, C 3. Orthorhombic P, C, I, F 4. Trigonal 18 P 5. Hexagonal P 6. Tetragonal P, I 7. Cubic P, I, F

Seven systems divide into 14 Bravais lattices : Notation 19 P: Primitive (lattice points only at the corners of the unit cell) I: Body-centred (lattice points at the corners + one lattice point at the centre of the unit cell) F: Face-centred (lattice points at the corners + lattice points at centres of all faces of the unit cell) C: End-centred or base-centred (lattice points at the corners + two lattice points at the centres of a pair of opposite faces)
Crystal Planes 20
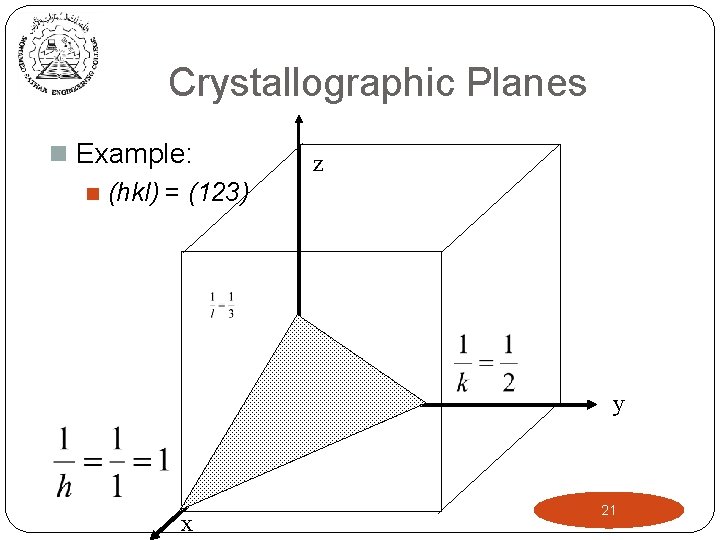
Crystallographic Planes n Example: n (hkl) = (123) z y x 21
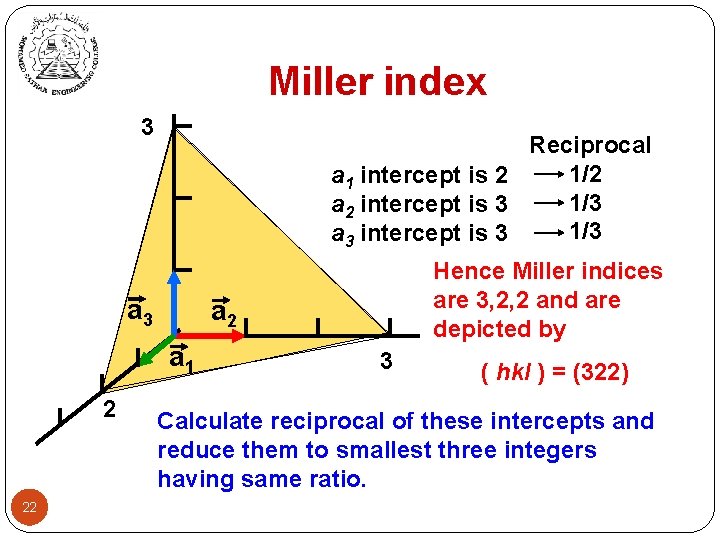
Miller index 3 Reciprocal 1/2 a 1 intercept is 2 1/3 a 2 intercept is 3 1/3 a 3 intercept is 3 a 2 a 1 2 22 Hence Miller indices are 3, 2, 2 and are depicted by 3 ( hkl ) = (322) Calculate reciprocal of these intercepts and reduce them to smallest three integers having same ratio.
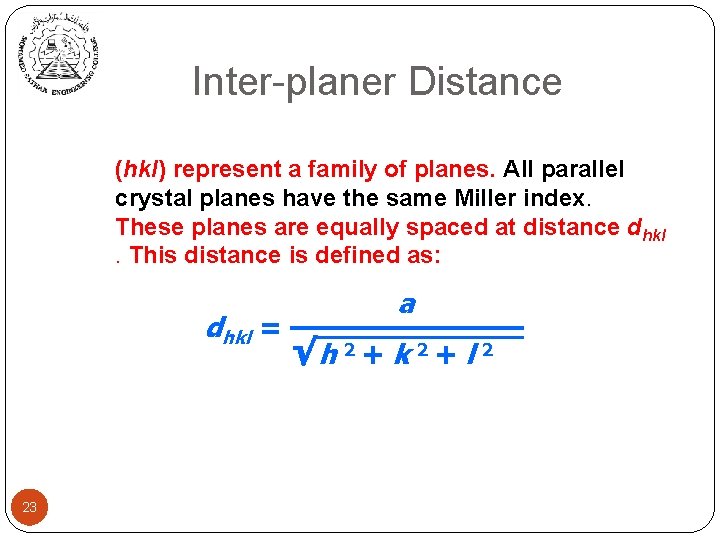
Inter-planer Distance (hkl) represent a family of planes. All parallel crystal planes have the same Miller index. These planes are equally spaced at distance dhkl. This distance is defined as: dhkl = 23 a √h 2 + k 2 + l 2
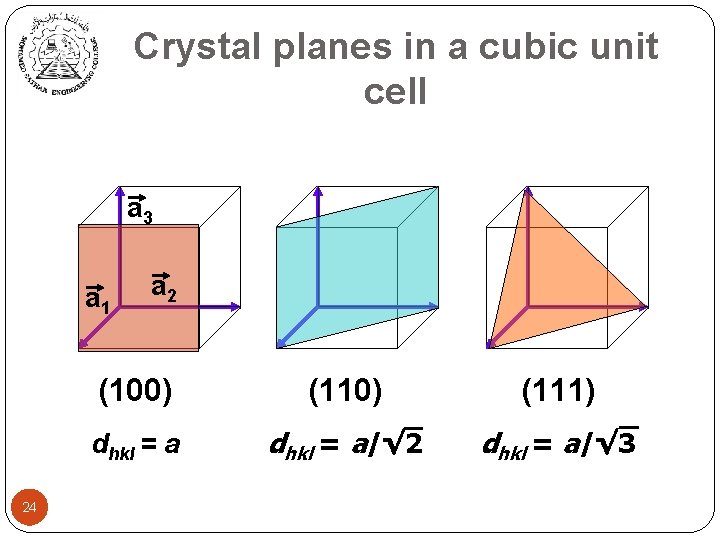
Crystal planes in a cubic unit cell a 3 a 1 24 a 2 (100) (111) dhkl = a/√ 2 dhkl = a/√ 3
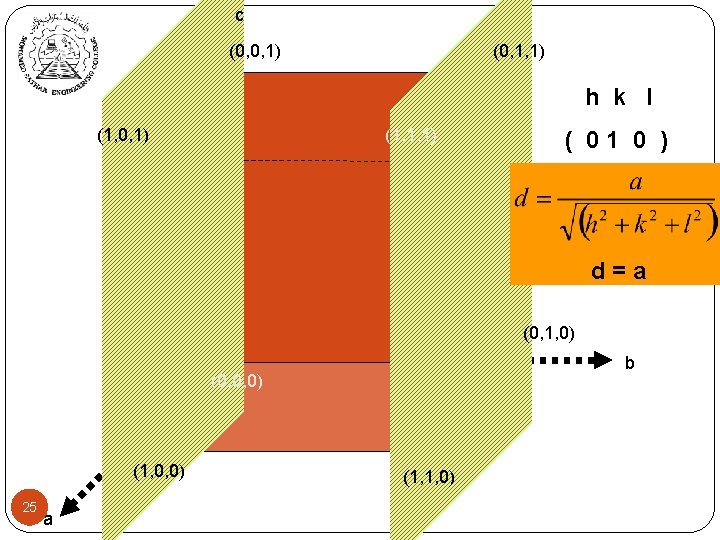
c (0, 0, 1) (0, 1, 1) h k l (1, 0, 1) (1, 1, 1) ( 01 0 ) d=a (0, 1, 0) b (0, 0, 0) (1, 0, 0) 25 a (1, 1, 0)
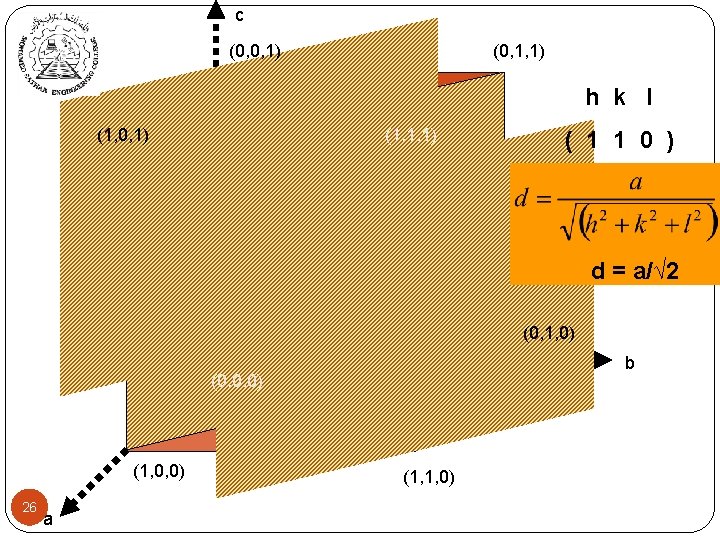
c (0, 0, 1) (0, 1, 1) h k l (1, 0, 1) (1, 1, 1) ( 1 1 0 ) d = a/√ 2 (0, 1, 0) b (0, 0, 0) (1, 0, 0) 26 a (1, 1, 0)
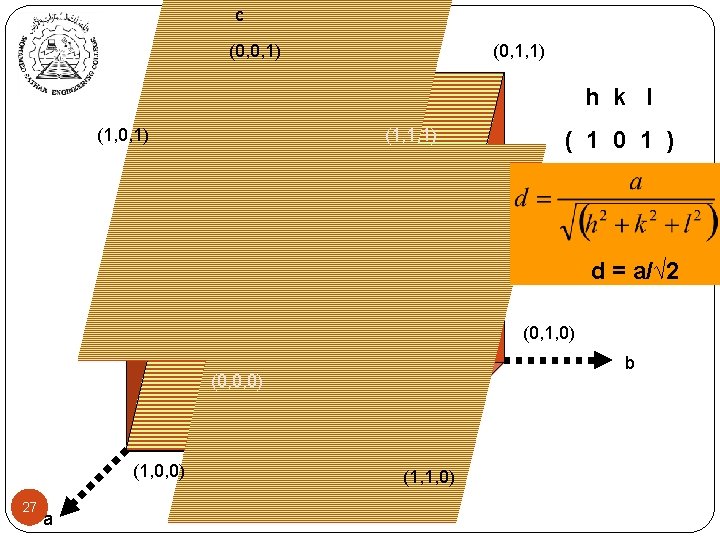
c (0, 0, 1) (0, 1, 1) h k l (1, 0, 1) (1, 1, 1) ( 1 0 1 ) d = a/√ 2 (0, 1, 0) b (0, 0, 0) (1, 0, 0) 27 a (1, 1, 0)
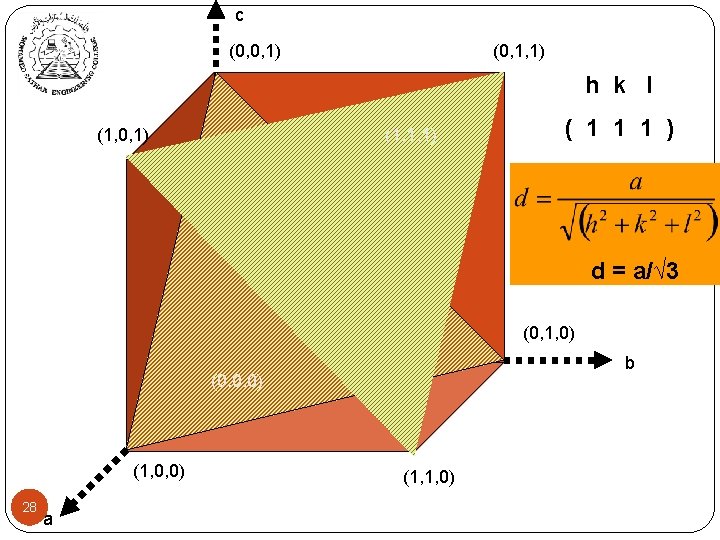
c (0, 0, 1) (0, 1, 1) h k l (1, 0, 1) (1, 1, 1) ( 1 1 1 ) d = a/√ 3 (0, 1, 0) b (0, 0, 0) (1, 0, 0) 28 a (1, 1, 0)
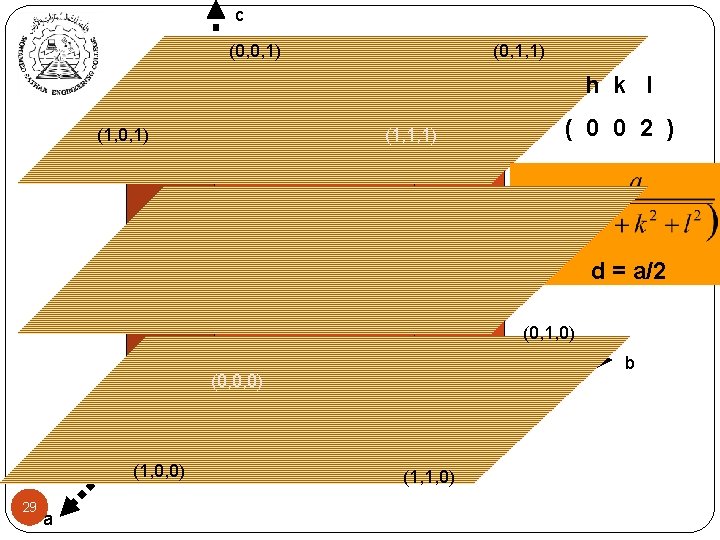
c (0, 0, 1) (0, 1, 1) h k l (1, 0, 1) (1, 1, 1) ( 0 0 2 ) d = a/2 (0, 1, 0) b (0, 0, 0) (1, 0, 0) 29 a (1, 1, 0)
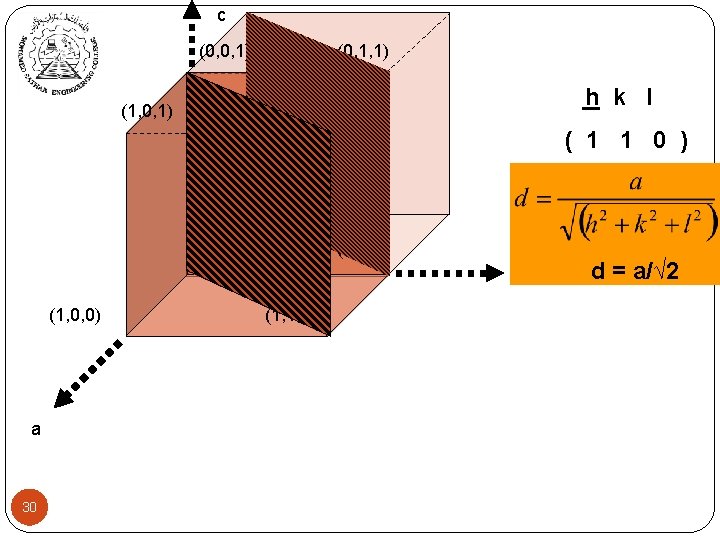
c (0, 0, 1) (1, 0, 1) (0, 1, 1) h k l (1, 1, 1) ( 1 1 0 ) (, -1 0, 0) (0, 1, 0) (0, 0, 0) (1, 0, 0) a 30 b (1, 1, 0) d = a/√ 2
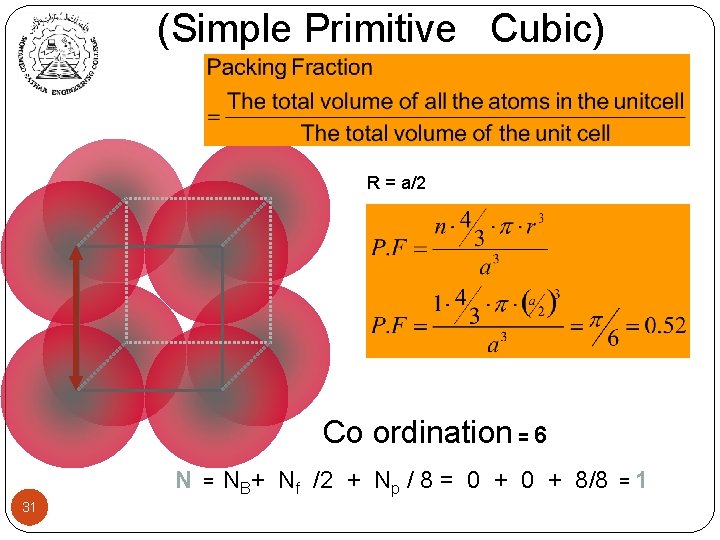
(Simple Primitive Cubic) R = a/2 Co ordination = 6 N 31 = NB+ Nf /2 + Np / 8 = 0 + 8/8 =1
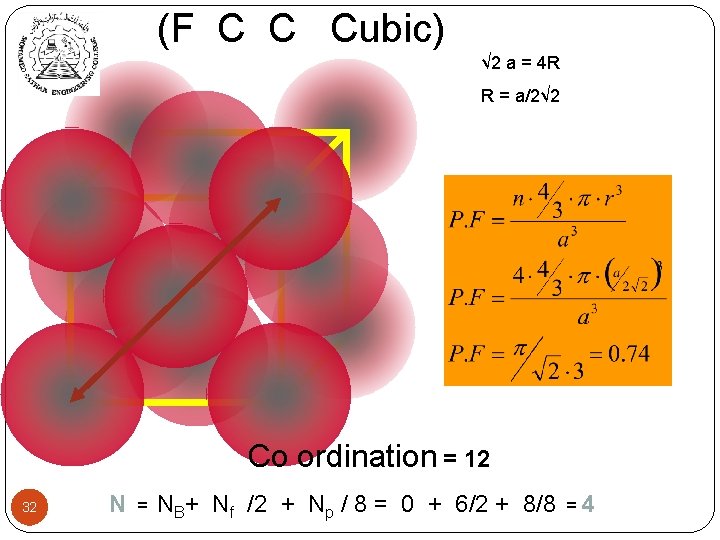
(F C C Cubic) √ 2 a = 4 R R = a/2√ 2 Co ordination = 12 32 N = NB+ Nf /2 + Np / 8 = 0 + 6/2 + 8/8 =4
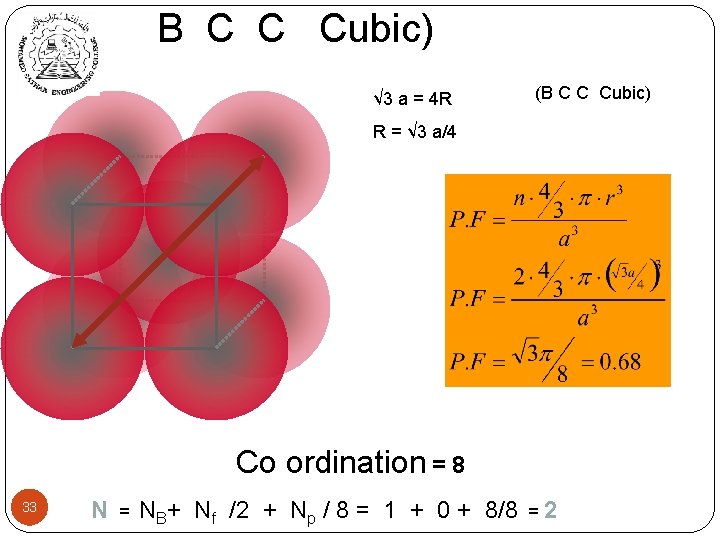
B C C Cubic) √ 3 a = 4 R (B C C Cubic) R = √ 3 a/4 Co ordination = 8 33 N = NB+ Nf /2 + Np / 8 = 1 + 0 + 8/8 =2
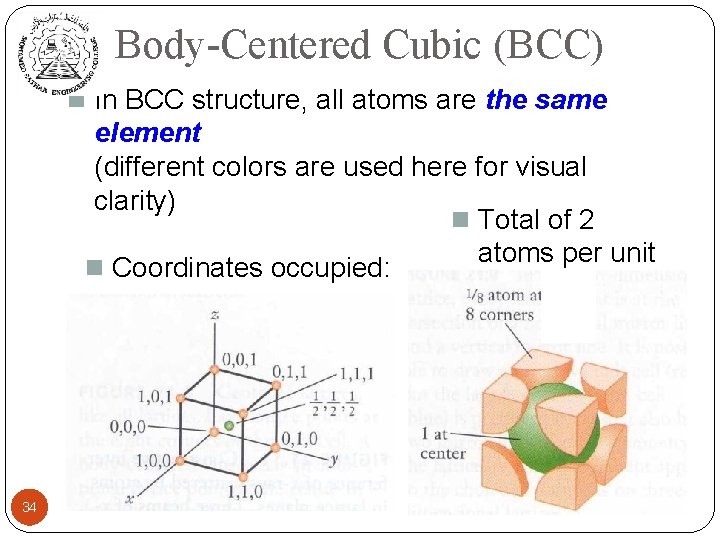
Body-Centered Cubic (BCC) n In BCC structure, all atoms are the same element (different colors are used here for visual clarity) n Total of 2 atoms per unit n Coordinates occupied: cell: 34
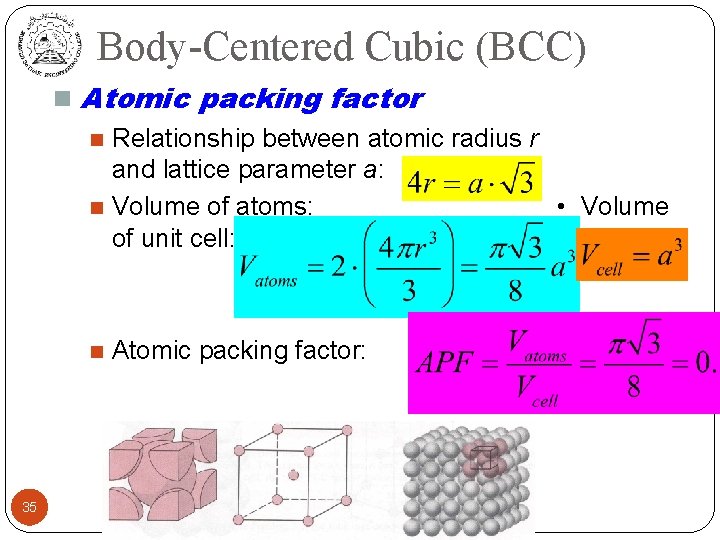
Body-Centered Cubic (BCC) n Atomic packing factor Relationship between atomic radius r and lattice parameter a: n Volume of atoms: • Volume of unit cell: n n 35 Atomic packing factor:
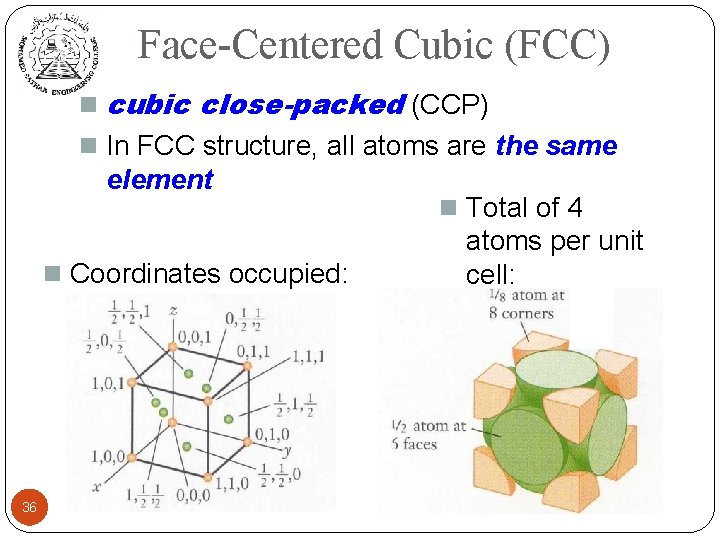
Face-Centered Cubic (FCC) n cubic close-packed (CCP) n In FCC structure, all atoms are the same element n Coordinates occupied: 36 n Total of 4 atoms per unit cell:
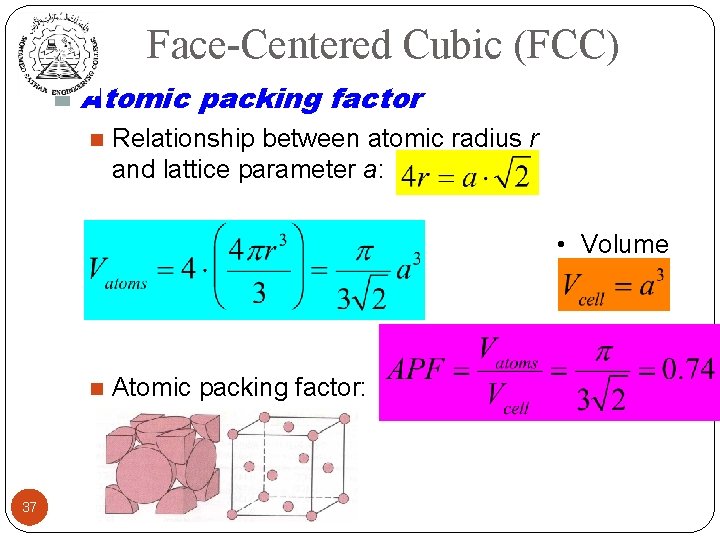
Face-Centered Cubic (FCC) n Atomic packing factor 37 n Relationship between atomic radius r and lattice parameter a: n Volume of atoms: of unit cell: n Atomic packing factor: • Volume
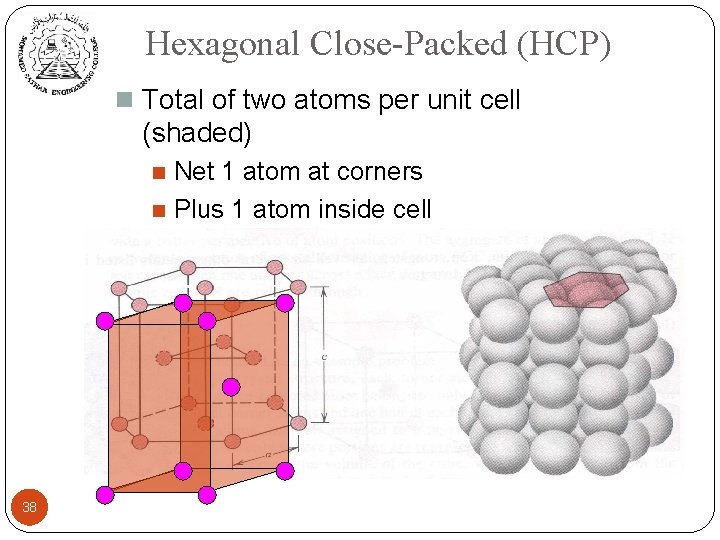
Hexagonal Close-Packed (HCP) n Total of two atoms per unit cell (shaded) Net 1 atom at corners n Plus 1 atom inside cell n 38
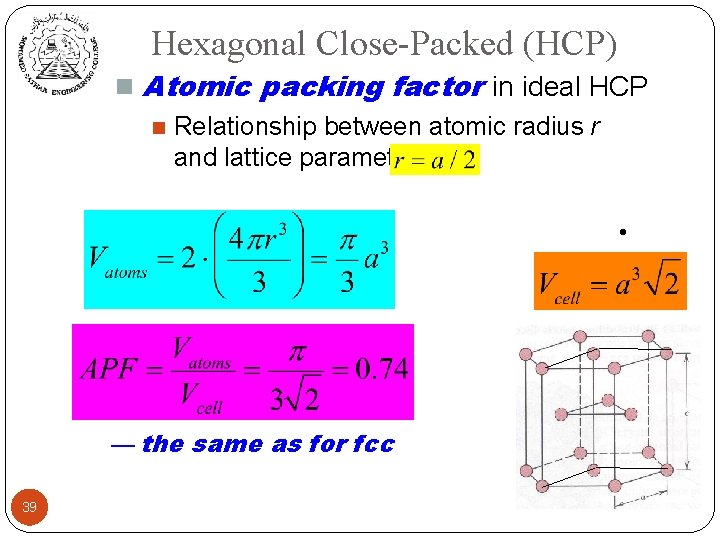
Hexagonal Close-Packed (HCP) n Atomic packing factor in ideal HCP n Relationship between atomic radius r and lattice parameter a: n Volume of atoms: Volume of unit cell: — the same as for fcc 39 •
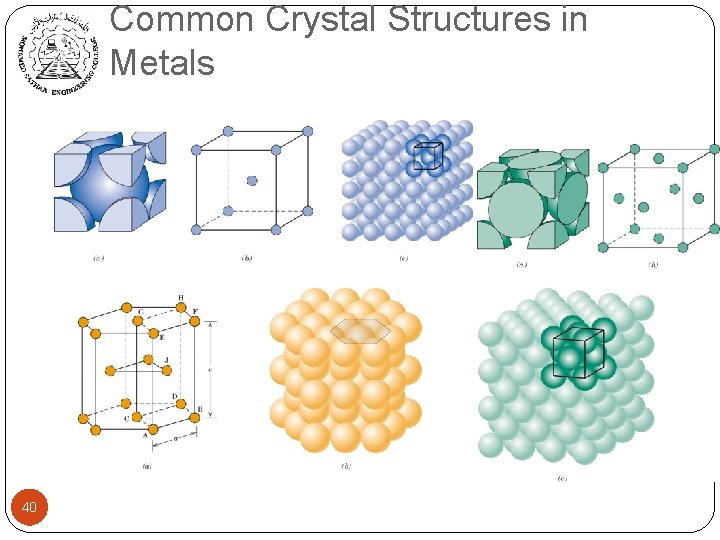
Common Crystal Structures in Metals 40
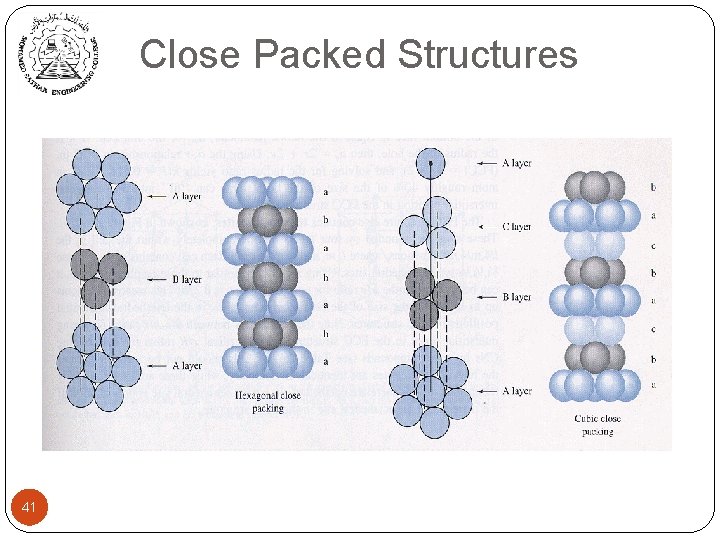
Close Packed Structures 41
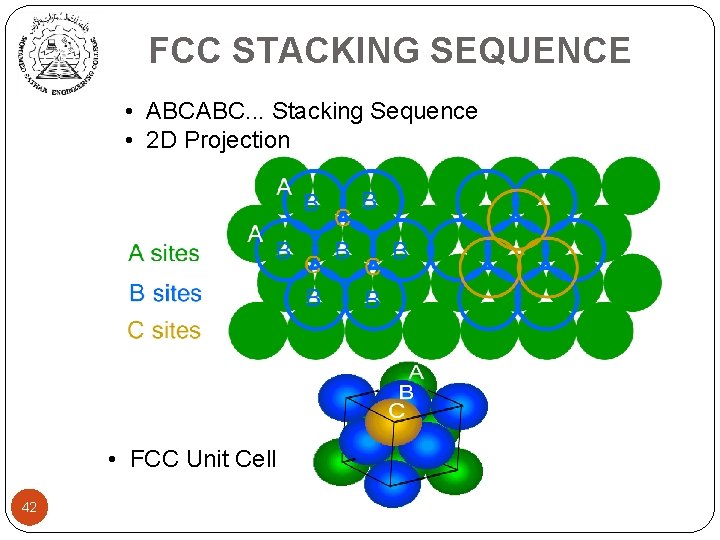
FCC STACKING SEQUENCE • ABCABC. . . Stacking Sequence • 2 D Projection • FCC Unit Cell 42
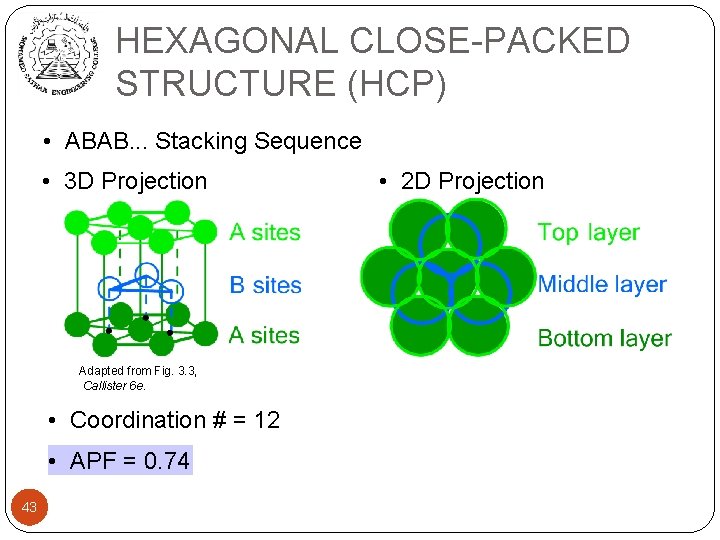
HEXAGONAL CLOSE-PACKED STRUCTURE (HCP) • ABAB. . . Stacking Sequence • 3 D Projection Adapted from Fig. 3. 3, Callister 6 e. • Coordination # = 12 • APF = 0. 74 43 • 2 D Projection
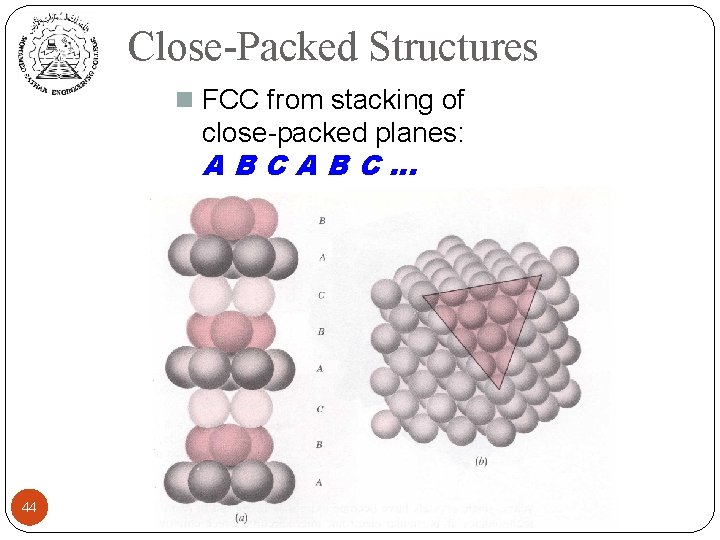
Close-Packed Structures n FCC from stacking of close-packed planes: ABCABC… 44
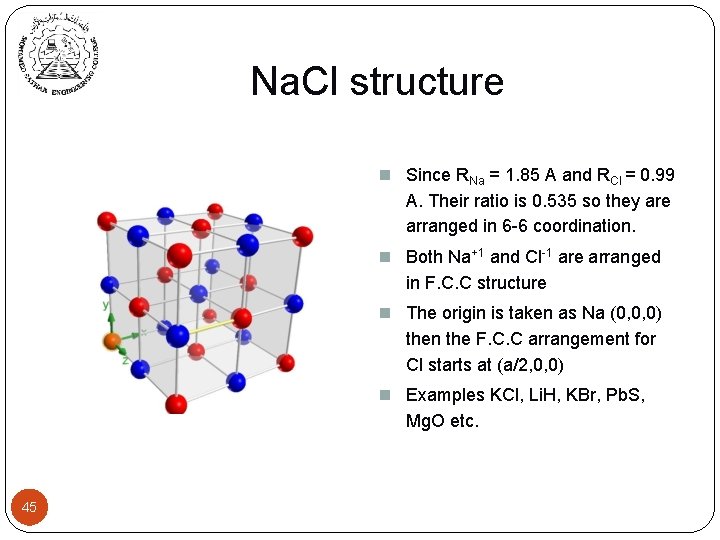
Na. Cl structure n Since RNa = 1. 85 A and RCl = 0. 99 A. Their ratio is 0. 535 so they are arranged in 6 -6 coordination. n Both Na+1 and Cl-1 are arranged in F. C. C structure n The origin is taken as Na (0, 0, 0) then the F. C. C arrangement for Cl starts at (a/2, 0, 0) n Examples KCl, Li. H, KBr, Pb. S, Mg. O etc. 45
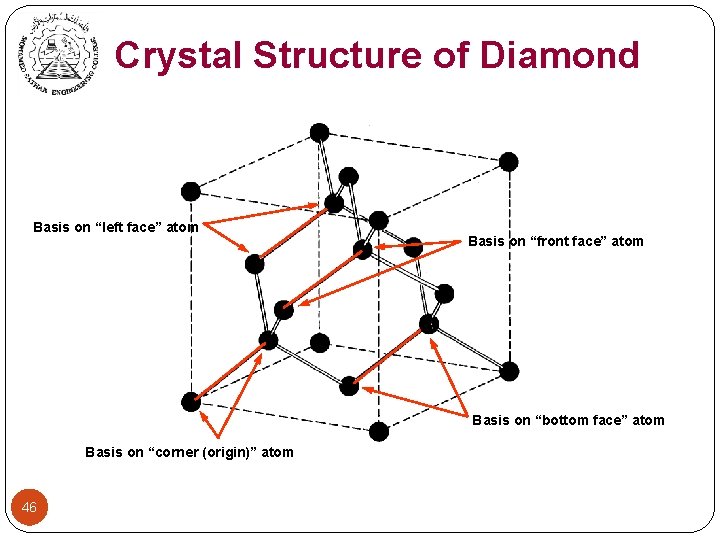
Crystal Structure of Diamond Basis on “left face” atom Basis on “front face” atom Basis on “bottom face” atom Basis on “corner (origin)” atom 46

CRYSTAL DEFECTS All atoms are at rest on their correct lattice position. Metals are not perfect neither at the macro level and nor at the micro level Contain a number of different types of crystalline defects (at the atomic level) Vibration of atoms can be regarded as a form of defects. Defects are important in many processes e. g. diffusion, deformation. 47

Classification of defects in solids Zero-dimensional (point) defects Vacancies, Interstitial atoms (ions), Foreign atoms (ions) One-dimensional (linear) defects Edge dislocation, screw dislocation Two-dimensional (flat) defects Antiphase boundary, shear plane, low angle twist boundary, low angle tilt boundary, grain boundary, surface Three-dimensional (spatial) defects Pores, foreign inclusions 48
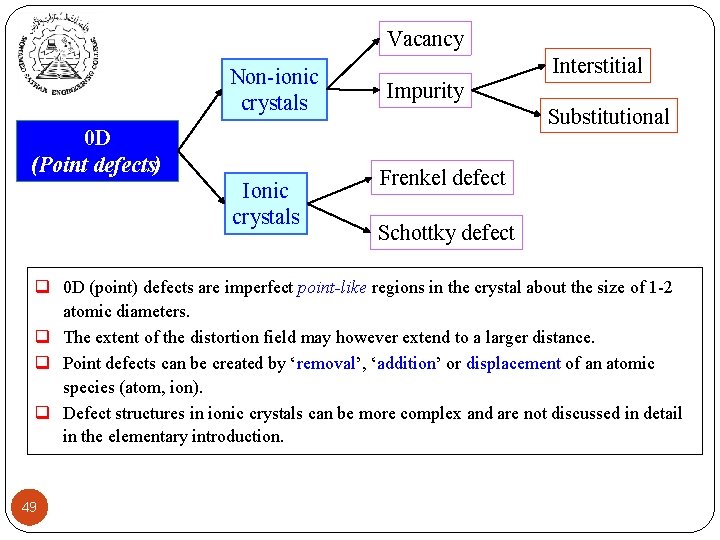
Vacancy Non-ionic crystals 0 D (Point defects) Ionic crystals Interstitial Impurity Substitutional Frenkel defect Schottky defect q 0 D (point) defects are imperfect point-like regions in the crystal about the size of 1 -2 atomic diameters. q The extent of the distortion field may however extend to a larger distance. q Point defects can be created by ‘removal’, ‘addition’ or displacement of an atomic species (atom, ion). q Defect structures in ionic crystals can be more complex and are not discussed in detail in the elementary introduction. 49
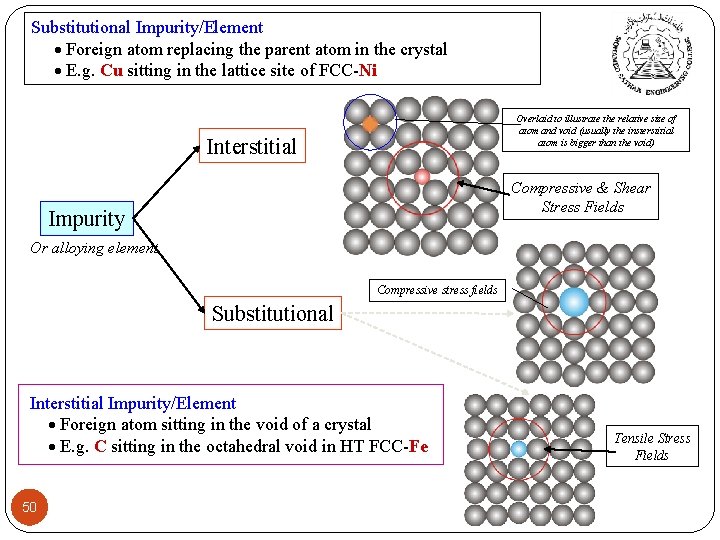
Substitutional Impurity/Element Foreign atom replacing the parent atom in the crystal E. g. Cu sitting in the lattice site of FCC-Ni Overlaid to illustrate the relative size of atom and void (usually the insterstitial atom is bigger than the void) Interstitial Compressive & Shear Stress Fields Impurity Or alloying element Compressive stress fields Substitutional Interstitial Impurity/Element Foreign atom sitting in the void of a crystal E. g. C sitting in the octahedral void in HT FCC-Fe 50 Tensile Stress Fields

One-dimensional (linear) defects Linear Defects (Dislocations) Are one-dimensional defects around which atoms are misaligned Ø Edge dislocation: Ø extra half-plane of atoms inserted in a crystal structure Ø b perpendicular ( ) to dislocation line Ø Screw dislocation: spiral planar ramp resulting from shear deformation b parallel ( ) to dislocation line ü Burger’s vector, b: measure of lattice distortion 51
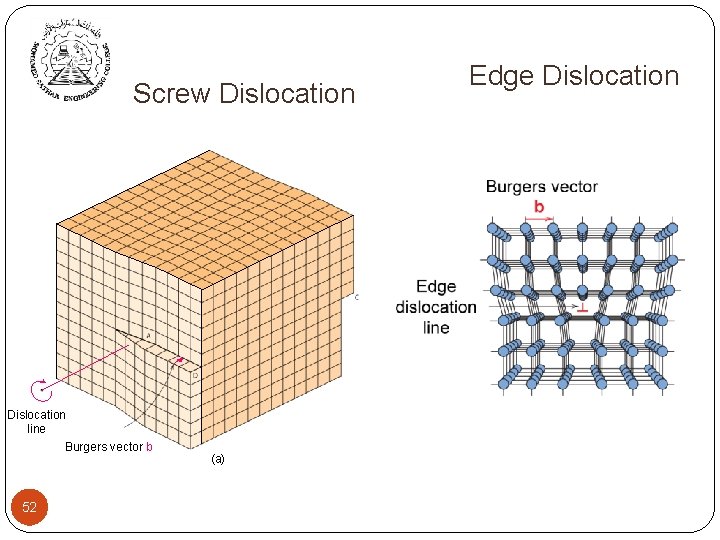
Screw Dislocation Edge Dislocation Screw Dislocation line Burgers vector b 52 (a) 52

Crystal Growth Techniques 1. Growth from solution Idea: homogeneous soln -> solid xtals + solvent 2. Growth from gas (vapor) phase Idea: Evaporate powder, deposit vapor onto seeds 3. Growth from liquid (melt) phase Idea: Melt polyxtals of desired materials, slowly cool 53
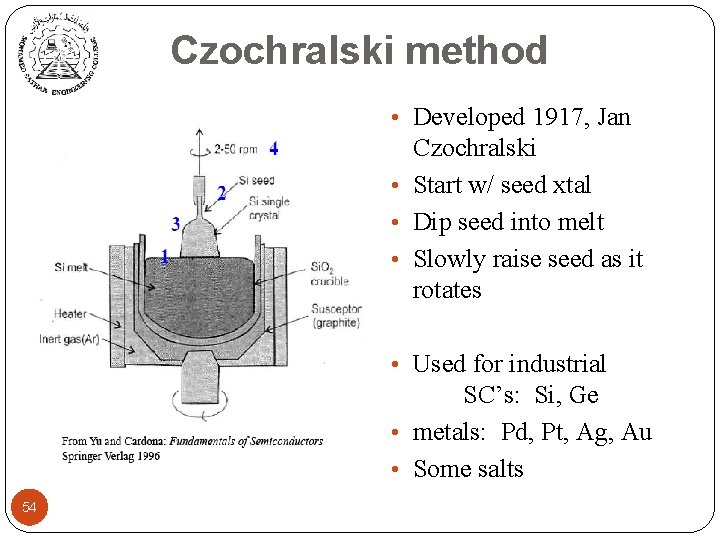
Czochralski method • Developed 1917, Jan Czochralski • Start w/ seed xtal • Dip seed into melt • Slowly raise seed as it rotates • Used for industrial SC’s: Si, Ge • metals: Pd, Pt, Ag, Au • Some salts 54
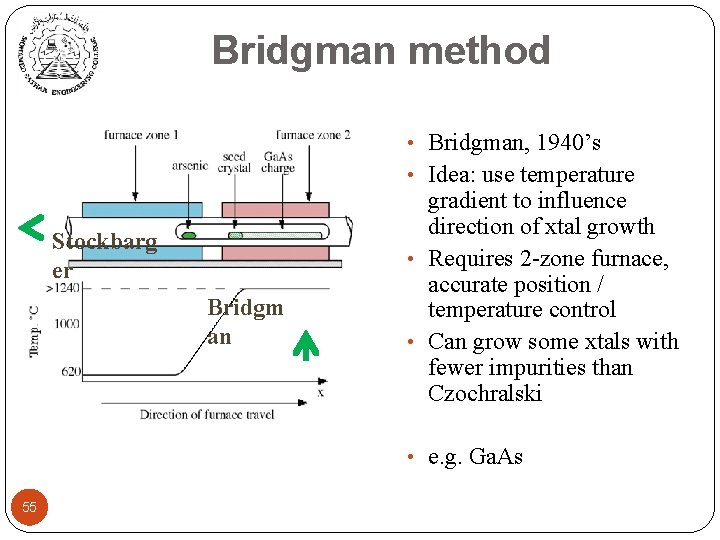
Bridgman method • Bridgman, 1940’s • Idea: use temperature Stockbarg er Bridgm an gradient to influence direction of xtal growth • Requires 2 -zone furnace, accurate position / temperature control • Can grow some xtals with fewer impurities than Czochralski • e. g. Ga. As 55
Low temperature solution growth Solution Growth Slow cooling method Temperature gradient method Slow evaporation method 56

Slow cooling method • Supersaturated solution • Systematic cooling • Solubility – High in higher temperature Low in lower temperature • Reduce the temperature – excess amount of solute (substance) gets crystallized • 45 -75°C 57

Slow evaporation method • Supersaturated solution • Constant temperature – Temperature stabilization - ± 0. 005°C – Rates of evaporation – few mm /hr 3 • Solubility – High in large amount of solvent Low in small amount of solvent • Evaporation of solvent – excess amount of solute (substance) gets crystallized 58

Temperature gradient method • Transport of the materials from hot (Source) to cold region Advantage Crystal grows at fixed temperature Insensitive to changes in temperature Economy of solvent and solute 59

Any Question ? Thank You. 60
- Slides: 60