GIANT IONIC LATTICES Ionic Bonding the net electrostatic
GIANT IONIC LATTICES
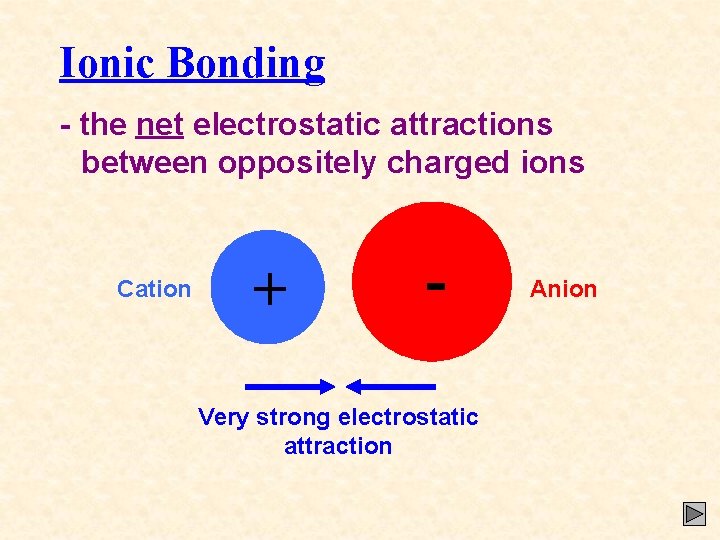
Ionic Bonding - the net electrostatic attractions between oppositely charged ions Cation + - Very strong electrostatic attraction Anion

How are ions formed? By atoms losing or gaining electrons More electronegative non-metal atoms GAIN electrons FROM metal atoms CATIONS (+) are formed when atoms (USUALLY METALS) lose electrons to non-metal Example Na - 1 s 22 p 63 s 1 loses its 3 s electron to form Na+ - 1 s 22 p 63 s 0 ANIONS (-) are formed when atoms (USUALLY NON-METALS) gain electrons from metal Example F - 1 s 22 p 5 gains an electron to form F- - 1 s 22 p 6 Ions are isoelectronic with nearest noble gas
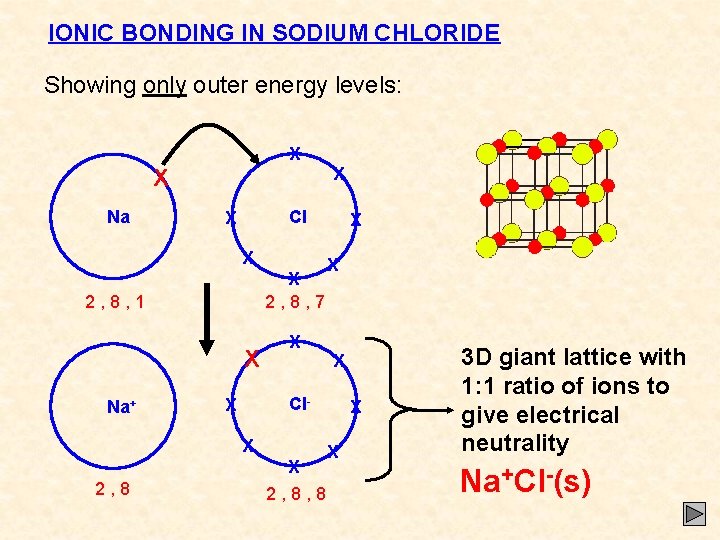
IONIC BONDING IN SODIUM CHLORIDE Showing only outer energy levels: X X Na Cl X X X 2, 8, 7 2, 8, 1 X Na+ X X Cl- X X X 2, 8, 8 X X 3 D giant lattice with 1: 1 ratio of ions to give electrical neutrality Na+Cl-(s)

General Ionic Structure • ions arrange in a giant lattice structure • cations and anions arranged alternately • 3 -dimensional array • lattice is very strong due to many ionic bonds
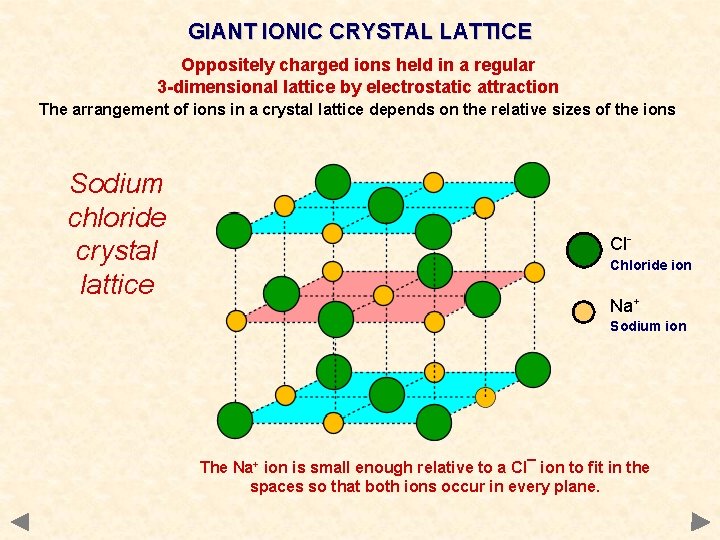
GIANT IONIC CRYSTAL LATTICE Oppositely charged ions held in a regular 3 -dimensional lattice by electrostatic attraction The arrangement of ions in a crystal lattice depends on the relative sizes of the ions Sodium chloride crystal lattice Cl. Chloride ion Na+ Sodium ion The Na+ ion is small enough relative to a Cl¯ ion to fit in the spaces so that both ions occur in every plane.
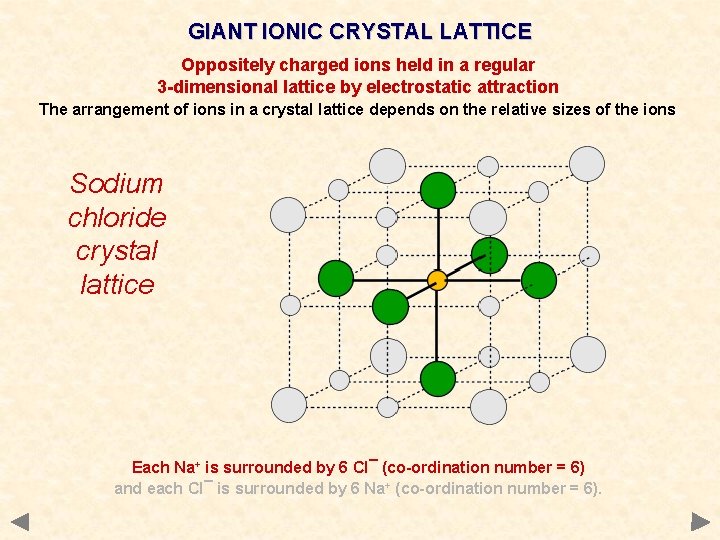
GIANT IONIC CRYSTAL LATTICE Oppositely charged ions held in a regular 3 -dimensional lattice by electrostatic attraction The arrangement of ions in a crystal lattice depends on the relative sizes of the ions Sodium chloride crystal lattice Each Na+ is surrounded by 6 Cl¯ (co-ordination number = 6) and each Cl¯ is surrounded by 6 Na+ (co-ordination number = 6).
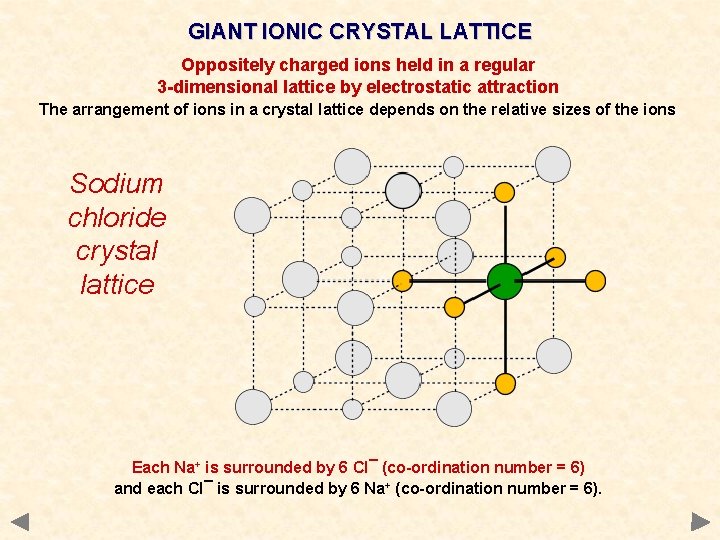
GIANT IONIC CRYSTAL LATTICE Oppositely charged ions held in a regular 3 -dimensional lattice by electrostatic attraction The arrangement of ions in a crystal lattice depends on the relative sizes of the ions Sodium chloride crystal lattice Each Na+ is surrounded by 6 Cl¯ (co-ordination number = 6) and each Cl¯ is surrounded by 6 Na+ (co-ordination number = 6).
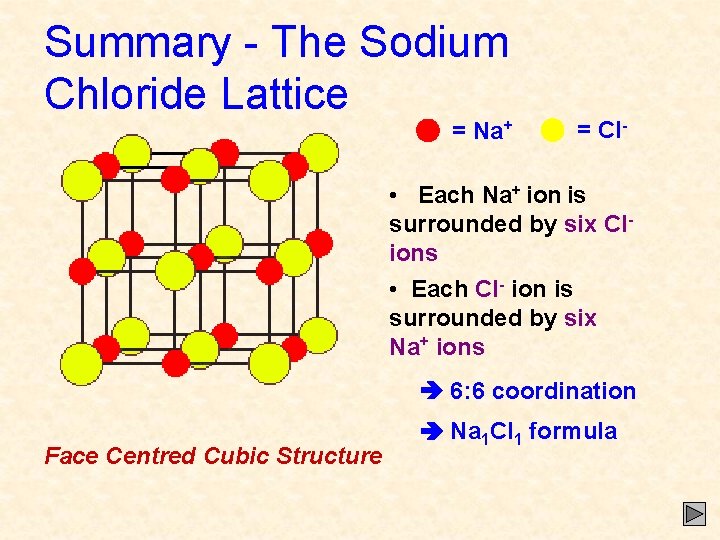
Summary - The Sodium Chloride Lattice = Na+ = Cl- • Each Na+ ion is surrounded by six Clions • Each Cl- ion is surrounded by six Na+ ions 6: 6 coordination Face Centred Cubic Structure Na 1 Cl 1 formula
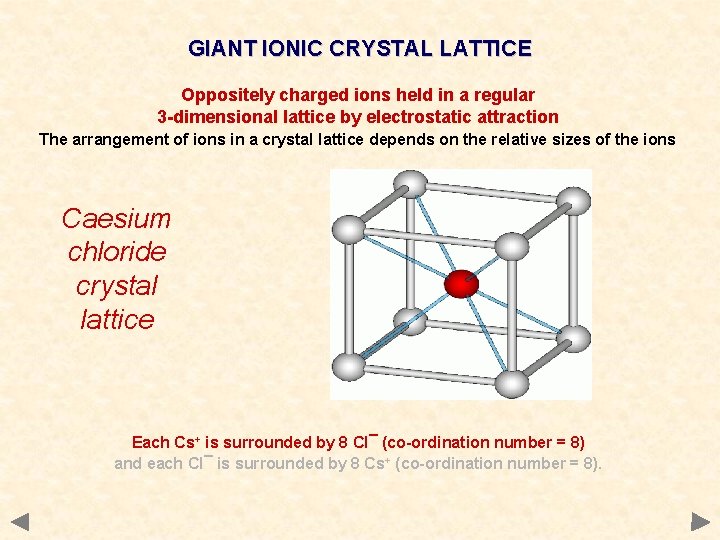
GIANT IONIC CRYSTAL LATTICE Oppositely charged ions held in a regular 3 -dimensional lattice by electrostatic attraction The arrangement of ions in a crystal lattice depends on the relative sizes of the ions Caesium chloride crystal lattice Each Cs+ is surrounded by 8 Cl¯ (co-ordination number = 8) and each Cl¯ is surrounded by 8 Cs+ (co-ordination number = 8).
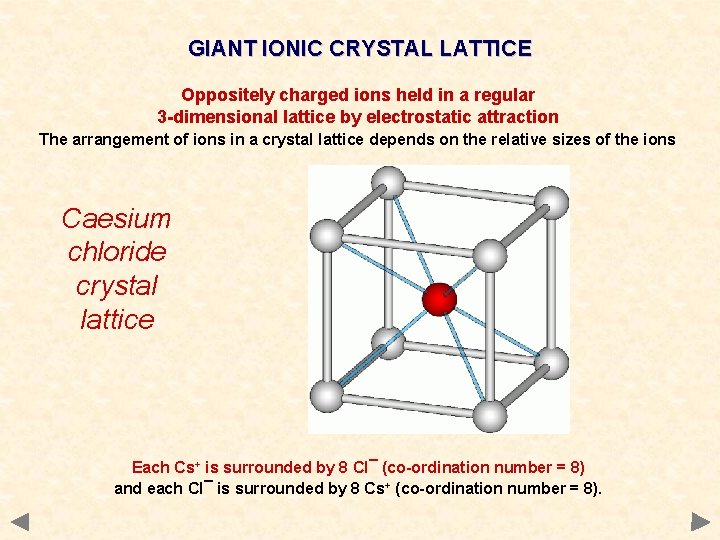
GIANT IONIC CRYSTAL LATTICE Oppositely charged ions held in a regular 3 -dimensional lattice by electrostatic attraction The arrangement of ions in a crystal lattice depends on the relative sizes of the ions Caesium chloride crystal lattice Each Cs+ is surrounded by 8 Cl¯ (co-ordination number = 8) and each Cl¯ is surrounded by 8 Cs+ (co-ordination number = 8).
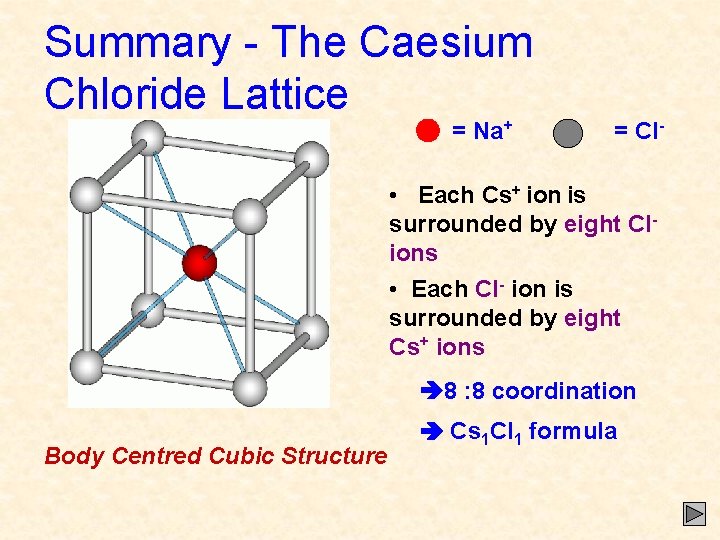
Summary - The Caesium Chloride Lattice = Na+ = Cl- • Each Cs+ ion is surrounded by eight Clions • Each Cl- ion is surrounded by eight Cs+ ions 8 : 8 coordination Body Centred Cubic Structure Cs 1 Cl 1 formula

A comparison of the sodium chloride and caesium chloride lattices Sodium Chloride Type of structure Face Centred Cubic Coordination number 6: 6 Caesium Chloride Body Centred Cubic 8: 8 The structures of Na. Cl and Cs. Cl are different because the Cs+ ion is larger than the Na+ ion, and is therefore able to surround itself with 8 Cl-ions, whereas the Na+ ion is smaller and can only fit 6 Cl- ions around itself

Physical properties of ionic compounds Melting point very high Strength Very brittle Electrical A large amount of energy must be put in to overcome the strong electrostatic attractions and separate the ions. Any dislocation leads to the layers moving and similarly charged ions being next to each other. The repulsion splits the crystal. do not conduct when solid - ions are held strongly in the lattice. conduct when molten or in aqueous solution - the ions become mobile and conduction takes place. Solubility. Insoluble in non-polar solvents but soluble in water Water as it is a polar solvent and stabilises the separated ions. Much energy is needed to overcome the electrostatic attraction and separate the ions stability attained by being surrounded by polar water molecules compensates for this
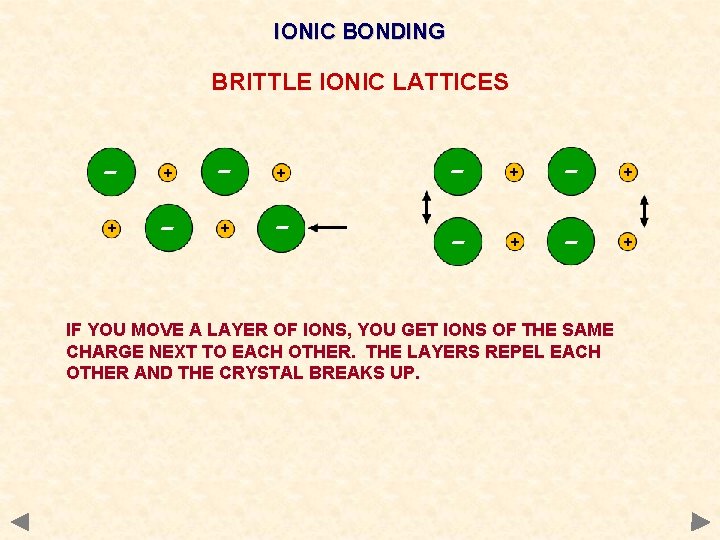
IONIC BONDING BRITTLE IONIC LATTICES - + + - - + - + IF YOU MOVE A LAYER OF IONS, YOU GET IONS OF THE SAME CHARGE NEXT TO EACH OTHER. THE LAYERS REPEL EACH OTHER AND THE CRYSTAL BREAKS UP.
- Slides: 15