3 Crystal Binding and Elastic Constants Crystals of

3. Crystal Binding and Elastic Constants • • • Crystals of Inert Gases Ionic Crystals Covalent Crystals Metals Hydrogen Bonds Atomic Radii Analysis of Elastic Strains Elastic Compliance and Stiffness Constants Elastic Waves in Cubic Crystals
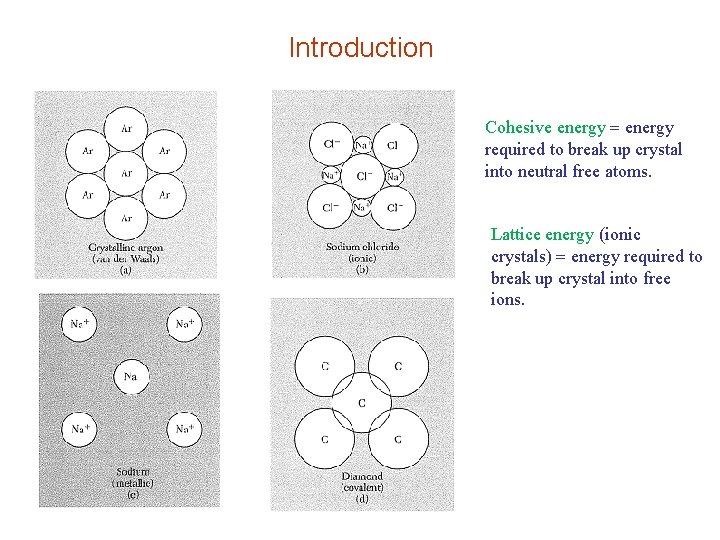
Introduction Cohesive energy required to break up crystal into neutral free atoms. Lattice energy (ionic crystals) energy required to break up crystal into free ions.
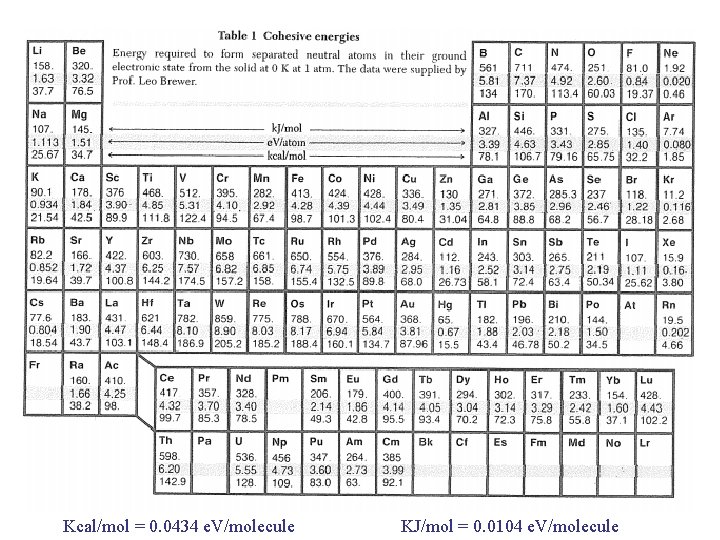
Kcal/mol = 0. 0434 e. V/molecule KJ/mol = 0. 0104 e. V/molecule
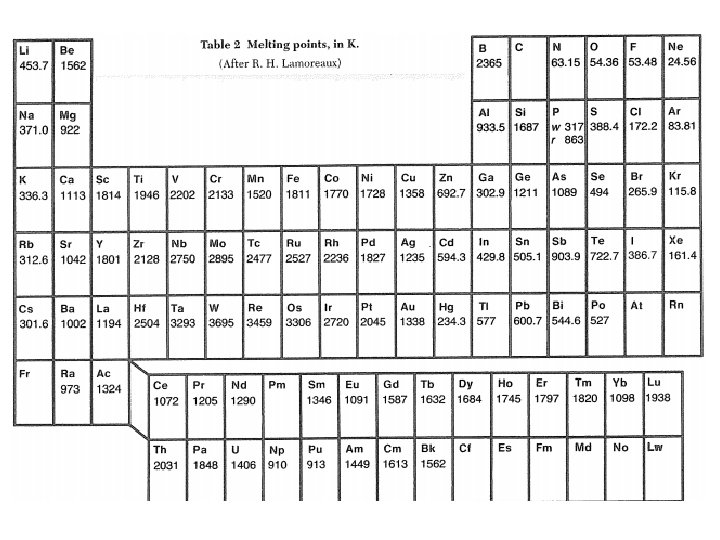
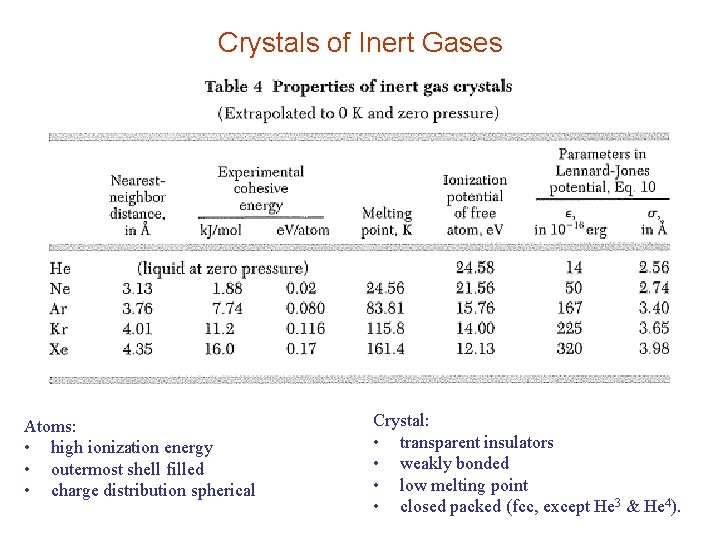
Crystals of Inert Gases Atoms: • high ionization energy • outermost shell filled • charge distribution spherical Crystal: • transparent insulators • weakly bonded • low melting point • closed packed (fcc, except He 3 & He 4).


Van der Waals – London Interaction Ref: A. Haug, “Theoretical Solid State Physics”, § 30, Vol I, Pergamon Press (1972). Van der Waals forces = induced dipole – dipole interaction between neutral atoms/molecules. Atom i charge +Q at Ri and charge –Q at Ri + xi. ( center of charge distributions )

H 0 = sum of atomic hamiltonians 0 = antisymmetrized product of ground state atomic functions 1 st order term vanishes if overlap of atomic functions negligible. 2 nd order term is negative & R 6 (van der Waals binding).
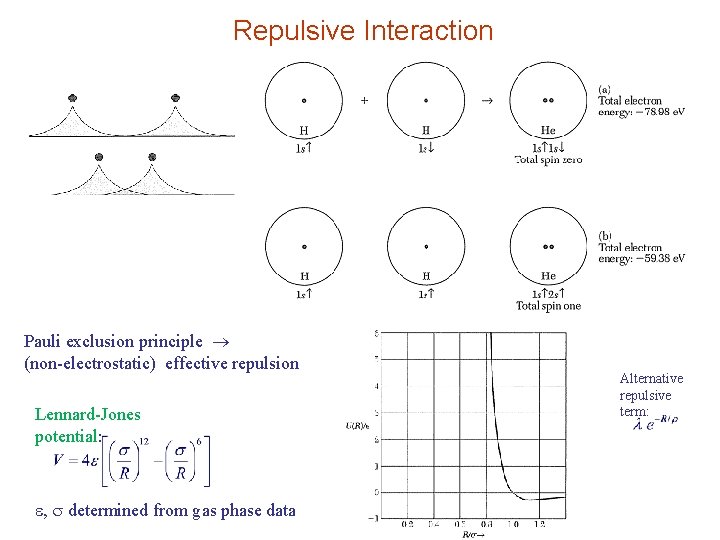
Repulsive Interaction Pauli exclusion principle (non-electrostatic) effective repulsion Lennard-Jones potential: , determined from gas phase data Alternative repulsive term:

Equilibrium Lattice Constants Neglecting K. E. For a fcc lattice: For a hcp lattice: At equilibrium: Experiment (Table 4): Error due to zero point motion R n. n. dist

Cohesive Energy for fcc lattices For low T, K. E. zero point motion. For a particle bounded within length , quantum correction is inversely proportional to the atomic mass: ~ 28, 10, 6, & 4% for Ne, Ar, Kr, Xe.
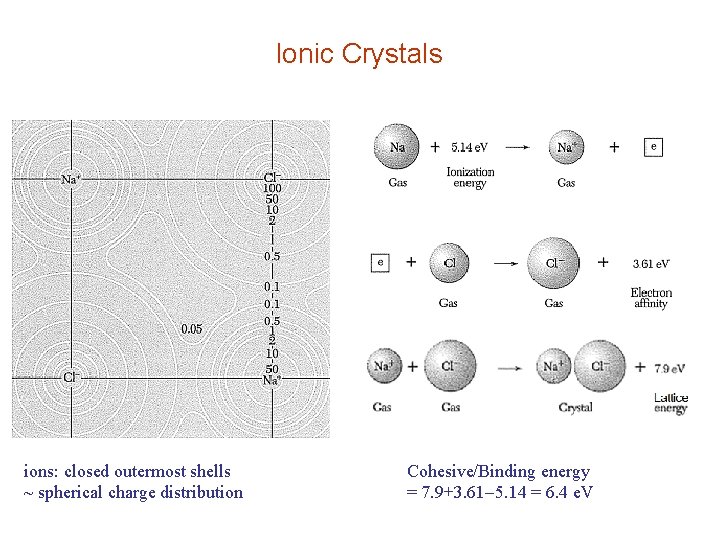
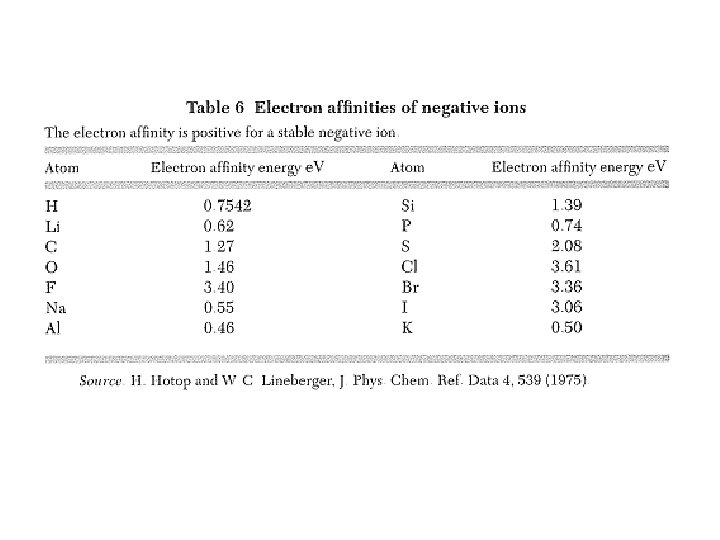
Ionic Crystals ions: closed outermost shells ~ spherical charge distribution Cohesive/Binding energy = 7. 9+3. 61 5. 14 = 6. 4 e. V
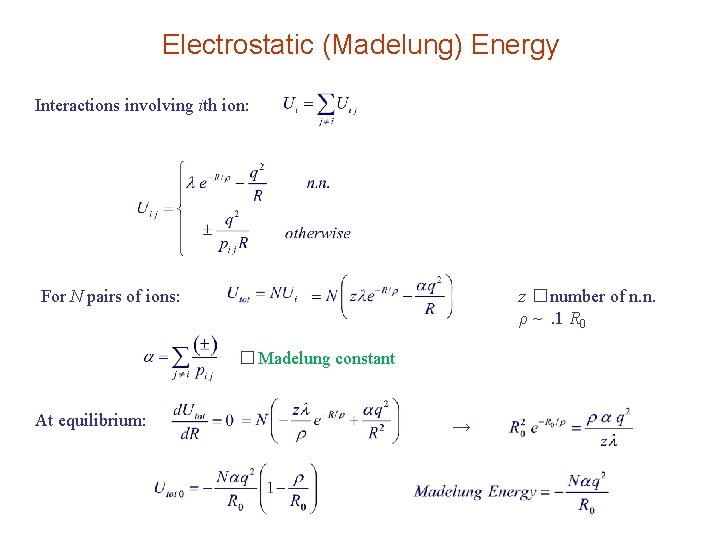
Electrostatic (Madelung) Energy Interactions involving ith ion: For N pairs of ions: z � number of n. n. ρ ~. 1 R 0 � Madelung constant At equilibrium: →
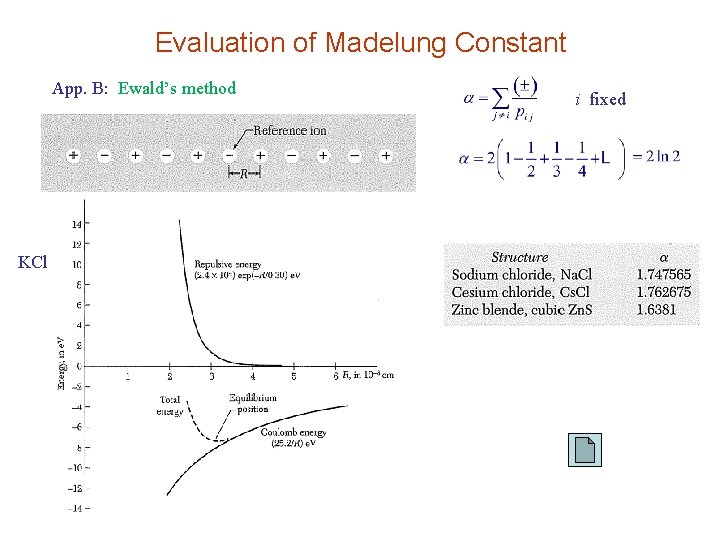
Evaluation of Madelung Constant App. B: Ewald’s method KCl i fixed
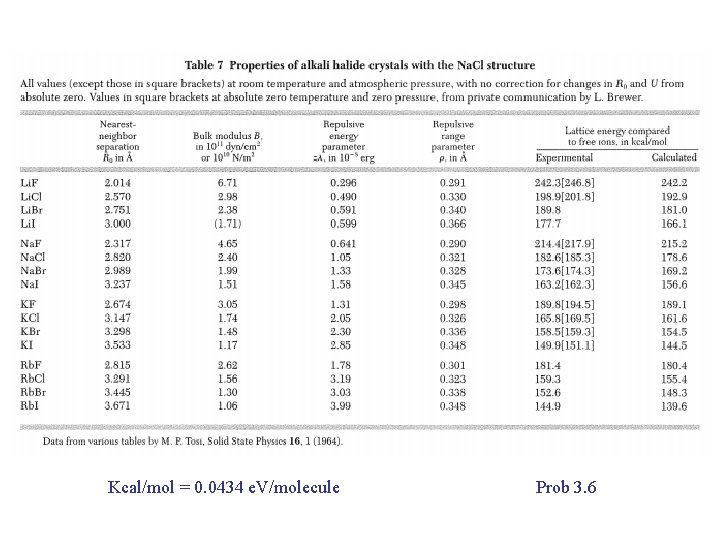
Kcal/mol = 0. 0434 e. V/molecule Prob 3. 6
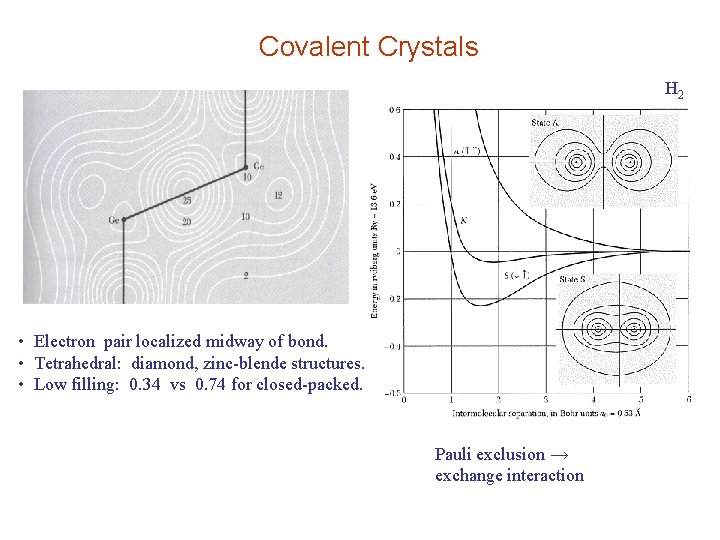
Covalent Crystals H 2 • Electron pair localized midway of bond. • Tetrahedral: diamond, zinc-blende structures. • Low filling: 0. 34 vs 0. 74 for closed-packed. Pauli exclusion → exchange interaction
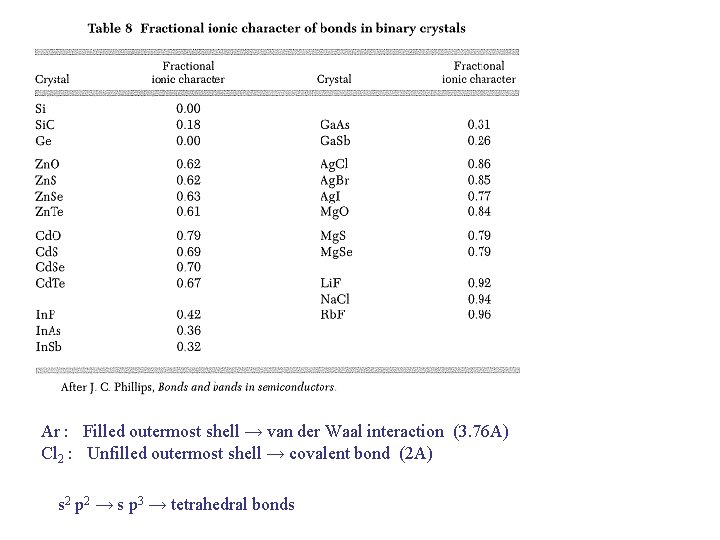
Ar : Filled outermost shell → van der Waal interaction (3. 76 A) Cl 2 : Unfilled outermost shell → covalent bond (2 A) s 2 p 2 → s p 3 → tetrahedral bonds

Metals Metallic bonding: • Non-directional, long-ranged. • Strength: vd. W < metallic < ionic < covalent • Structure: closed packed (fcc, hcp, bcc) • Transition metals: extra binding of d-electrons.

Hydrogen Bonds • • Energy ~ 0. 1 e. V Largely ionic ( between most electronegative atoms like O & N ). Responsible (together with the dipoles) for characteristics of H 2 O. Important in ferroelectric crystals & DNA.
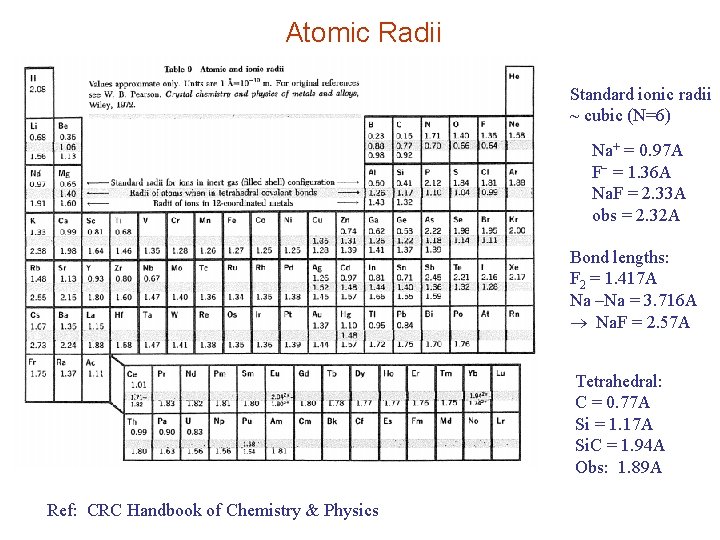
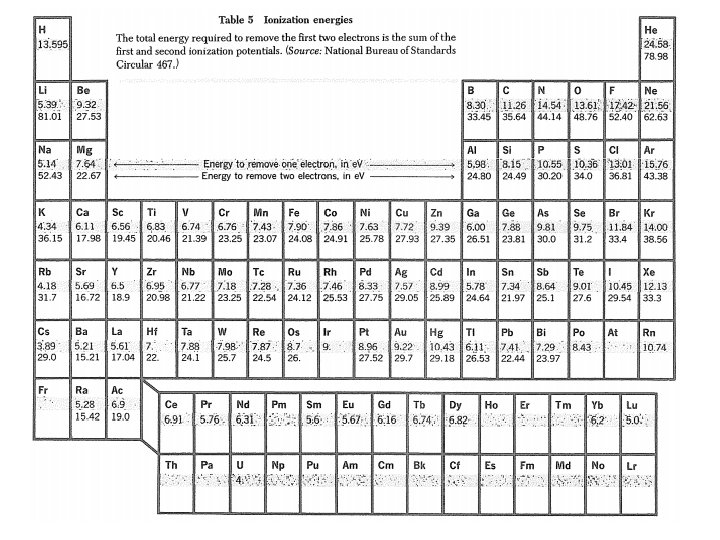
Atomic Radii Standard ionic radii ~ cubic (N=6) Na+ = 0. 97 A F = 1. 36 A Na. F = 2. 33 A obs = 2. 32 A Bond lengths: F 2 = 1. 417 A Na –Na = 3. 716 A Na. F = 2. 57 A Tetrahedral: C = 0. 77 A Si = 1. 17 A Si. C = 1. 94 A Obs: 1. 89 A Ref: CRC Handbook of Chemistry & Physics

Ionic Crystal Radii E. g. Ba. Ti. O 3 : a = 4. 004 A Ba++ – O– – : D 12 = 1. 35 + 1. 40 + 0. 19 = 2. 94 A → a = 4. 16 A Ti++++ – O – – : D 6 = 0. 68 + 1. 40 = 2. 08 A → a = 4. 16 A Bonding has some covalent character.

Analysis of Elastic Strains Ref: L. D. Landau, E. M. Lifshitz, “Theory of Elasticity”, Pergamon Press (59/86) Continuum approximation: good for λ > 30 A. Description of deformation (Cartesian coordinates): Displacement vector field u(r). Material point Nearby point = strain tensor = linear strain tensor

Dilation uik is symmetric → diagonalizable → principal axes such that (no summation over i ) → Fractional volume change Trace of uik

Stress Total force acting on a volume element inside solid f force density Newton’s 3 rd law → internal forces cancel each other → only forces on surface contribute This is guaranteed if so that σ stress tensor σik ith component of force acting on the surface element normal to the xk axis. Moment on volume element Only forces on surface contribute → (σ is symmetric)

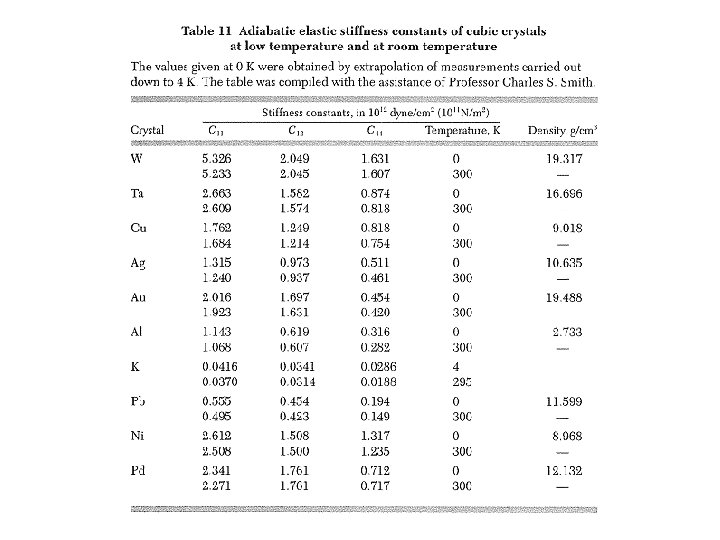
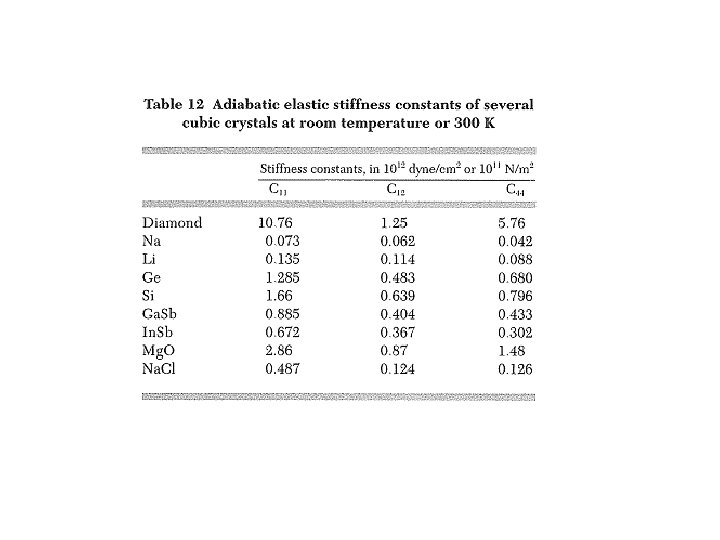
Elastic Compliance & Stiffness Constants σ and u are symmetric → they have at most 6 independent components Compact index notations (i , j) → α : (1, 1) → 1, (2, 2) → 2, (3, 3) → 3, (1, 2) = (2, 1) → 4, (2, 3) = (3, 2) → 5, (3, 1) = (1, 3) → 6 Elastic energy density: i , j , k, l = 1, 2, 3 α , β = 1, 2, …, 6 21 where elastic stiffness constants elastic modulus tensor Stress: uik & uki treated as independent S α β elastic compliance constants

Elastic Stiffness Constants for Cubic Crystals Invariance under reflections xi → –xi C with odd numbers of like indices vanishes Invariance under C 3 , i. e. , All C i j k l = 0 except for (summation notation suspended):

where

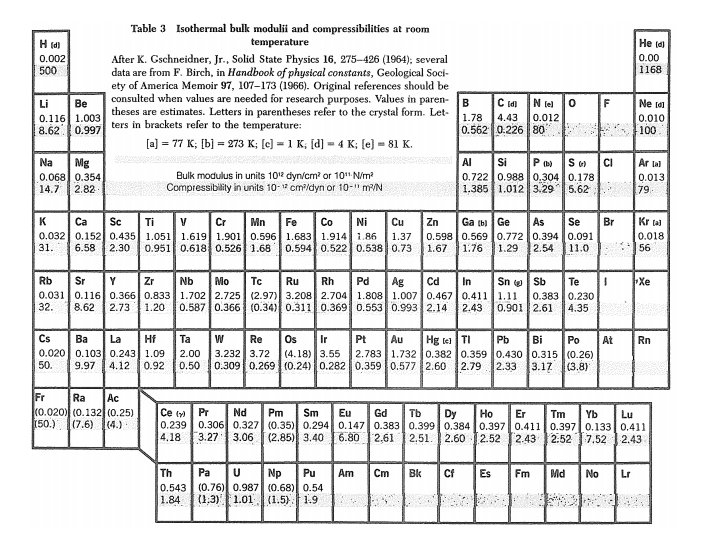
Bulk Modulus & Compressibility Uniform dilation: δ = Tr uik = fractional volume change B = Bulk modulus = 1/κ See table 3 for values of B & κ. κ = compressibility

Elastic Waves in Cubic Crystals Newton’s 2 nd law: don’t confuse ui with uα → Similarly

Dispersion Equation → dispersion equation
![Waves in the [100] direction → Longitudinal Transverse, degenerate Waves in the [100] direction → Longitudinal Transverse, degenerate](http://slidetodoc.com/presentation_image_h/c6c4e5f795b691babf257276ae981ba5/image-34.jpg)
Waves in the [100] direction → Longitudinal Transverse, degenerate
![Waves in the [110] direction → Lonitudinal Transverse Waves in the [110] direction → Lonitudinal Transverse](http://slidetodoc.com/presentation_image_h/c6c4e5f795b691babf257276ae981ba5/image-35.jpg)
Waves in the [110] direction → Lonitudinal Transverse
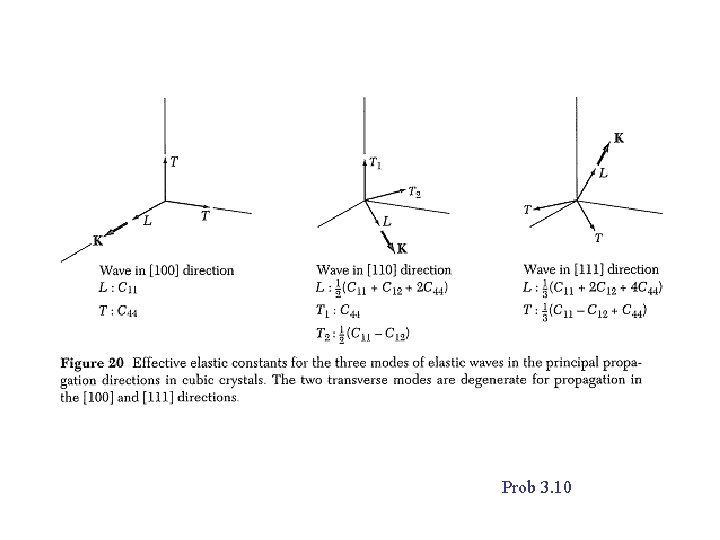
Prob 3. 10
- Slides: 38