Why Study Solid State Physics Ideal Crystal An









































- Slides: 71

Why Study Solid State Physics?

Ideal Crystal • An ideal crystal is a periodic array of structural units, such as atoms or molecules. • It can be constructed by the infinite repetition of these identical structural units in space. • Structure can be described in terms of a lattice, with a group of atoms attached to each lattice point. The group of atoms is the basis.

Bravais Lattice • An infinite array of discrete points with an arrangement and orientation that appears exactly the same, from any of the points the array is viewed from. • A three dimensional Bravais lattice consists of all points with position vectors R that can be written as a linear combination of primitive vectors. The expansion coefficients must be integers.

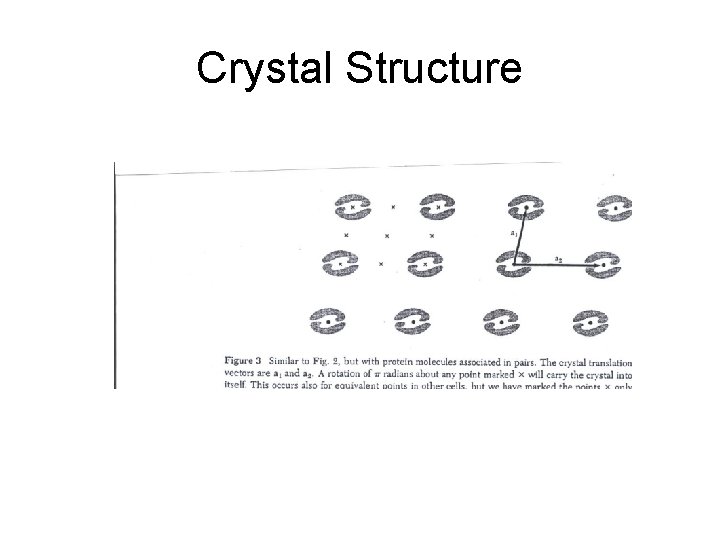
Crystal lattice: Proteins

Crystal Structure

Honeycomb: NOT Bravais

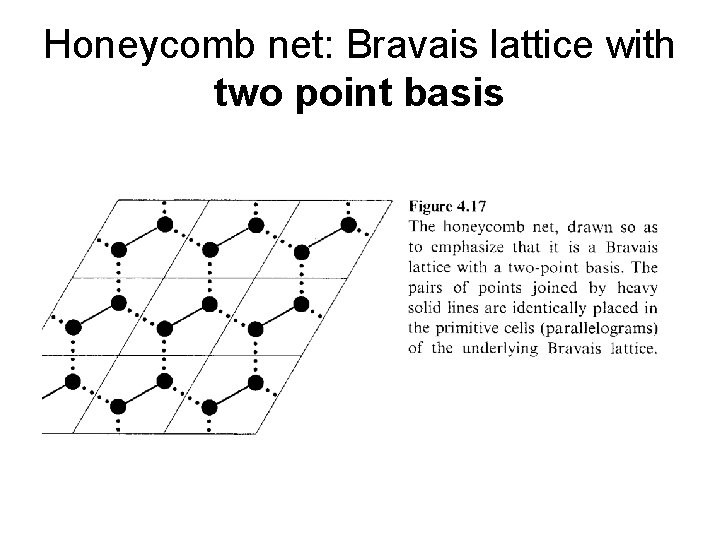
Honeycomb net: Bravais lattice with two point basis

Crystal structure: basis

Translation Vector T

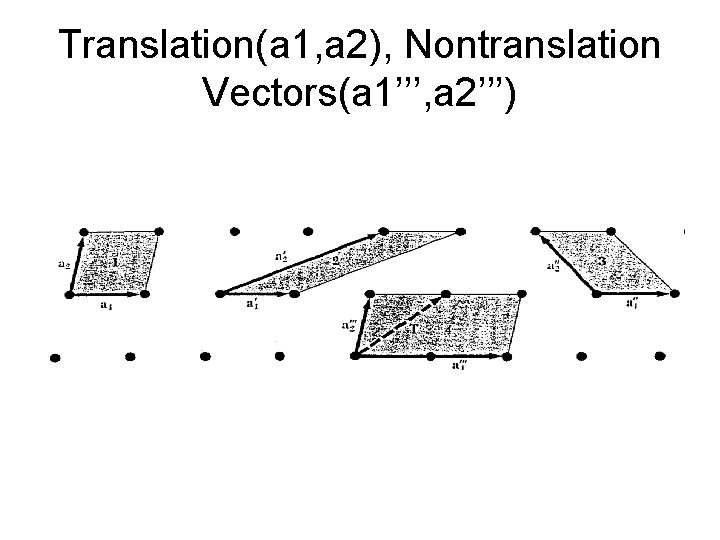
Translation(a 1, a 2), Nontranslation Vectors(a 1’’’, a 2’’’)

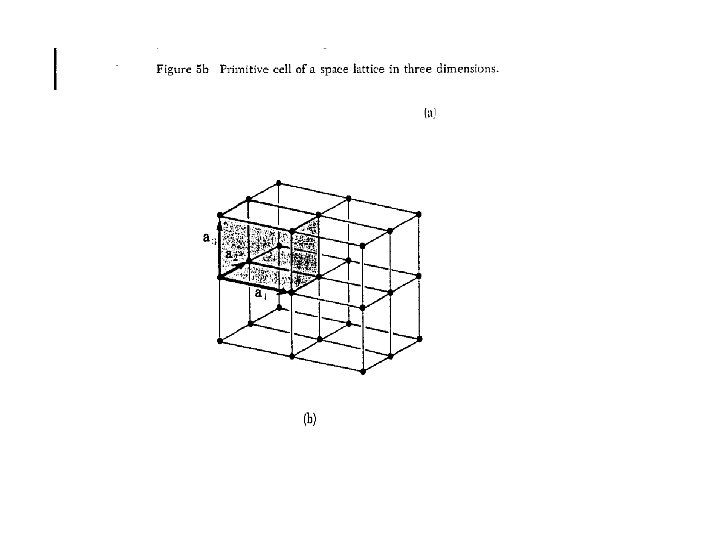
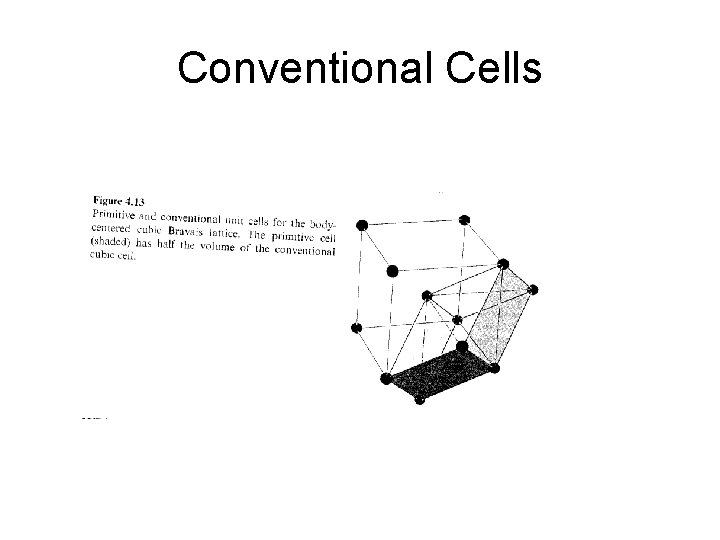
Primitive Unit Cell • A primitive cell or primitive unit cell is a volume of space that when translated through all the vectors in a Bravais lattice just fills all of space without either overlapping itself or leaving voids. • A primitive cell must contain precisely one lattice point.


Fundamental Types of Lattices • Crystal lattices can be mapped into themselves by the lattice translations T and by various other symmetry operations. • A typical symmetry operation is that of rotation about an axis that passes through a lattice point. Allowed rotations of : 2 π, 2π/2, 2π/3, 2π/4, 2π/6 • (Note: lattices do not have rotation axes for 1/5, 1/7 …) times 2π

Five fold axis of symmetry cannot exist

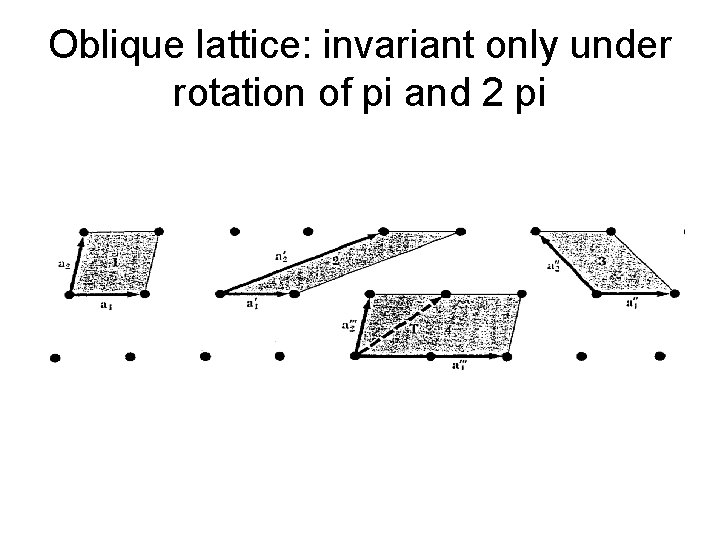
Two Dimensional Lattices • There is an unlimited number of possible lattices, since there is no restriction on the lengths of the lattice translation vectors or on the angle between them. An oblique lattice has arbitrary a 1 and a 2 and is invariant only under rotation of π and 2 π about any lattice point.

Oblique lattice: invariant only under rotation of pi and 2 pi

Two Dimensional Lattices

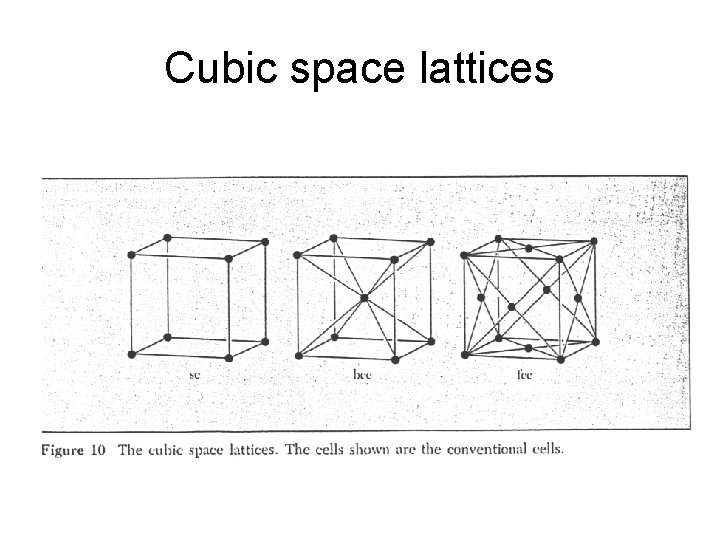
Three Dimensional Lattice Types

Wigner-Seitz Primitive Cell: Full symmetry of Bravais Lattice

Conventional Cells

Cubic space lattices

Cubic lattices

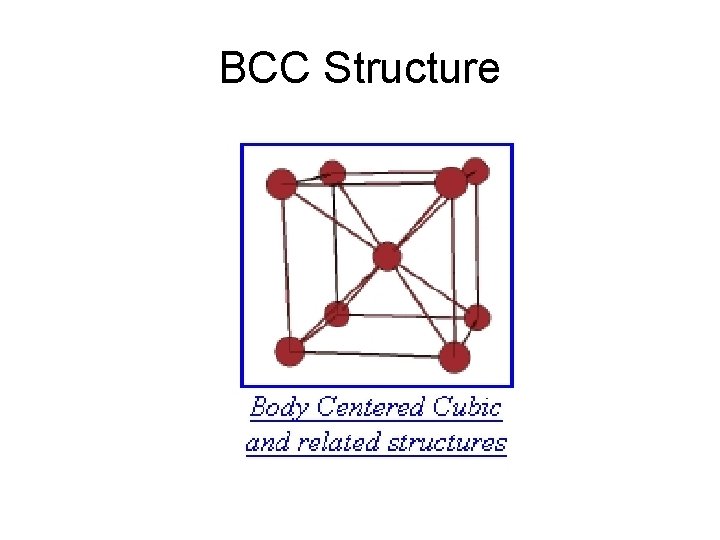
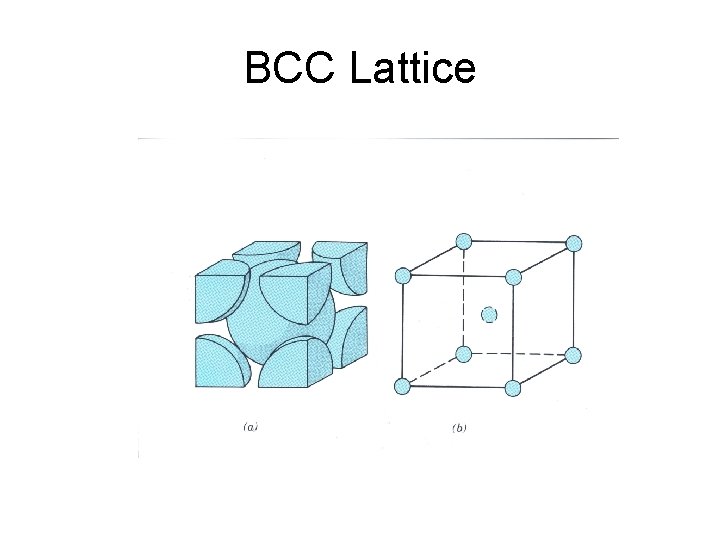
BCC Structure

BCC Crystal

BCC Lattice

Primitive vectors BCC

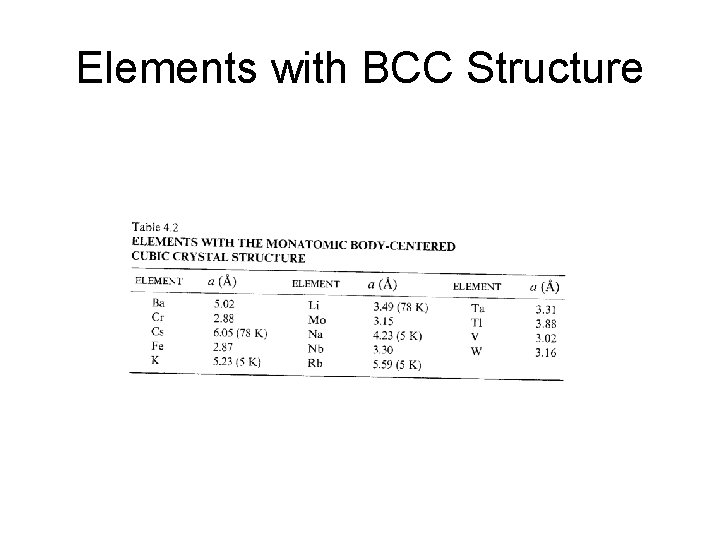
Elements with BCC Structure

Summary: Bravais Lattices (Nets) in Two Dimensions

Escher loved two dimensional structures too

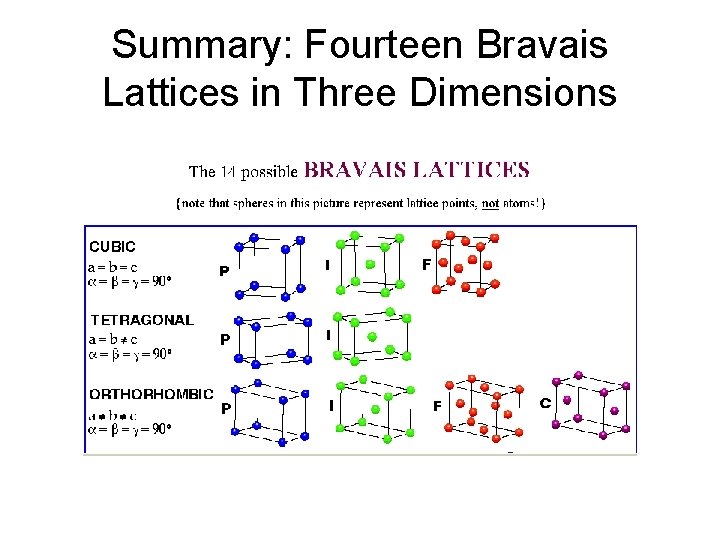
Summary: Fourteen Bravais Lattices in Three Dimensions

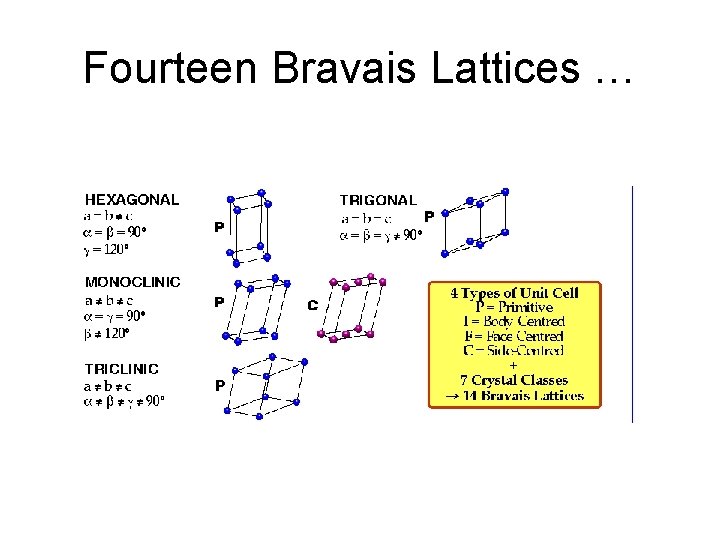
Fourteen Bravais Lattices …

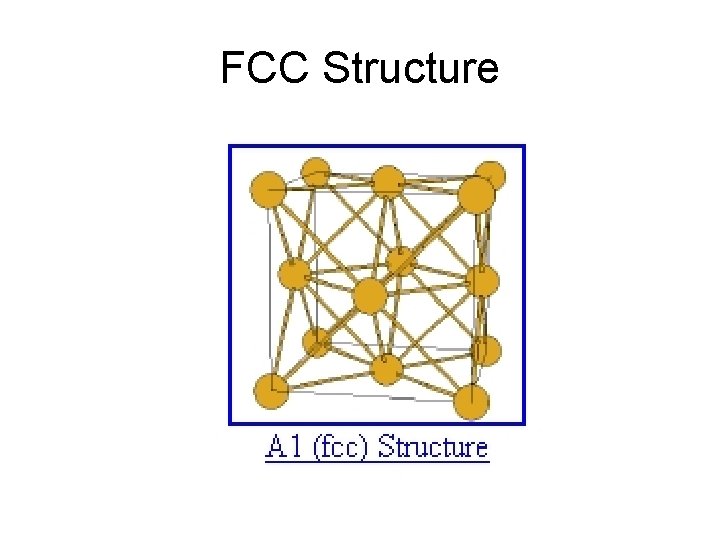
FCC Structure

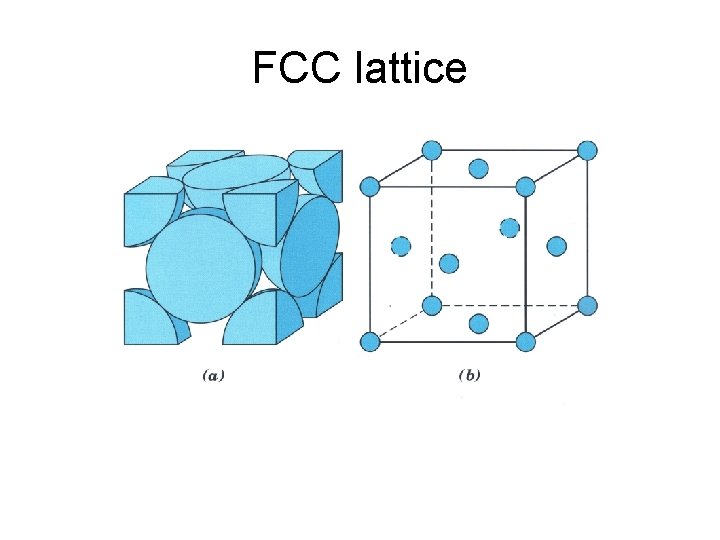
FCC lattice

Primitive Cell: FCC Lattice

FCC: Conventional Cell With Basis • We can also view the FCC lattice in terms of a conventional unit cell with a four point basis. • Similarly, we can view the BCC lattice in terms of a conventional unit cell with a two point basis.

Elements That Have FCC Structure

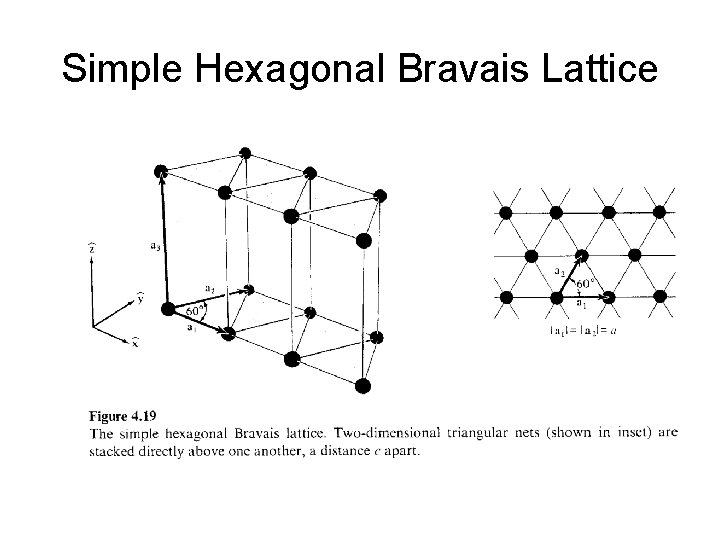
Simple Hexagonal Bravais Lattice

Primitive Cell: Hexagonal System

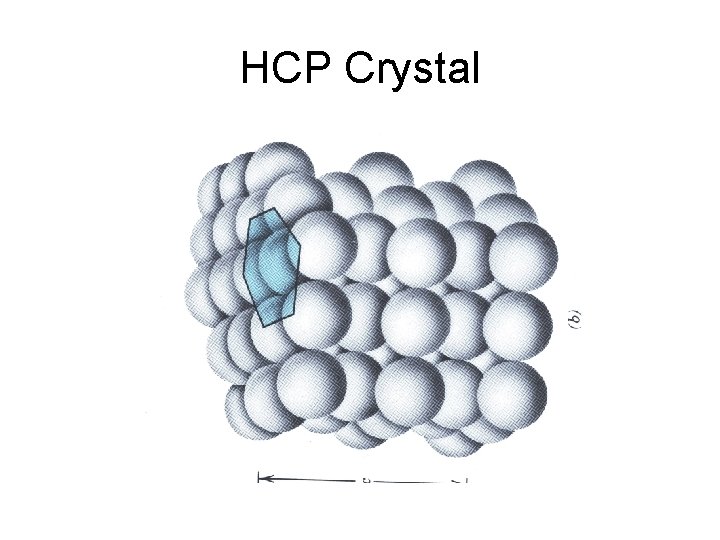
HCP Crystal

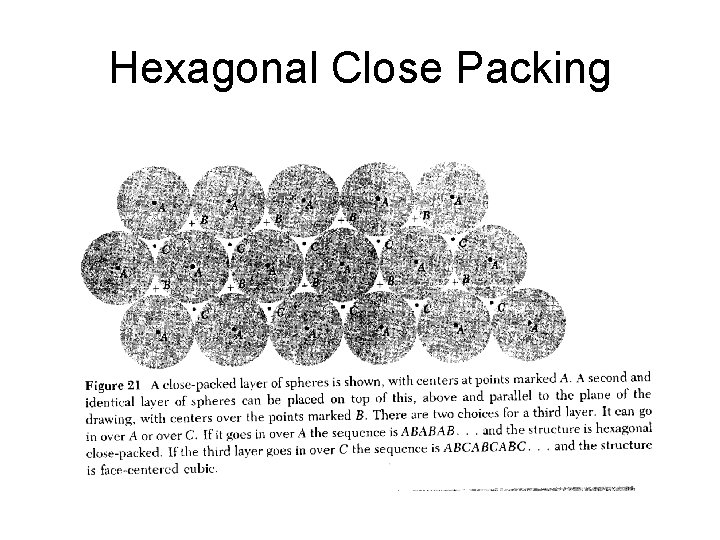
Hexagonal Close Packing

Hexagonal. Close. Packed HCP lattice is not a Bravais lattice, because orientation of the environment Of a point varies from layer to layer along the c-axis.

HCP: Simple Hexagonal Bravais With Basis of Two Atoms Per Point

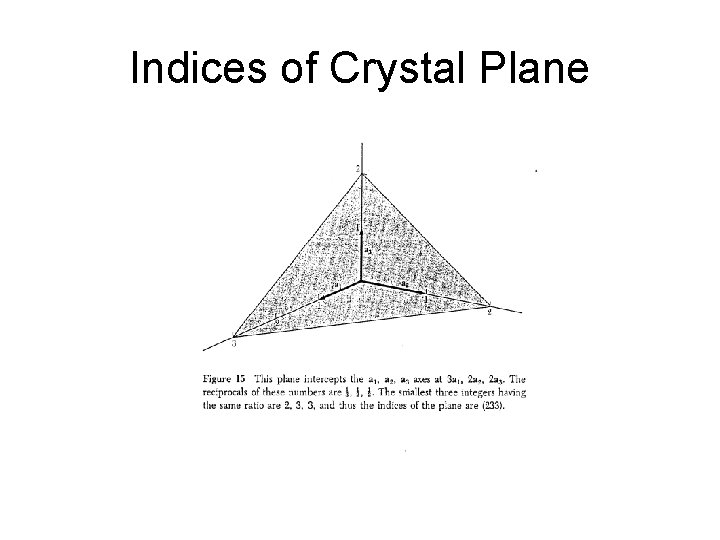
Miller indices of lattice plane • The indices of a crystal plane (h, k, l) are defined to be a set of integers with no common factors, inversely proportional to the intercepts of the crystal plane along the crystal axes:

Indices of Crystal Plane

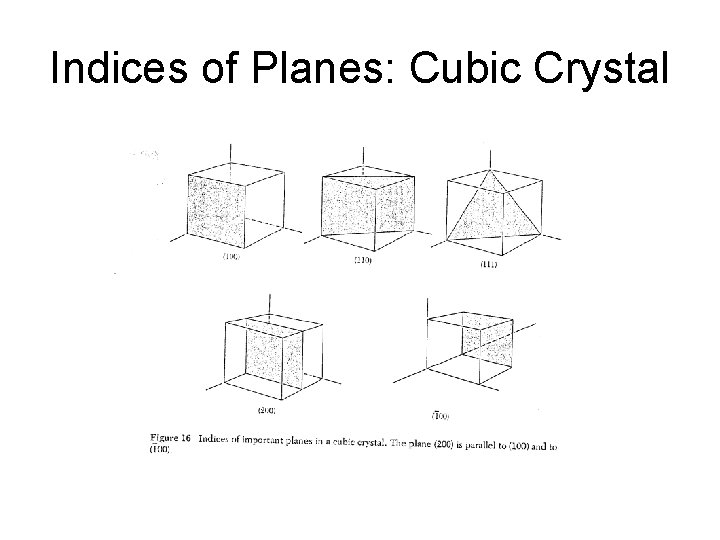
Indices of Planes: Cubic Crystal

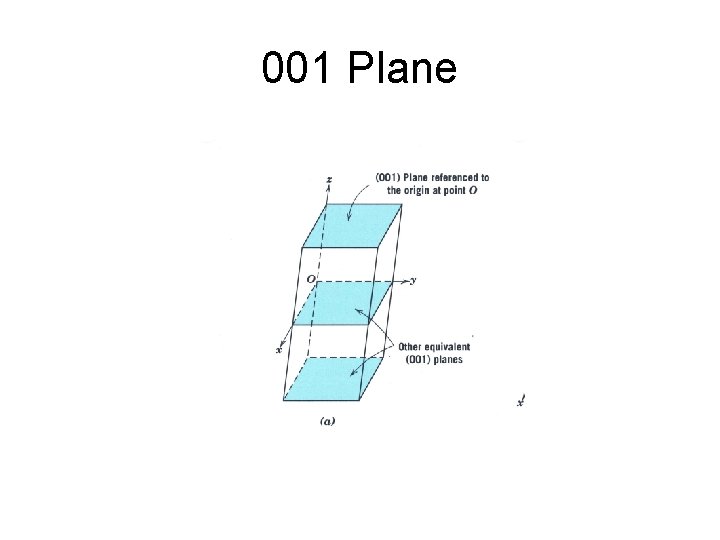
001 Plane

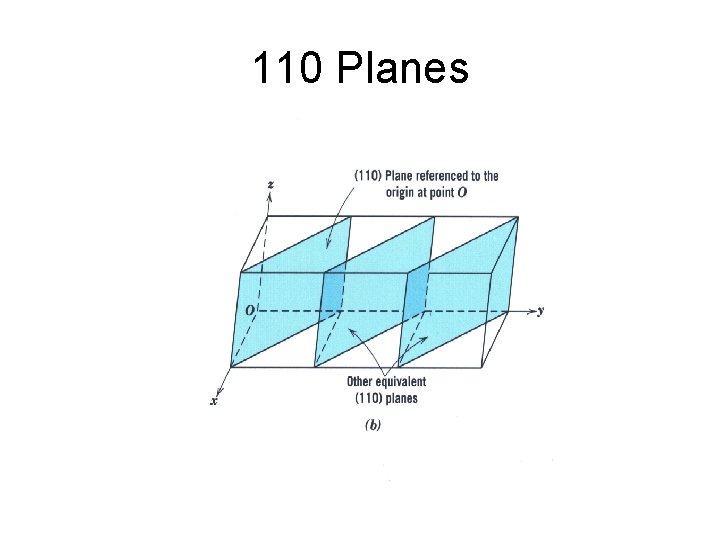
110 Planes

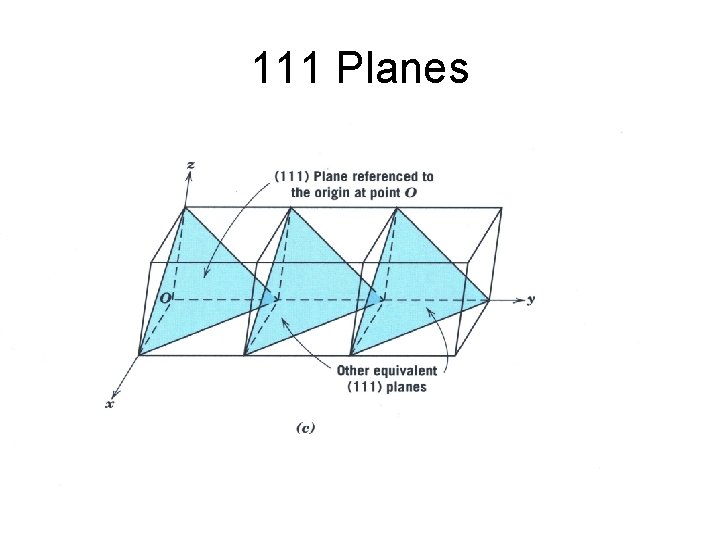
111 Planes

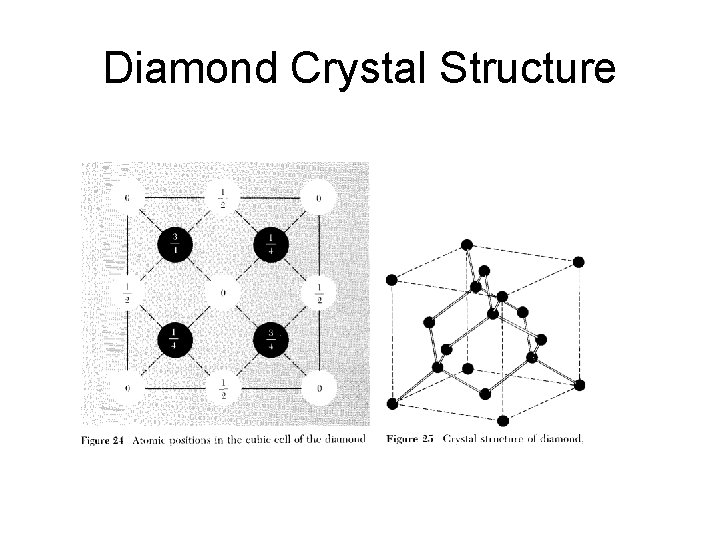
Simple Crystal Structures • There are several crystal structures of common interest: sodium chloride, cesium chloride, hexagonal close-packed, diamond and cubic zinc sulfide. • Each of these structures have many different realizations.

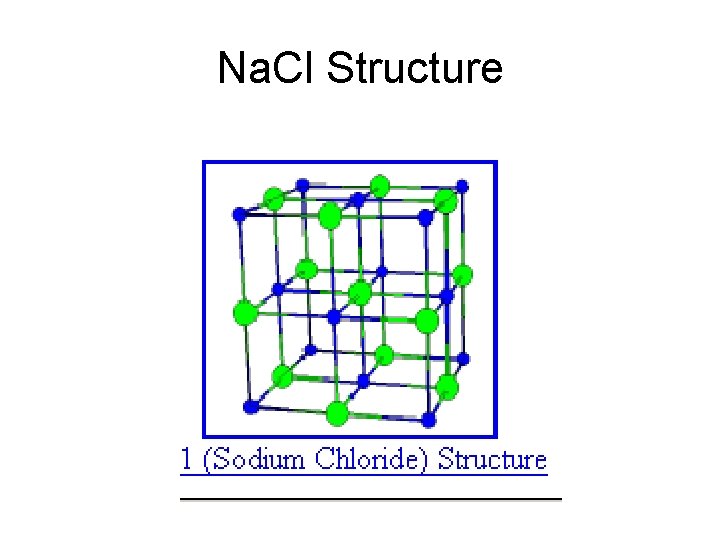
Na. Cl Structure

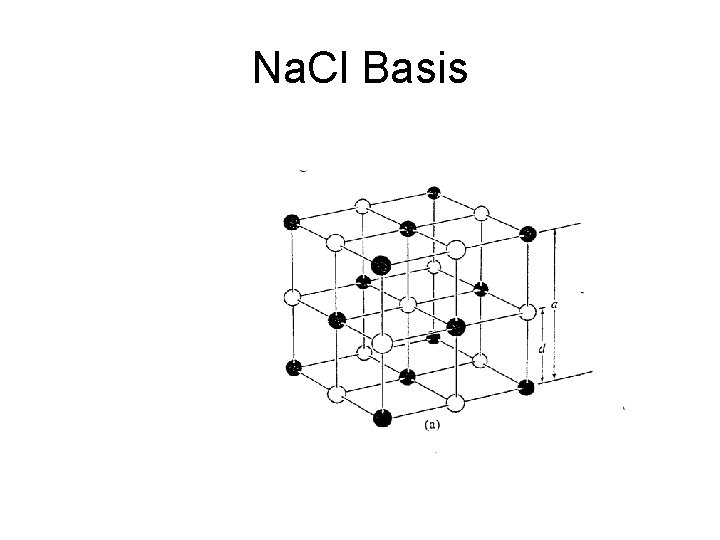
Na. Cl Basis

Na. Cl Type Elements

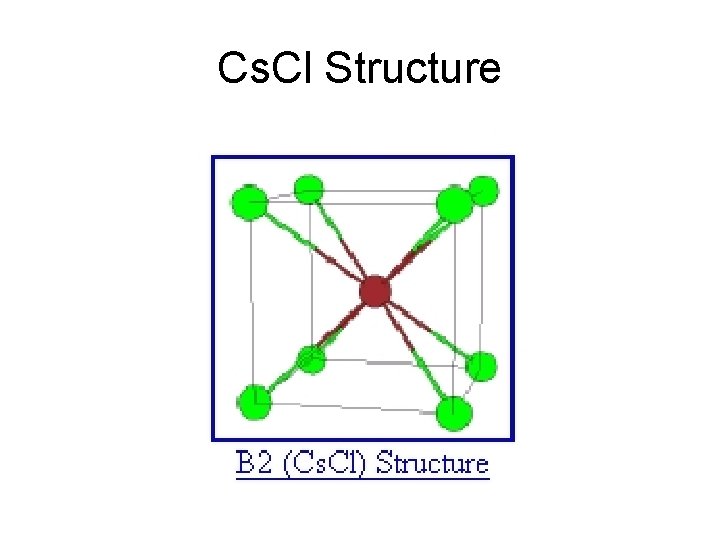
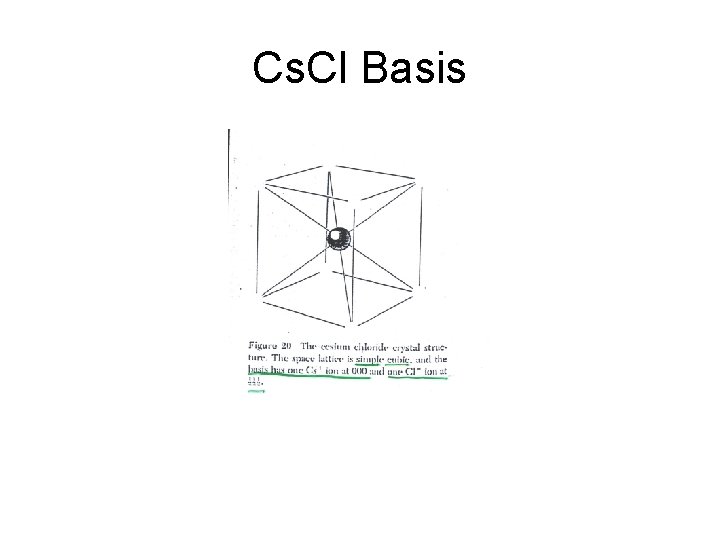
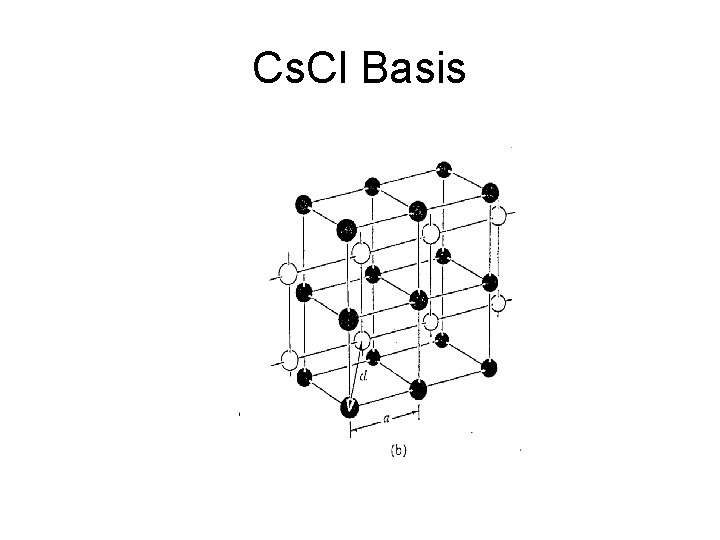
Cs. Cl Structure

Cs. Cl Basis

Cs. Cl Basis

Ce. Cl Crystals

Diamond Crystal Structure

Zinc. Blende structure

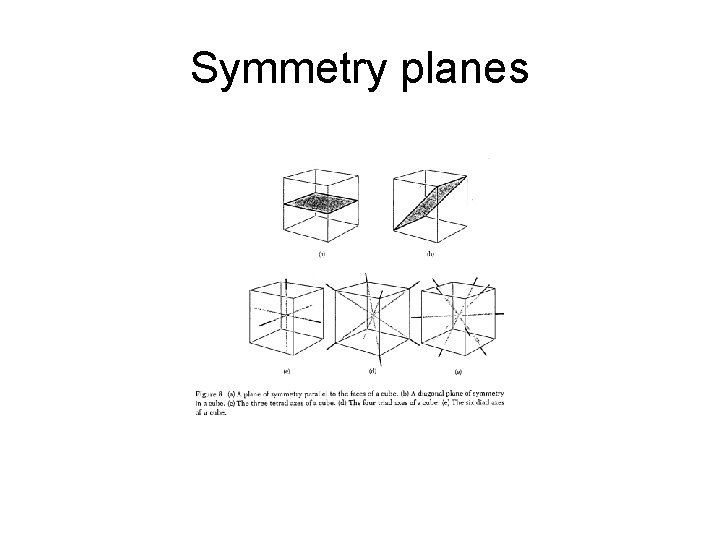
Symmetry planes

The End: Chapter 1


Bravais Lattice: Two Definitions The expansion coefficients n 1, n 2, n 3 must be integers. The vectors a 1, a 2, a 3 are primitive vectors and span the lattice.

HCP Close Packing

HCP Close Packing

Close Packing 2

Close Packing 3

Close Packing 4

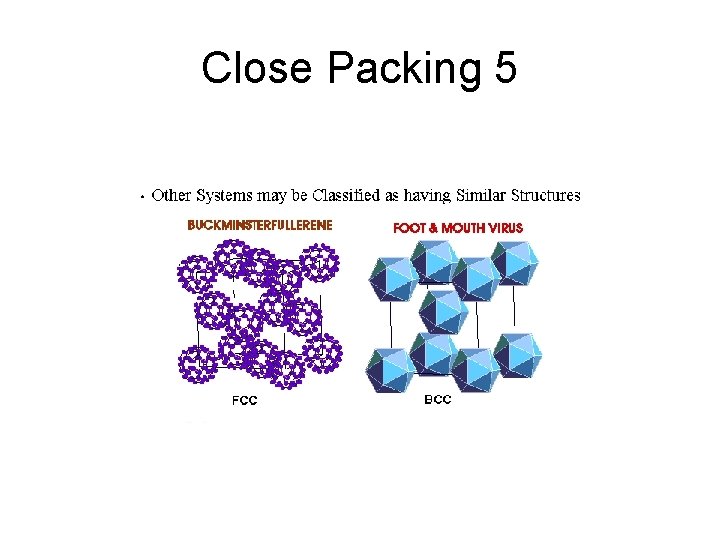
Close Packing 5

Na. Cl Basis

Close Packing of Spheres
