Coulombs Law Credit Wikimedia user The Elementary Concept

Coulomb’s Law Credit: Wikimedia user
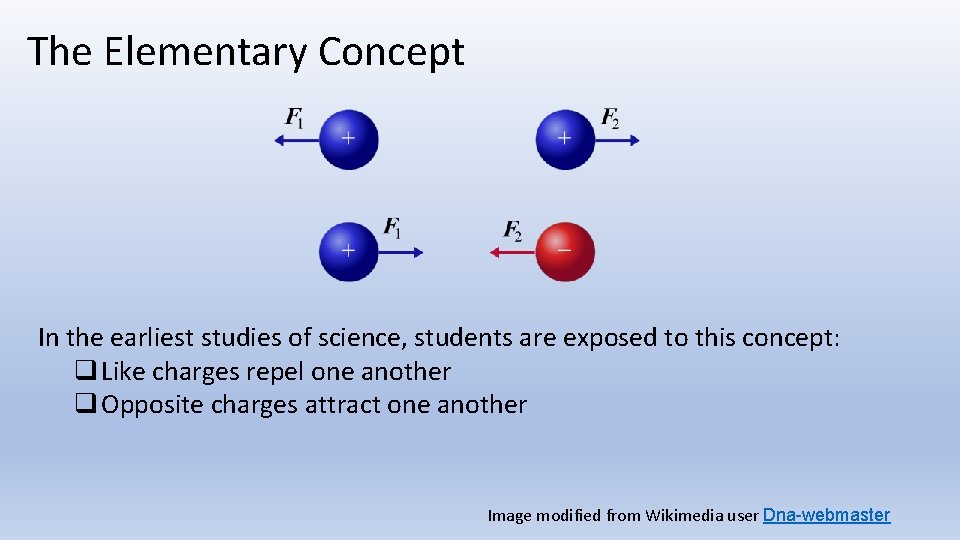
The Elementary Concept In the earliest studies of science, students are exposed to this concept: q Like charges repel one another q Opposite charges attract one another Image modified from Wikimedia user Dna-webmaster
Importance of the Concept These forces are at the heart of chemical processes such as q Chemical bonding q Ionization energy q Solubility q Acid and base behavior Because of this, the subject must be understood at greater depth than the simple “attract” and “repel” statements Image modified from Wikimedia user Dna-webmaster

Coulomb’s Law q q 1 and q 2 are the charges on the particles q r is the distance between their centers This means that 1. Greater charge = Greater Force 2. Smaller r = Greater Force Credit: Wikimedia user Dna-webmaster
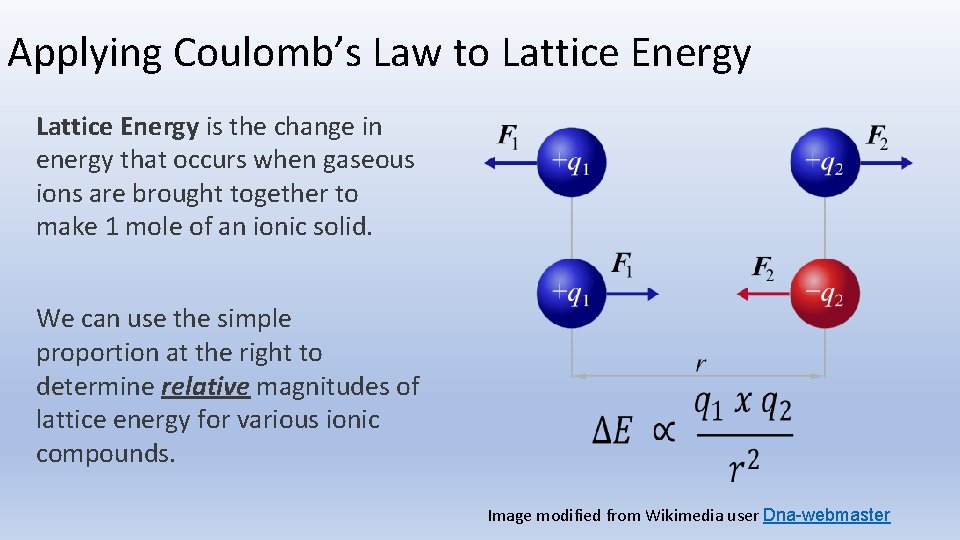
Applying Coulomb’s Law to Lattice Energy is the change in energy that occurs when gaseous ions are brought together to make 1 mole of an ionic solid. We can use the simple proportion at the right to determine relative magnitudes of lattice energy for various ionic compounds. Image modified from Wikimedia user Dna-webmaster

Example #1: Which compound is expected to have the greater lattice energy? Na. Cl or Mg. Cl 2 For Na 1+Cl 1 -: q 1 x q 2 = (+1)(-1) = -1 For Mg 2+Cl 21 -: q 1 x q 2 = (+2)(-1) = -2 Negative value means “exothermic” – the product solid is more stable than the gaseous ions Because -2 is greater than -1, Mg. Cl 2 is expected to have the greater lattice energy. Differences in radius cannot overcome the two-fold difference in the numerator.

Example #1: Which compound is expected to have the greater lattice energy? Na. Cl or Mg. Cl 2 For Na 1+Cl 1 -: q 1 x q 2 = (+1)(-1) = -1 For Mg 2+Cl 21 -: q 1 x q 2 = (+2)(-1) = -2 Negative value means “exothermic” – the product solid is more stable than the gaseous ions Notice that we have NOT calculated lattice energy. We have only used a simple proportion to determine RELATIVE magnitudes.

Example #2: Which compound is expected to have the greater lattice energy? Mg. Cl 2 or Ca. O For Mg 2+Cl 21 -: q 1 x q 2 = (+2)(-1) = -2 For Ca 2+O 2 -: q 1 x q 2 = (+2)(-2) = -4 Because -4 is greater than -2, Ca. O is expected to have the greater lattice energy. Differences in radius cannot overcome the two-fold difference in the numerator.

Example #3: Which compound is expected to have the greater lattice energy? Na. Cl or KCl Clearly, for both, the product of q 1 and q 2 is -1. The radius becomes the tiebreaker. K, potassium, is below sodium, Na in Group 1 on the periodic table. As a result, a potassium ion has a greater radius than a sodium ion. A larger denominator means a smaller overall value, so Na. Cl has the greater lattice energy.

Example #4: Which compound is expected to have the greater lattice energy? Na. Cl or Li. F Clearly, for both, the product of q 1 and q 2 is -1. The radius becomes the tiebreaker. Na 1+ has a larger radius than Li 1+, and Cl 1 - has a larger radius than F 1 - (if you don’t know why, review Periodic Behavior). A smaller denominator means a larger overall value, so Li. F has the greater lattice energy.

Example #5: Which compound is expected to have the greater lattice energy? Na 2 O or Li 2 S Clearly, for both, the product of q 1 and q 2 is -2. The radius becomes the tiebreaker. Na 1+ has a larger radius than Li 1+, BUT S 2 - has a larger radius than O 2 -. Without knowing actual values for the ionic radius, we cannot answer definitively. This problem does not lend itself to a qualitative solution.

Example #6: Use Coulomb’s Law to explain why Na. Cl is much more soluble in water than Ca. O (this is jumping ahead a bit, but illustrates the power of the concept to explain observable phenomena). § For Na. Cl, the product of q 1 and q 2 is -1. § For Ca. O, the product of q 1 and q 2 is -4. § Because Ca. O has a much larger lattice energy, it is much harder for water to break apart the ionic lattice of Ca. O. As a result, it does not dissolve readily.
- Slides: 12