Group 1 and 2 Nitrates And Carbonates The

Group 1 and 2 Nitrates And Carbonates

The s-block elements • The s-block elements all form salts when they react. • All compounds contain M+ and M 2+ cations. • Various factors determine the stability of these (ionic) compounds. – Main one is lattice energy – which is determined by two factors – What are they?

Lattice energy trends • One is distance between the centre of the cation and the anion. • The other is charge on the cation.

General stability rule of thumb • Stability of an ionic compound increases as: Cation radius decreases The charge on the ion increases
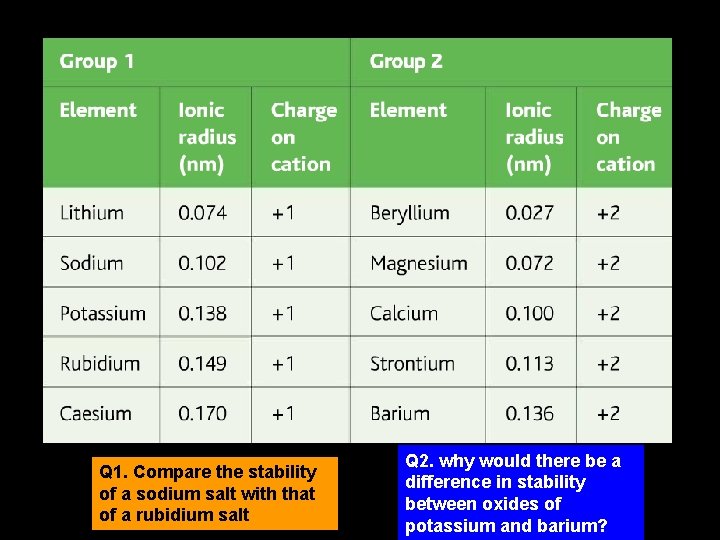
Q 1. Compare the stability of a sodium salt with that of a rubidium salt Q 2. why would there be a difference in stability between oxides of potassium and barium?

Thermal stability of nitrates and carbonates • When talking stability, you don’t only think about stability of a salt in relation to its elements, but also in relation to other compounds that may be formed. • For example: – The carbonates (CO 32 -) anion may also decompose to form carbon dioxide and the oxide anion: • E. g. Ca. CO 3(s) → Ca. O(s) + CO 2(g) – The nitrate (NO 3 -) anion may decompose to form an oxide ion too, or the larger nitrate anion. • E. g. 2 Mg(NO 3)2(s) → 2 Mg. O(s) + 4 NO 2(g) + O 2(g) • E. g. 2 KNO 3(s) → 2 KNO 2(s) + O 2(g) http: //www. chemguide. co. uk/inorganic/group 2/thermstab. html

Lattice energy trends • As you travel down the group 2, for example, the lattice energies of both carbonates and oxides becomes less negative. • However, not at the same rate – the oxide value falls faster than that of the carbonate. • The value of overall enthalpy change for decomposition reaction becomes more positive as you go down as a result:

Lattice energy trends • Why do we think the lattice energy of the carbonates falls and oxides falls at different rates? • Oxide ion is smaller (0. 140 nm) than the carbonate ion. • As you go down from Mg to Ca, the distance increases from 0. 212 nm to 0. 240 nm (13%). • For the carbonate ion, distance would increase by same amount – as a percentage the distance increase will be less.

Thermal Stability of Nitrates • Group 1 Nitrates (except Li): • Decompose to form nitrites and oxygen: • This is because NO 2 - ion is smaller than NO 3 - ion; so lattice of (Na+ NO 2 -) is more stable than of (Na+ NO 3 -). • Considering the small size of the group 1 cations, this is sufficient to achieve thermal stability and avoid further decomposition to the oxide.

Thermal Stability of Nitrates • Group 2 Nitrates (and Li): • Decompose to form the metal oxide, nitrogen dioxide and oxygen: • This is because the O 2 - ion is smaller and more highly charged than the NO 3 - ion. • The smaller cations needing a much smaller anion than the nitrite one to give them stability.

Thermal Stability of Carbonates • Carbonates of Group 1 are thermally stable: – Exception is lithium carbonate which decomposes to give the oxide: • All group 2 carbonates decompose to form stable oxides. E. g.
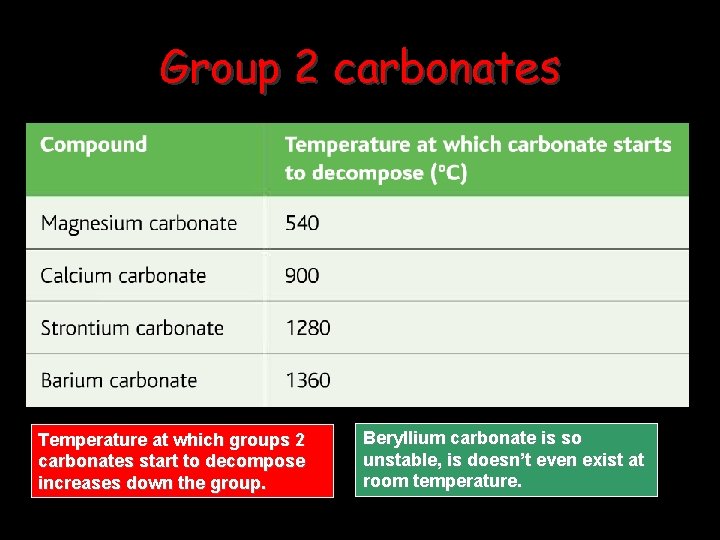
Group 2 carbonates Temperature at which groups 2 carbonates start to decompose increases down the group. Beryllium carbonate is so unstable, is doesn’t even exist at room temperature.

Thermal Stability of Hydroxides. • These follow same pattern as carbonates and nitrates. • The group 1 hydroxides are stable to quite high temperatures, with lithium hydroxide first to decompose at 650°C. • Group 2 hydroxides all decompose to give the oxide and water:

Questions

Answers
- Slides: 15