Lattice Energy Properties affected by bond strength meltingboiling

Lattice Energy

Properties affected by bond strength • melting/boiling point (stronger bonds = higher melting/boiling point)

Properties affected by bond strength • melting/boiling point (stronger bonds = higher melting point/boiling point) • hardness (stronger bonds = harder crystals)

Properties affected by bond strength • melting/boiling point (stronger bonds = higher melting point/boiling point) • hardness (stronger bonds = harder crystals) • odor (stronger bonds = weaker odor)

Properties affected by bond strength • melting/boiling point (stronger bonds = higher melting point/boiling point) • hardness (stronger bonds = harder crystals) • odor (stronger bonds = weaker odor) • state at room temperature (stronger bonds are more likely to be solids)

Bond formation • Whenever a new bond forms, energy is released. The stronger the bond, the more energy that is released.

Bond formation • Whenever a new bond forms, energy is released. The stronger the bond, the more energy that is released. • For ionic bonds, this energy is called lattice energy.

Bond formation • Whenever a new bond forms, energy is released. The stronger the bond, the more energy that is released. • For ionic bonds, this energy is called lattice energy. • Higher lattice energy values correspond to stronger bonds.

Bond formation • Whenever a new bond forms, energy is released. The stronger the bond, the more energy that is released. • For ionic bonds, this energy is called lattice energy. • Higher lattice energy values correspond to stronger bonds. • The amount of energy released when a bond forms is identical to the amount of energy required to break the bond.

Predicting lattice energy • Two factors will determine the lattice energy for ionic compounds.

Predicting lattice energy • Two factors will determine the lattice energy for ionic compounds. – Bigger charges produce higher lattice energies (2+ and 2 - produce stronger bonds than 1+ and 1 -)

Predicting lattice energy • Two factors will determine the lattice energy for ionic compounds. – Bigger charges produce higher lattice energies (2+ and 2 - produce stronger bonds than 1+ and 1 -) – Fewer energy levels result in greater lattice energies (Li. F will have a greater lattice energy than Na. Cl)

Predicting lattice energy • Two factors will determine the lattice energy for ionic compounds. – Bigger charges produce higher lattice energies (2+ and 2 - produce stronger bonds than 1+ and 1 -) – Fewer energy levels result in greater lattice energies (Li. F will have a greater lattice energy than Na. Cl) – Charge is by far the bigger factor and should always be compared first!!!
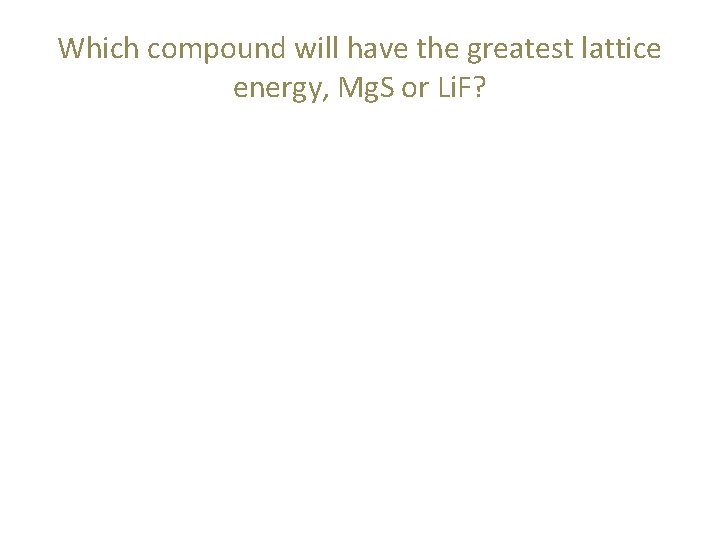
Which compound will have the greatest lattice energy, Mg. S or Li. F?

Which compound will have the greatest lattice energy, Mg. S or Li. F? • Magnitude of charge is the first thing we should look at.

Which compound will have the greatest lattice energy, Mg. S or Li. F? • Magnitude of charge is the first thing we should look at. – magnesium (2+ charge), sulfide (2 - charge)

Which compound will have the greatest lattice energy, Mg. S or Li. F? • Magnitude of charge is the first thing we should look at. – magnesium (2+ charge), sulfide (2 - charge) – lithium (1+ charge), fluoride (1 - charge)

Which compound will have the greatest lattice energy, Mg. S or Li. F? • Magnitude of charge is the first thing we should look at. – magnesium (2+ charge), sulfide (2 - charge) – lithium (1+ charge), fluoride (1 - charge) – Mg. S has the greater individual charges, so Mg. S has the greater lattice energy.

Which compound will have a lower melting point, Na 2 S or Be. O?

Which compound will have a lower melting point, Na 2 S or Be. O? • A lower melting point means we need to select the compound with the lower lattice energy. We check the charges first.

Which compound will have a lower melting point, Na 2 S or Be. O? • A lower melting point means we need to select the compound with the lower lattice energy. We check the charges first. – sodium (1+), sulfide (2 -)

Which compound will have a lower melting point, Na 2 S or Be. O? • A lower melting point means we need to select the compound with the lower lattice energy. We check the charges first. – sodium (1+), sulfide (2 -) – beryllium (2+), oxide (2 -)

Which compound will have a lower melting point, Na 2 S or Be. O? • A lower melting point means we need to select the compound with the lower lattice energy. We check the charges first. – sodium (1+), sulfide (2 -) – beryllium (2+), oxide (2 -) – sodium sulfide has smaller individual charges, so it has the lower melting point.
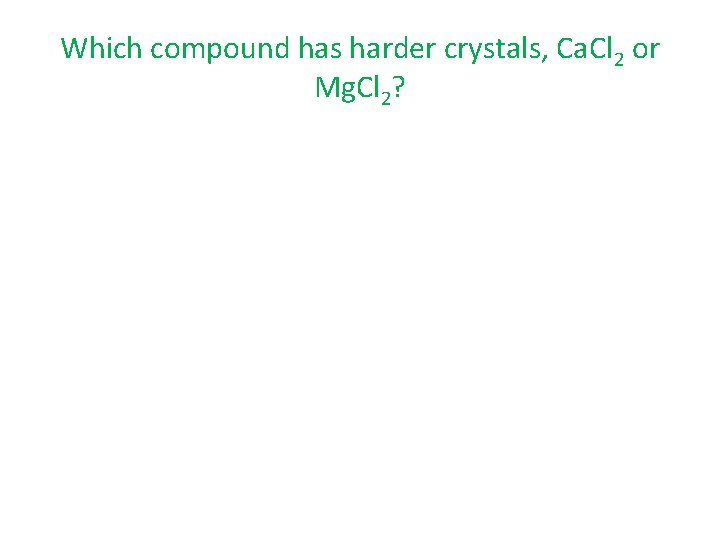
Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2?

Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2? • Harder crystals require a higher lattice energy. First, check the charges.

Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2? • Harder crystals require a higher lattice energy. First, check the charges. – calcium (2+), chloride (1 -)

Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2? • Harder crystals require a higher lattice energy. First, check the charges. – calcium (2+), chloride (1 -) – magnesium (2+), chloride (1 -)

Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2? • Harder crystals require a higher lattice energy. First, check the charges. – calcium (2+), chloride (1 -) – magnesium (2+), chloride (1 -) – the charges are the same, so we need to see which ions have fewer energy levels.

Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2? – the charges are the same, so we need to see which ions have fewer energy levels. – calcium (4), chloride (3)

Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2? – the charges are the same, so we need to see which ions have fewer energy levels. – calcium (4), chloride (3) – magnesium (3), chloride (3)

Which compound has harder crystals, Ca. Cl 2 or Mg. Cl 2? – the charges are the same, so we need to see which ions have fewer energy levels. – calcium (4), chloride (3) – magnesium (3), chloride (3) – fewer energy levels give Mg. Cl 2 the higher lattice energy and therefore, the harder crystals.

Which compound has the lower boiling point, Ag. NO 3 or K 2 SO 4?

Which compound has the lower boiling point, Ag. NO 3 or K 2 SO 4? • Lower boiling point means a lower lattice energy.

Which compound has the lower boiling point, Ag. NO 3 or K 2 SO 4? • Lower boiling point means a lower lattice energy. – silver (1+), nitrate (1 -)

Which compound has the lower boiling point, Ag. NO 3 or K 2 SO 4? • Lower boiling point means a lower lattice energy. – silver (1+), nitrate (1 -) – potassium (1+), sulfate (2 -)

Which compound has the lower boiling point, Ag. NO 3 or K 2 SO 4? • Lower boiling point means a lower lattice energy. – silver (1+), nitrate (1 -) – potassium (1+), sulfate (2 -) – Charges indicate a lower lattice energy for Ag. NO 3. Lower lattice energy means a lower boiling point.
- Slides: 36