Chapter 2 Atoms Molecules and Ions Early History









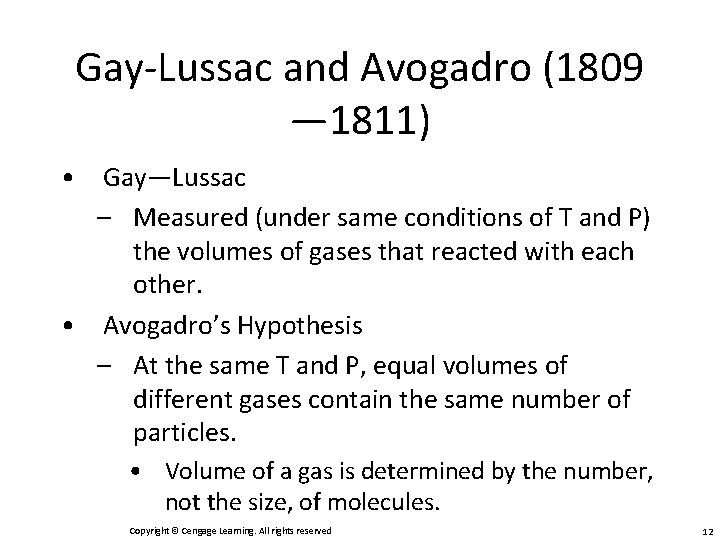


















































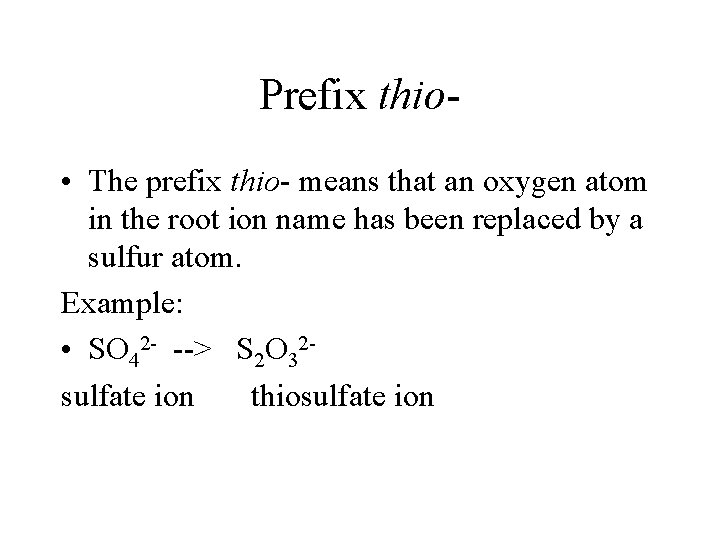






















- Slides: 110

Chapter 2 Atoms, Molecules, and Ions

Early History of Chemistry • Greeks were the first to attempt to explain why chemical changes occur. • Alchemy dominated for 2000 years. – Several elements discovered. – Mineral acids prepared. • Robert Boyle was the first “chemist”. – Performed quantitative experiments. – Developed first experimental definition of an element. Copyright © Cengage Learning. All rights reserved 2

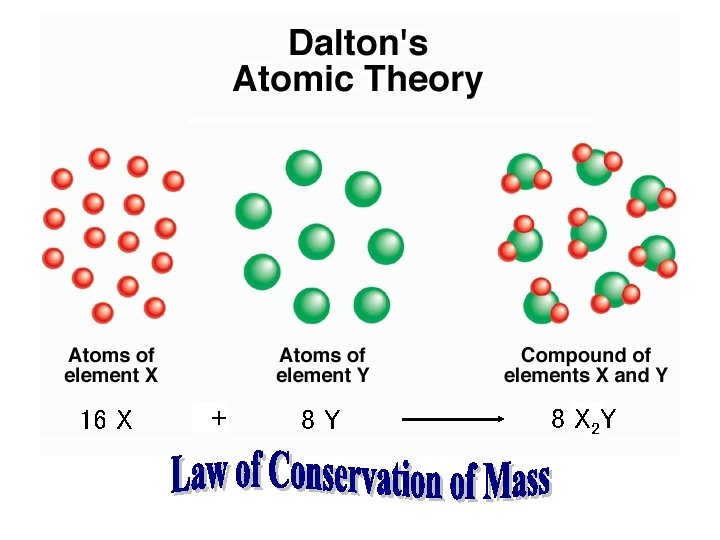
Three Important Laws • Law of conservation of mass (Lavoisier): – Mass is neither created nor destroyed in a chemical reaction. • Law of definite proportion (Proust): – A given compound always contains exactly the same proportion of elements by mass. Copyright © Cengage Learning. All rights reserved 3

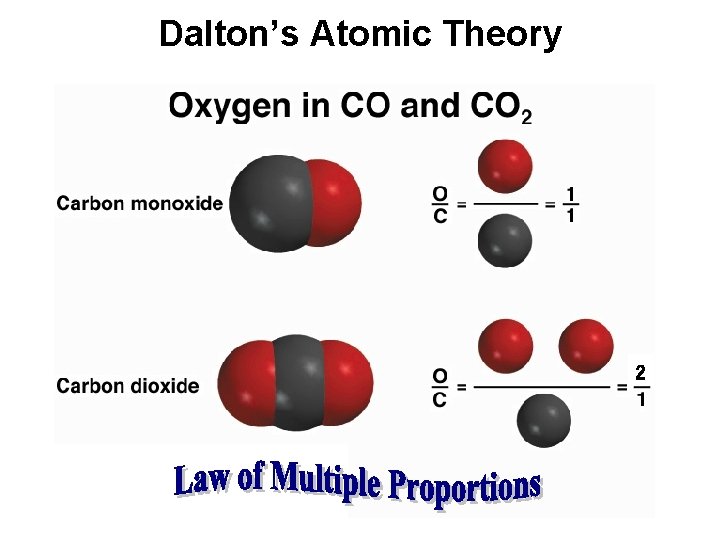
Three Important Laws (continued) • Law of multiple proportions (Dalton): – When two elements form a series of compounds, the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole numbers. Copyright © Cengage Learning. All rights reserved 4

Composition of the Atom • Understanding the structure of the atom will help to understand the properties of the elements. • Keep in mind that these, as all theories, are subject to constant refinement. The picture of the atom isn’t final.

Development of the Atomic Theory • Dalton’s Atomic Theory - the first experimentally based theory of atomic structure of the atom. – John Dalton – early 1800’s • Much of Dalton’s Theory is still regarded as correct today. *See starred items. *

Postulates of Dalton’s Atomic Theory 1. All matter consists of tiny particles called atoms. * 2. An atom cannot be created, divided, destroyed, or converted to any other type of atom. 3. Atoms of a particular element have identical properties.

4. Atoms of different elements have different properties. * 5. Atoms of different elements combine in simple whole-number ratios to produce compounds (stable aggregates of atoms. )* 6. Chemical change involves joining, separating, or rearranging atoms. * * These postulates are still regarded as true.

CONCEPT CHECK! Which of the following statements regarding Dalton’s atomic theory are still believed to be true? I. Elements are made of tiny particles called atoms. II. All atoms of a given element are identical. III. A given compound always has the same relative numbers and types of atoms. IV. Atoms are indestructible. Copyright © Cengage Learning. All rights reserved 9

Dalton’s Atomic Theory 2

16 X + 8 Y 8 X 2 Y
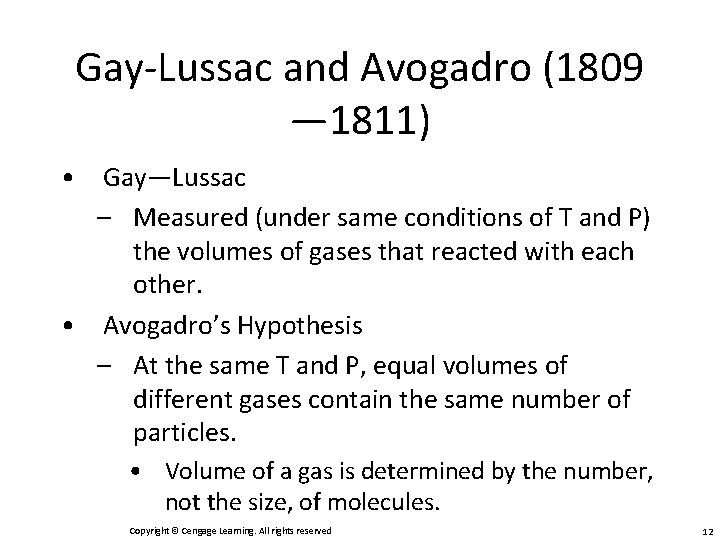
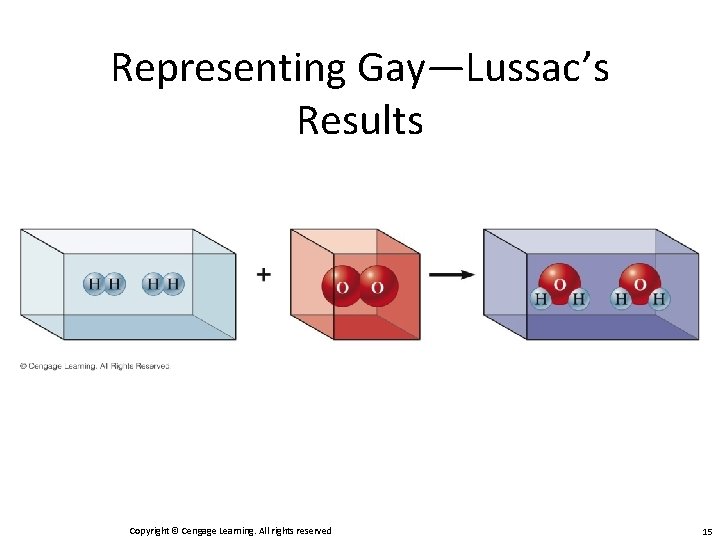
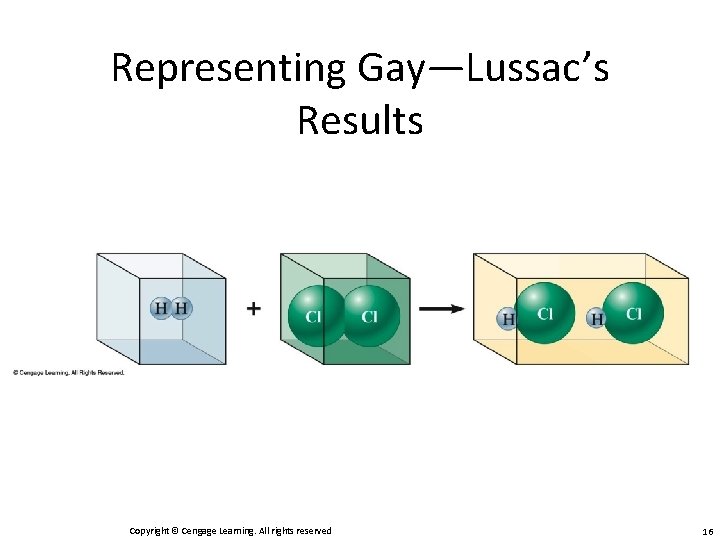
Gay-Lussac and Avogadro (1809 — 1811) • Gay—Lussac – Measured (under same conditions of T and P) the volumes of gases that reacted with each other. • Avogadro’s Hypothesis – At the same T and P, equal volumes of different gases contain the same number of particles. • Volume of a gas is determined by the number, not the size, of molecules. Copyright © Cengage Learning. All rights reserved 12

Representing Gay—Lussac’s Results Copyright © Cengage Learning. All rights reserved 13

Representing Gay—Lussac’s Results Copyright © Cengage Learning. All rights reserved 14

Representing Gay—Lussac’s Results Copyright © Cengage Learning. All rights reserved 15

Representing Gay—Lussac’s Results Copyright © Cengage Learning. All rights reserved 16

We will learn that atoms consist of three primary particles. electrons protons neutrons • Nucleus - small, dense, positively charged region in the center of the atom. Contains: - protons - positively charged particles - neutrons - uncharged particles • Surrounding the nucleus is a diffuse region of negative charge populated by: - electrons - negatively charged particles

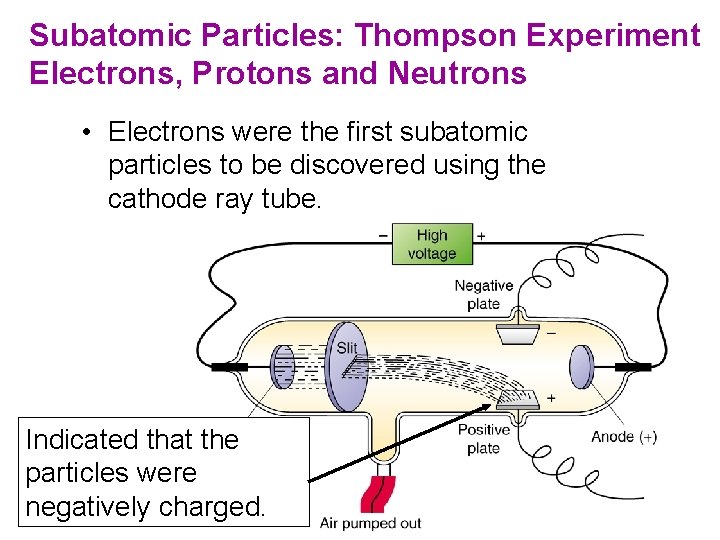
J. J. Thomson (1898— 1903) • Postulated the existence of negatively charged particles, that we now call electrons, using cathode-ray tubes. • Determined the charge-to-mass ratio of an electron. • The atom must also contain positive particles that balance exactly the negative charge carried by electrons. Copyright © Cengage Learning. All rights reserved 18

Subatomic Particles: Thompson Experiment Electrons, Protons and Neutrons • Electrons were the first subatomic particles to be discovered using the cathode ray tube. Indicated that the particles were negatively charged.

Cathode Ray Tube

Joseph John Thomson (1856 -1940). Photo courtesy of The Cavendish Laboratory.

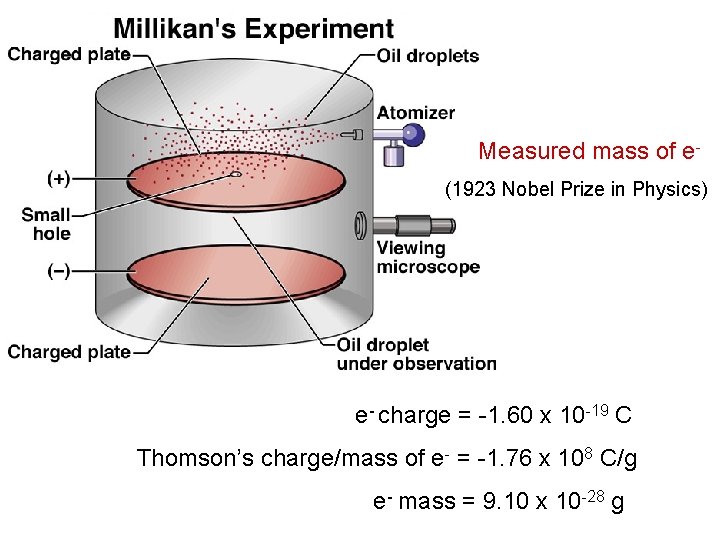
Robert Millikan (1909) • Performed experiments involving charged oil drops. • Determined the magnitude of the charge on a single electron. • Calculated the mass of the electron – (9. 11 × 10 -31 kg). Copyright © Cengage Learning. All rights reserved 22

Measured mass of e(1923 Nobel Prize in Physics) e- charge = -1. 60 x 10 -19 C Thomson’s charge/mass of e- = -1. 76 x 108 C/g e- mass = 9. 10 x 10 -28 g

Millikan Oil Drop Experiment To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 24

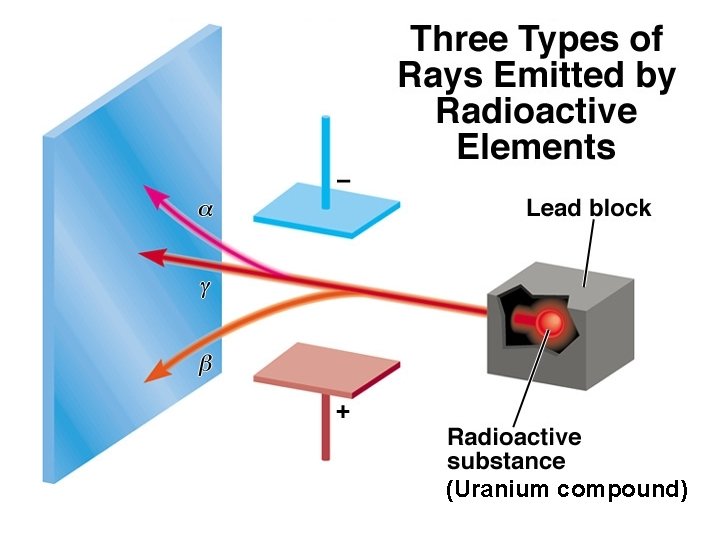
Henri Becquerel (1896) • Discovered radioactivity by observing the spontaneous emission of radiation by uranium. • Three types of radioactive emission exist: – Gamma rays (ϒ) – high energy light – Beta particles (β) – a high speed electron – Alpha particles (α) – a particle with a 2+ charge Copyright © Cengage Learning. All rights reserved 25

(Uranium compound)


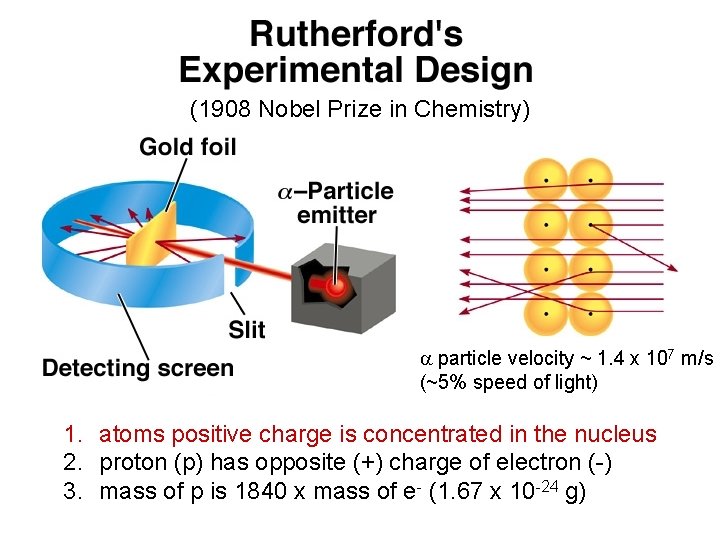
Ernest Rutherford (1911) • Explained the nuclear atom. • The atom has a dense center of positive charge called the nucleus. • Electrons travel around the nucleus at a large distance relative to the nucleus. Copyright © Cengage Learning. All rights reserved 28

(1908 Nobel Prize in Chemistry) a particle velocity ~ 1. 4 x 107 m/s (~5% speed of light) 1. atoms positive charge is concentrated in the nucleus 2. proton (p) has opposite (+) charge of electron (-) 3. mass of p is 1840 x mass of e- (1. 67 x 10 -24 g)

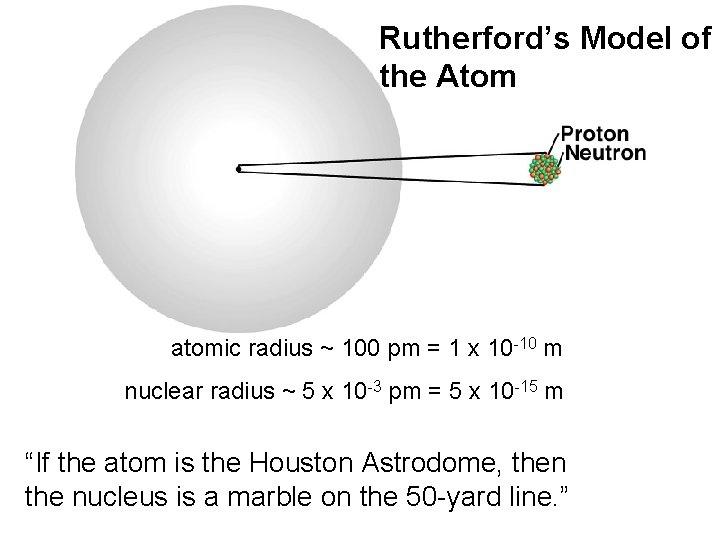
Rutherford’s Model of the Atom atomic radius ~ 100 pm = 1 x 10 -10 m nuclear radius ~ 5 x 10 -3 pm = 5 x 10 -15 m “If the atom is the Houston Astrodome, then the nucleus is a marble on the 50 -yard line. ”

• The nucleus is: – Small compared with the overall size of the atom. – Extremely dense; accounts for almost all of the atom’s mass. Copyright © Cengage Learning. All rights reserved 31

Nuclear Atom Viewed in Cross Section Copyright © Cengage Learning. All rights reserved

Discovery of the Proton • The proton was discovered in 1918 by Ernest Rutherford. He noticed that when alpha particles were shot into nitrogen gas, his scintillation detectors showed the signatures of hydrogen nuclei. Rutherford determined that the only place this hydrogen could have come from was the nitrogen, and therefore nitrogen must contain hydrogen nuclei. He thus suggested that the hydrogen nucleus, which was known to have an atomic number of 1, was an elementary particle. This he named proton, from protos, the Greek for "first".

Chadwick’s Experiment (1932) (1935 Noble Prize in Physics) H atoms - 1 p; He atoms - 2 p mass He/mass H should = 2 measured mass He/mass H = 4 a + 9 Be 1 n + 12 C + energy neutron (n) is neutral (charge = 0) n mass ~ p mass = 1. 67 x 10 -24 g

Atom constitution summary • The atom contains: – Electrons – found outside the nucleus; negatively charged. – Protons – found in the nucleus; positive charge equal in magnitude to the electron’s negative charge. – Neutrons – found in the nucleus; no charge; virtually same mass as a proton. Copyright © Cengage Learning. All rights reserved 35

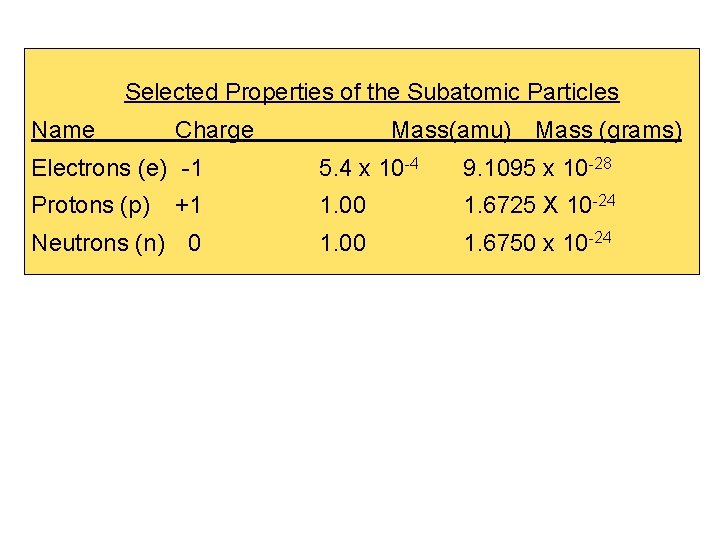
Selected Properties of the Subatomic Particles Name Charge Mass(amu) Mass (grams) Electrons (e) -1 5. 4 x 10 -4 9. 1095 x 10 -28 Protons (p) +1 1. 00 1. 6725 X 10 -24 Neutrons (n) 0 1. 00 1. 6750 x 10 -24

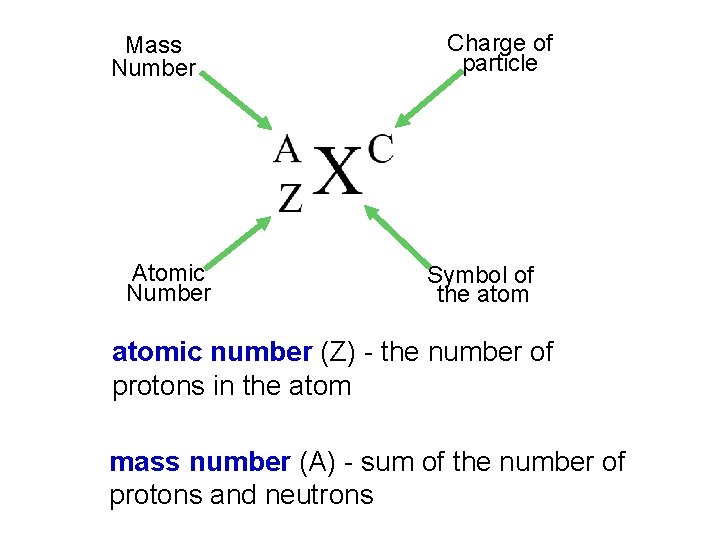
Atomic number, Mass number and Isotopes Atomic number (Z) = number of protons in nucleus Mass number (A) = number of protons + number of neutrons = atomic number (Z) + number of neutrons Isotopes are atoms of the same element (X) with different numbers of neutrons in their nuclei Mass Number A ZX Atomic Number 1 1 H 235 92 2 1 H U Element Symbol (D) 238 92 3 1 H U (T)

Mass Number Atomic Number Charge of particle Symbol of the atomic number (Z) - the number of protons in the atom mass number (A) - sum of the number of protons and neutrons

Calculate the number of protons, neutrons and electrons in each of the following:

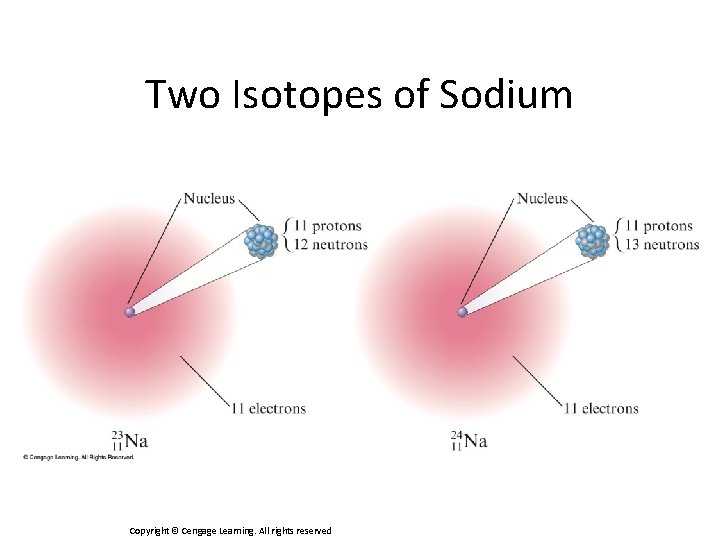
Isotopes • Atoms with the same number of protons but different numbers of neutrons. • Show almost identical chemical properties; chemistry of atom is due to its electrons. • In nature most elements contain mixtures of isotopes. Copyright © Cengage Learning. All rights reserved 40

Two Isotopes of Sodium Copyright © Cengage Learning. All rights reserved

• Isotopes - atoms of the same element having different masses. – contain same number of protons – contain different numbers of neutrons Isotopes of Hydrogen (Hydrogen 1) Tritium Deuterium (Hydrogen - 2) (Hydrogen - 3)

Do You Understand Isotopes? How many protons, neutrons, and electrons are 14 in 6 C ? 6 protons, 8 (14 - 6) neutrons, 6 electrons How many protons, neutrons, and electrons are 11 in 6 C ? 6 protons, 5 (11 - 6) neutrons, 6 electrons

EXERCISE! A certain isotope X contains 23 protons and 28 neutrons. • What is the mass number of this isotope? • Identify the element. Mass Number = 51 Vanadium Copyright © Cengage Learning. All rights reserved 44

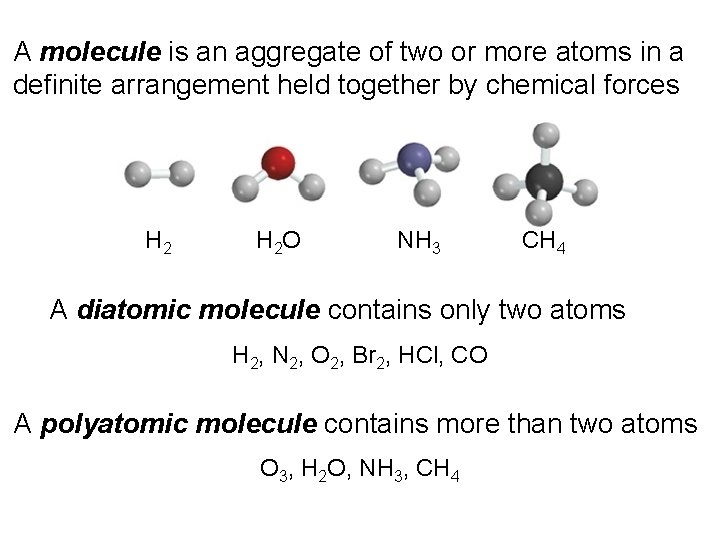
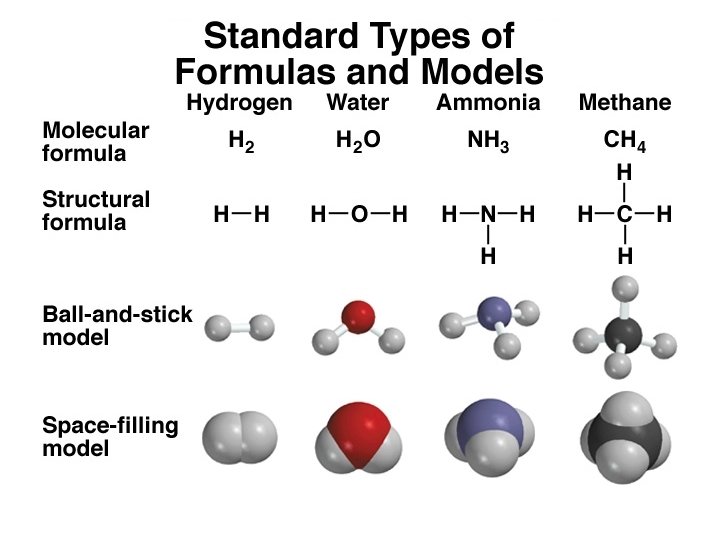
A molecule is an aggregate of two or more atoms in a definite arrangement held together by chemical forces H 2 O NH 3 CH 4 A diatomic molecule contains only two atoms H 2, N 2, O 2, Br 2, HCl, CO A polyatomic molecule contains more than two atoms O 3, H 2 O, NH 3, CH 4

Chemical Bonds • Covalent Bonds – Bonds form between atoms by sharing electrons. – Resulting collection of atoms is called a molecule. Copyright © Cengage Learning. All rights reserved 46

Covalent Bonding To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 47

Chemical Bonds • Ionic Bonds – Bonds form due to force of attraction between oppositely charged ions. – Ion – atom or group of atoms that has a net positive or negative charge. – Cation – positive ion; lost electron(s). – Anion – negative ion; gained electron(s). Copyright © Cengage Learning. All rights reserved 48

Chemical Formulas; Molecular and Ionic Substances • Ionic substances Although many substances are molecular, others are composed of ions. An ion is an electrically charged particle obtained from an atom or chemically bonded group of atoms by adding or removing electrons. Sodium chloride is a substance made up of ions.

An ion is an atom, or group of atoms, that has a net positive or negative charge. cation – ion with a positive charge If a neutral atom loses one or more electrons it becomes a cation. Na 11 protons 11 electrons Na+ 11 protons 10 electrons anion – ion with a negative charge If a neutral atom gains one or more electrons it becomes an anion. Cl 17 protons 17 electrons Cl- 17 protons 18 electrons

How many protons, neutrons and electrons are in the following ions?

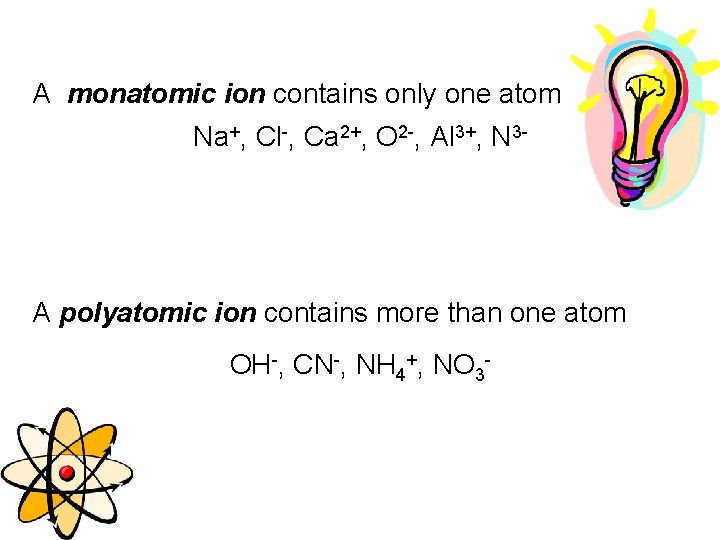
A monatomic ion contains only one atom Na+, Cl-, Ca 2+, O 2 -, Al 3+, N 3 - A polyatomic ion contains more than one atom OH-, CN-, NH 4+, NO 3 -

Do You Understand Ions? How many protons and electrons are in 27 3+ 13 Al ? 13 protons, 10 (13 – 3) electrons How many protons and electrons are in 78 2 Se ? 34 34 protons, 36 (34 + 2) electrons

Molecular vs Ionic Compounds To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 54

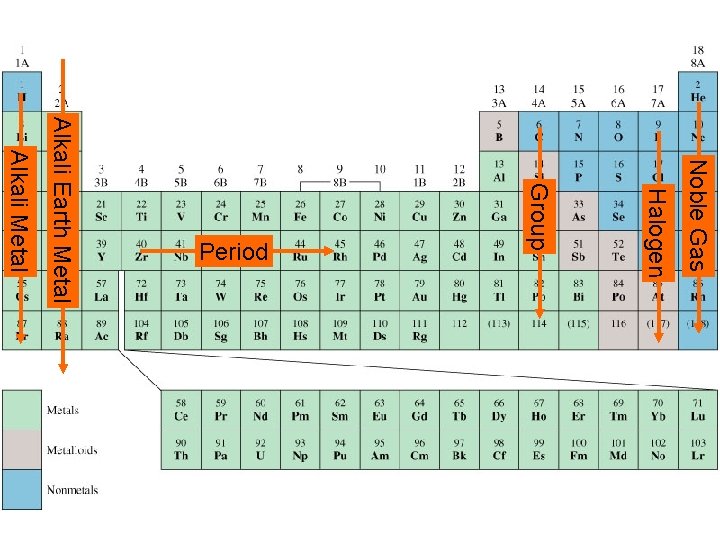
The Periodic Table • In 1869, Dmitri Mendeleev discovered that if the known elements were arranged in order of atomic number, they could be placed in horizontal rows such that the elements in the vertical columns had similar properties. A tabular arrangement of elements in rows and columns, highlighting the regular repetition of properties of the elements, is called a periodic table.

The Periodic Table • Periods and Groups A period consists of the elements in one horizontal role of the periodic table. A group consists of the elements in any one column of the periodic table. The groups are usually numbered. The eight groups are called main group (or representative) elements.

The Periodic Table • Periods and Groups The “B” groups are called transition elements. The two rows of elements at the bottom of the table are called inner transition elements. Elements in any one group have similar properties.

The Periodic Table • Periods and Groups The elements in group IA, often known as the alkali metals, are soft metals that react easily with water. The group VIIA elements, known as the halogens, are also reactive elements.

The Periodic Table • Metals, Nonmetals, and Metalloids A metal is a substance or mixture that has a characteristic luster and is generally a good conductor of heat and electricity. A nonmetal is an element that does not exhibit the characteristics of the metal. A metalloid, or semi-metal, is an element having both metallic and nonmetallic properties.

Noble Gas Halogen Group Alkali Metal Alkali Earth Metal Period

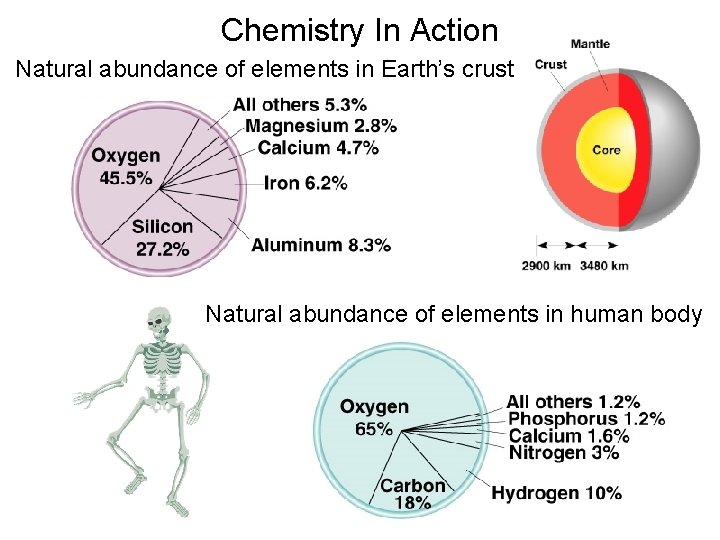
Chemistry In Action Natural abundance of elements in Earth’s crust Natural abundance of elements in human body


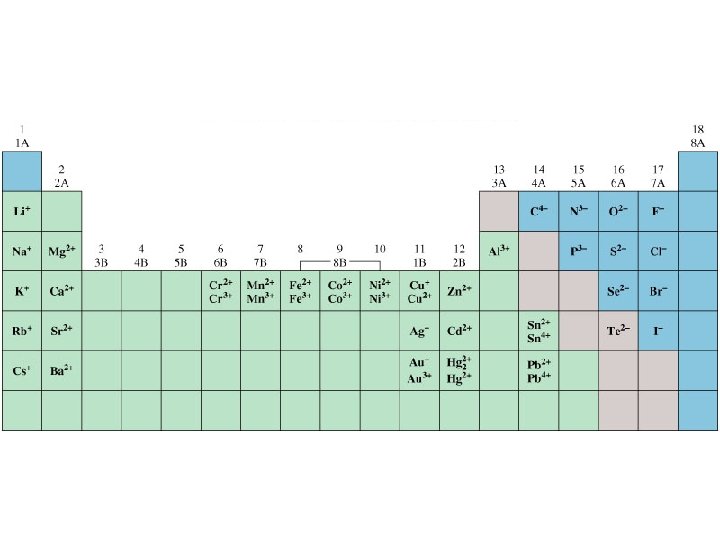
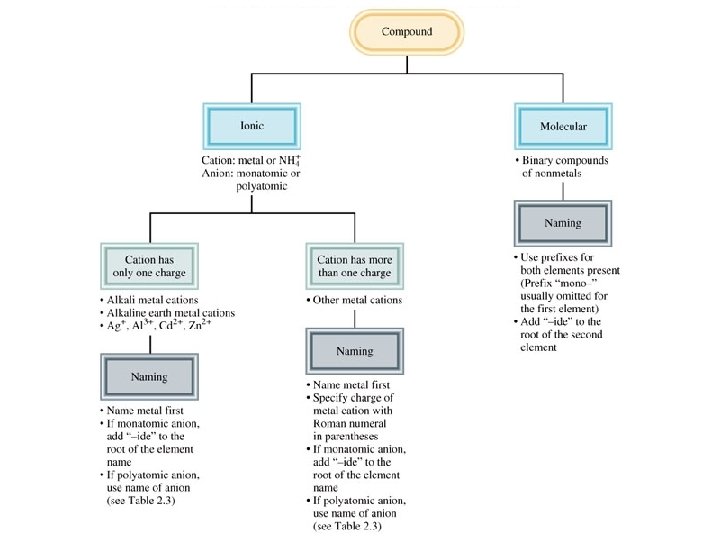
Chemical Substances; Formulas and Names A monatomic ion is an ion formed from a single atom.

Chemical Substances; Formulas and Names • Rules for predicting charges on monatomic ions Most of the main group metals form cations with the charge equal to their group number. The charge on a monatomic anion for a nonmetal equals the group number minus 8. Most transition elements form more than one ion, each with a different charge.

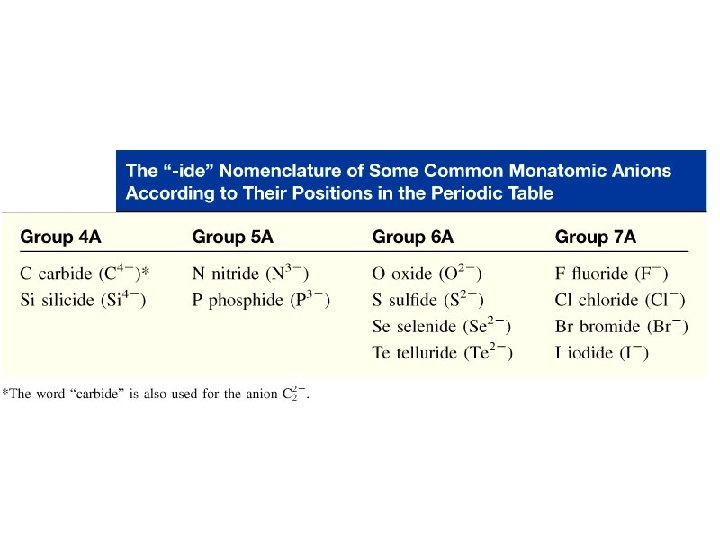
Chemical Substances; Formulas and Names • Rules for naming monatomic ions Monatomic cations are named after the element – if there is only one such ion. For example, Al 3+ is called the aluminum ion. If there is more than one cation of an element, a Roman numeral in parentheses denoting the charge on the ion is used. This often occurs with transition elements. i. e. Fe 2+ = Iron(II); Fe 3+ = Iron(III) The names of the monatomic anions use the stem name of the element followed by the suffix – ide. For example, Br- is called the bromide ion.


Naming Compounds • Binary Compounds – Composed of two elements – Ionic and covalent compounds included • Binary Ionic Compounds – Metal—nonmetal • Binary Covalent Compounds – Nonmetal—nonmetal Copyright © Cengage Learning. All rights reserved 67

Binary Ionic Compounds (Type I) 1. The cation is always named first and the anion second. 2. A monatomic cation takes its name from the name of the parent element. 3. A monatomic anion is named by taking the root of the element name and adding –ide. Copyright © Cengage Learning. All rights reserved 68

Binary Ionic Compounds (Type I) • Examples: KCl Potassium chloride Mg. Br 2 Magnesium bromide Ca. O Calcium oxide Copyright © Cengage Learning. All rights reserved 69

Naming Binary Compounds • Na. F • Li. Cl • Mg. O -

Naming Binary Compounds • Na. F • Li. Cl • Mg. O - Sodium Fluoride Lithium Chloride Magnesium Oxide

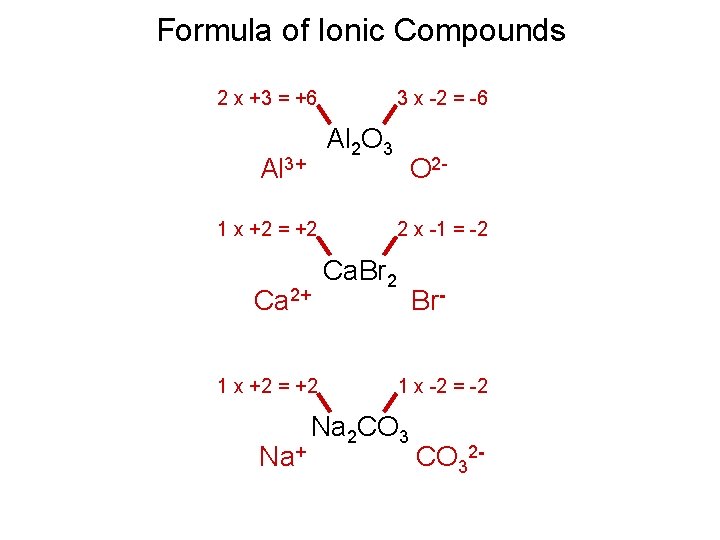
Formula of Ionic Compounds 2 x +3 = +6 3 x -2 = -6 Al 2 O 3 Al 3+ 1 x +2 = +2 Ca 2+ 1 x +2 = +2 Na+ O 22 x -1 = -2 Ca. Br 2 Br 1 x -2 = -2 Na 2 CO 32 -

Formula of Ionic Compounds 1 x +2 = +2 Mg 2+ 1 x -2 = -2 Mg. O O 2 - NOT 1 x +2 = +2 Mg 2+ 1 x -2 = -2 Mg 2 O 2 O 2 - Use lowest common denominator

Binary Ionic Compounds (Type II) • Metals in these compounds form more than one type of positive ion. • Charge on the metal ion must be specified. • Roman numeral indicates the charge of the metal cation. • Transition metal cations usually require a Roman numeral. • Elements that form only one cation do not need to be identified by a roman numeral. Copyright © Cengage Learning. All rights reserved 74

Binary Ionic Compounds (Type II) • Examples: Cu. Br Copper(I) bromide Fe. S Iron(II) sulfide Pb. O 2 Lead(IV) oxide Copyright © Cengage Learning. All rights reserved 75

• Transition metal ionic compounds – indicate charge on metal with Roman numerals Fe. Cl 2 2 Cl- -2 so Fe is +2 iron(II) chloride Fe. Cl 3 3 Cl- -3 so Fe is +3 iron(III) chloride Cr 2 S 3 3 S-2 -6 so Cr is +3 (6/2) chromium(III) sulfide

Polyatomic Ions • Must be memorized (see Table 2. 5 on pg. 65 in text). • Examples of compounds containing polyatomic ions: Na. OH Sodium hydroxide Mg(NO 3)2 Magnesium nitrate (NH 4)2 SO 4 Ammonium sulfate Copyright © Cengage Learning. All rights reserved 77

Minimum Polyatomic Ions You Should Know • • • NH 4+ - Ammonium OH- - Hydroxide CN- - Cyanide SO 42 - - Sulfate Cl. O 4 - - Perchlorate • • O 22 - - Peroxide PO 43 - - Phosphate CO 32 - - Carbonate HCO 3 - - Hydrogen carbonate

Chemical Substances; Formulas and Names • Polyatomic ions A polyatomic ion is an ion consisting of two or more atoms chemically bonded together and carrying a net electric charge.
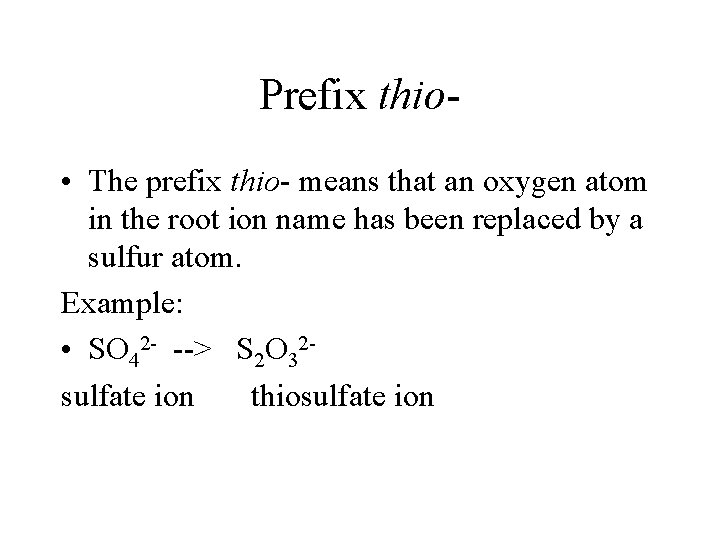
Prefix thio • The prefix thio- means that an oxygen atom in the root ion name has been replaced by a sulfur atom. Example: • SO 42 - --> S 2 O 32 sulfate ion thiosulfate ion


Chemical Nomenclature • Ionic Compounds – often a metal + nonmetal – anion (nonmetal), add “ide” to element name Ba. Cl 2 barium chloride K 2 O potassium oxide Mg(OH)2 magnesium hydroxide KNO 3 potassium nitrate

More Practice • Na 2 SO 4 Sodium Sulfate • Ag. CN Silver Cyanide • Ca(OCl)2 Calcium Hypochlorite Na 2 SO 3 Sodium Sulfite Cd(OH)2 Cadmium Hydroxide KCl. O 4 Potassium Perchlorate

Formation of Ionic Compounds To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 84

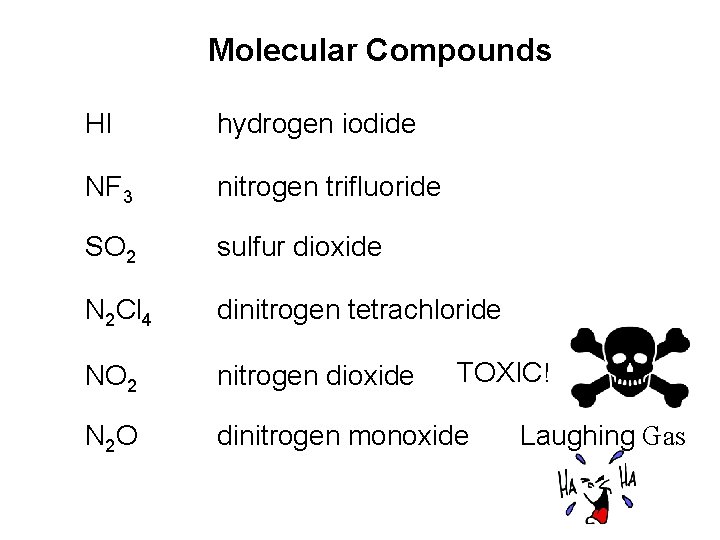
• Molecular compounds • nonmetals or nonmetals + metalloids • common names • H 2 O, NH 3, CH 4, C 60 • element further left in periodic table is 1 st • element closest to bottom of group is 1 st • if more than one compound can be formed from the same elements, use prefixes to indicate number of each kind of atom • last element ends in ide


Binary Covalent Compounds (Type III) • Formed between two nonmetals. 1. The first element in the formula is named first, using the full element name. 2. The second element is named as if it were an anion. 3. Prefixes are used to denote the numbers of atoms present. 4. The prefix mono- is never used for naming the first element. Copyright © Cengage Learning. All rights reserved 87

Prefixes Used to Indicate Number in Chemical Names Copyright © Cengage Learning. All rights reserved 88

Binary Covalent Compounds (Type III) • Examples: CO 2 Carbon dioxide SF 6 Sulfur hexafluoride N 2 O 4 Dinitrogen tetroxide Copyright © Cengage Learning. All rights reserved 89

Molecular Compounds HI hydrogen iodide NF 3 nitrogen trifluoride SO 2 sulfur dioxide N 2 Cl 4 dinitrogen tetrachloride NO 2 nitrogen dioxide N 2 O dinitrogen monoxide TOXIC! Laughing Gas


Acids • Acids can be recognized by the hydrogen that appears first in the formula—HCl. • Molecule with one or more H+ ions attached to an anion. Copyright © Cengage Learning. All rights reserved 92

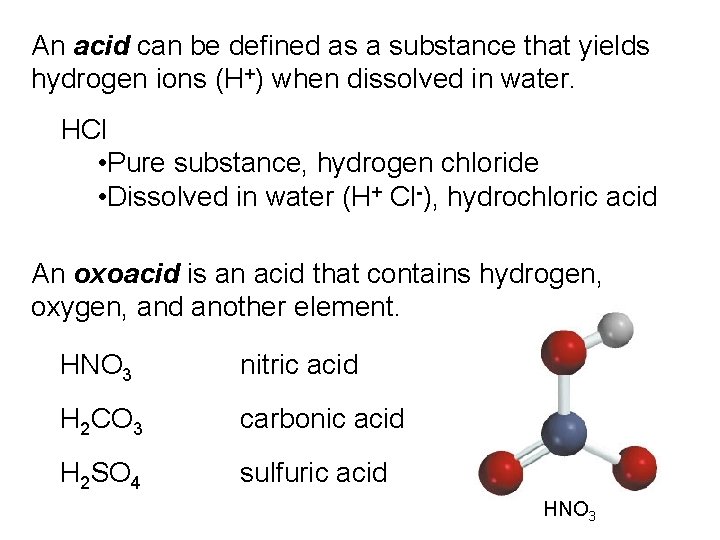
An acid can be defined as a substance that yields hydrogen ions (H+) when dissolved in water. HCl • Pure substance, hydrogen chloride • Dissolved in water (H+ Cl-), hydrochloric acid An oxoacid is an acid that contains hydrogen, oxygen, and another element. HNO 3 nitric acid H 2 CO 3 carbonic acid H 2 SO 4 sulfuric acid HNO 3

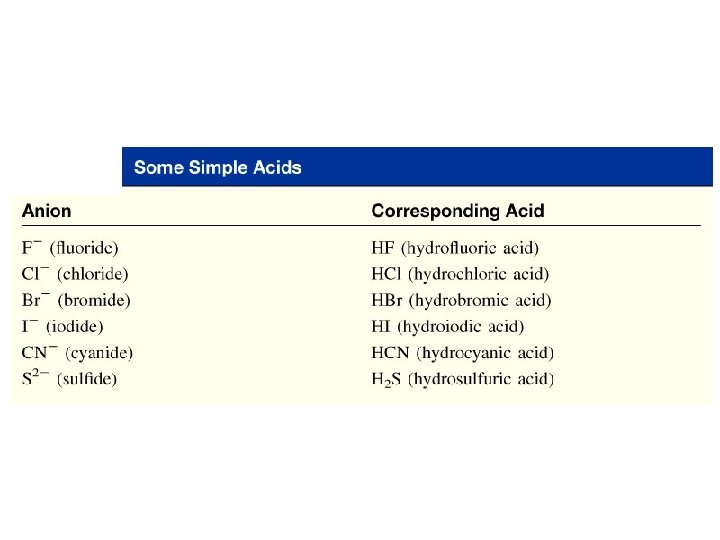
Acids • If the anion does not contain oxygen, the acid is named with the prefix hydro– and the suffix –ic. • Examples: HCl Hydrochloric acid HCN Hydrocyanic acid H 2 S Hydrosulfuric acid Copyright © Cengage Learning. All rights reserved 94


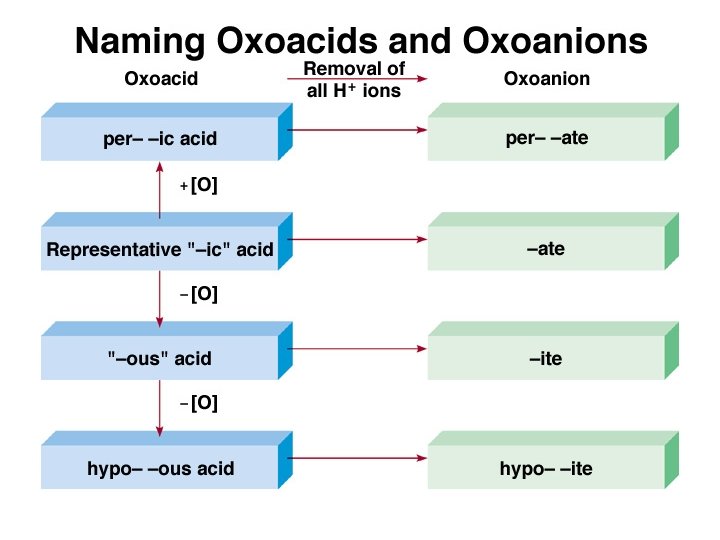
Oxoacid • If the anion does contain oxygen: – The suffix –ic is added to the root name if the anion name ends in –ate. • Examples: HNO 3 Nitric acid H 2 SO 4 Sulfuric acid HC 2 H 3 O 2 Acetic acid Copyright © Cengage Learning. All rights reserved 96

Oxoacid • If the anion does contain oxygen: – The suffix –ous is added to the root name if the anion name ends in –ite. • Examples: HNO 2 Nitrous acid H 2 SO 3 Sulfurous acid HCl. O 2 Chlorous acid Copyright © Cengage Learning. All rights reserved 97


Acid anions (provide + H ions) • Uses of mono– and di • Rule: to be used if oxyanion is bonded to one or more hydrogen ions. Example: • HPO 42 - = monohydrogen phosphate ion • H 2 PO 4 - = dihydrogen phosphate ion


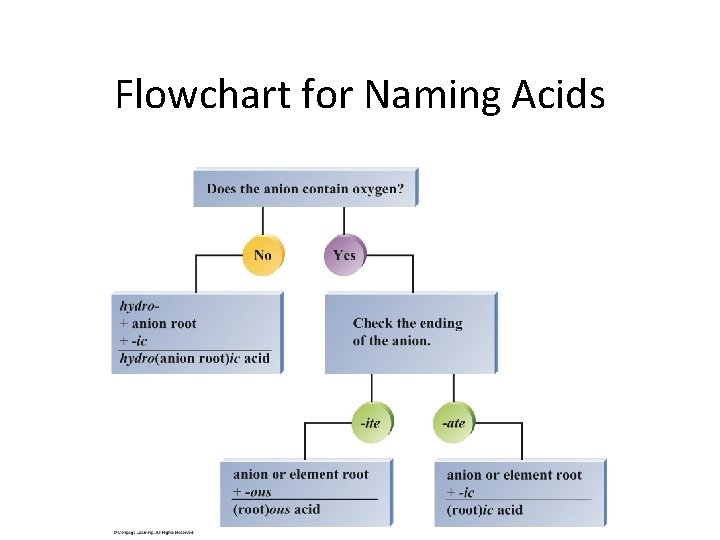
Flowchart for Naming Acids Copyright © Cengage Learning. All rights reserved

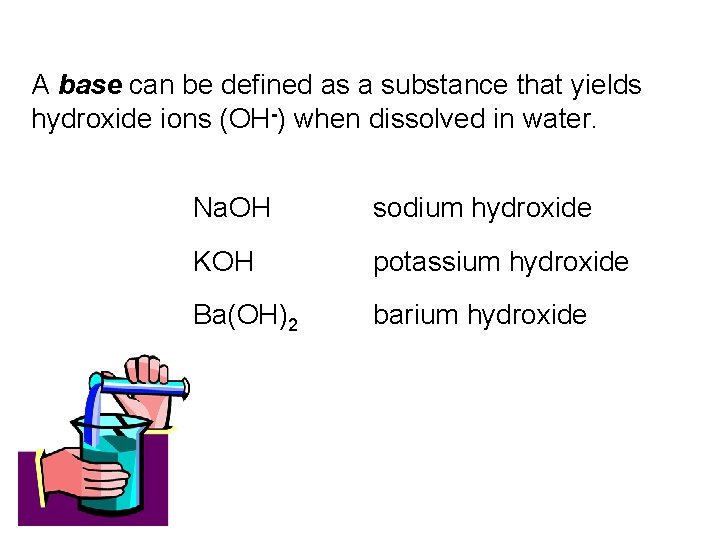
A base can be defined as a substance that yields hydroxide ions (OH-) when dissolved in water. Na. OH sodium hydroxide KOH potassium hydroxide Ba(OH)2 barium hydroxide

EXERCISE! Which of the following compounds is named incorrectly? a) KNO 3 b) Ti. O 2 c) Sn(OH)4 d) PBr 5 e) Ca. Cr. O 4 potassium nitrate titanium(II) oxide tin(IV) hydroxide phosphorus pentabromide calcium chromate Copyright © Cengage Learning. All rights reserved 103

Worked Example 2. 1





