Counting Atoms and Balancing Chemical Equations Elements Compounds

Counting Atoms and Balancing Chemical Equations

Elements, Compounds, Mixtures l l Hydrogen is an element. Oxygen is an element. When hydrogen and oxygen bond they make the compound water. When salt and water are combined, a mixture is created. Compounds in mixtures retain their individual properties. The ocean is a mixture.

Elements, compounds, and mixtures l Elements are pure substances. When the subatomic particles of an element are separated from its atom, it no longer retains the properties of that element. l Mixtures can be separated by physical means. l Compounds can only be separated by chemical means.

Identifying Compounds Each new element is identified by a capital letter l Example: H 2 SO 4 l l The elements in Sulfuric Acid l Hydrogen l Sulfur l Oxygen

Subscripts l C 12 H 22 O 11 l There are 12 atoms of Carbon l There are 22 atoms of Hydrogen l There are 11 atoms of Oxygen If there is not a subscript listed, it is understood to be 1. l Example: Na. Cl l l There is one atom of Sodium l There is one atom of Chlorine

You Practice! Na. HCO 3 HCl Sodium – 1 Hydrogen – 1 Carbon – 1 Oxygen -3 Hydrogen – 1 Chlorine - 1

There are times you will see a compound with parenthesis. Pb(NO 3)2 The 2 after the parenthesis indicates there are two sets of the parenthesis. Pb(NO 3) So, in counting the atoms, you would have the following: Lead – 1 Oxygen - 6 Nitrogen -2

You Practice!! (NH 4)3 PO 4 Mg(OH)2 Nitrogen – 3 Hydrogen – 12 Phosphorus – 1 Oxygen – 4 Magnesium – 1 Oxygen – 2 Hydrogen – 2

Coefficient 2 H 2 SO 4 This means there are 2 compounds of Sulfuric Acid. Think: H 2 SO 4 Counting the atoms: Hydrogen – 4 Sulfur – 2 Oxygen - 8

You Practice!! 3 H 3 PO 4 2 H 2 O Hydrogen – 9 Phosphorus – 3 Oxygen - 12 Hydrogen – 4 Oxygen - 2

Got It? ?


Chemical Equations Chemical equations express what is happening in a chemical reaction using symbols. 2 K + Cl 2 Reactant yields 2 KCl Product

Law of Conservation of Mass In a chemical reaction, matter cannot be created or destroyed. It can only be changed. Therefore the Reactants must equal the Products for the equation to be balanced

What does that mean? ? The reactants MUST contain the same elements and the same number of each element as the product. BUT they CAN be in different compounds.

HUH? ? ? H 2 + O 2 Reactants H=2 O=2 H 2 O Products H =2 O=1 This cannot happen. An oxygen atom cannot be destroyed.

So what do we do? The chemical equation must be written according to the law. AGAIN --- HUH? ? ? H 2 + O 2 H 2 O is the production of water. Well, how it actually happens in nature is this…. 2 H 2 + O 2 Reactants: H=4 O=2 2 H 2 O Products: H=4 O=2

Do I Need to Know How to Balance? NOT YET. You need to be able to recognize if the chemical reaction follows the law of conservation of mass. Na + O 2 Mg + 2 HCl Na 2 O NOPE Mg. Cl 2 + H 2 YUP!!

You Practice!!! Hg. O Hg + O 2 Hg = O= NOPE YUP N 2 + 3 H 2 2 NH 3 YUP Zn + 2 HCl Zn. Cl 2 + H 2
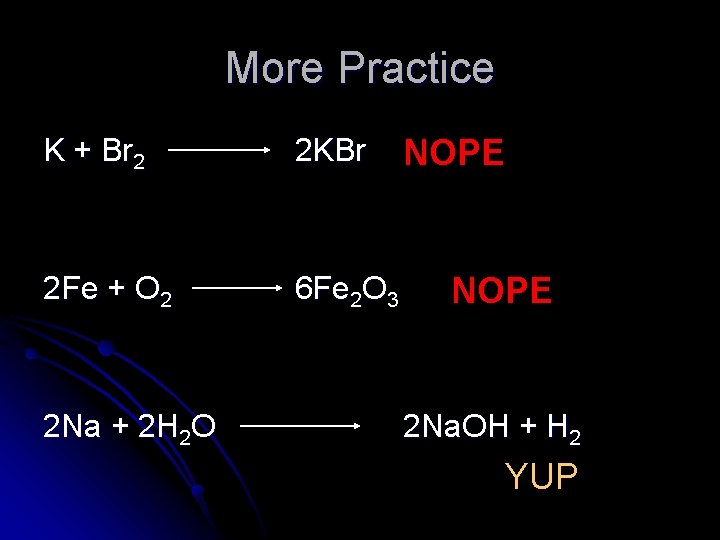
More Practice K + Br 2 2 KBr 2 Fe + O 2 6 Fe 2 O 3 2 Na + 2 H 2 O NOPE 2 Na. OH + H 2 YUP

Got It? ? ?

Balancing Chemical Equations H 2 + O 2 Reactants: H = 2 O=2 H 2 O Products: H = 2 O=1 What do we multiply by 1 to get 2?

Keep Going…. H 2 + O 2 Reactants: H = 2 O=2 2 H 2 O Products: H = 4 O=2 Now the Oxygen is balanced but Hydrogen is not, so what do we multiply by 2 to get 4?

Almost There!!! 2 H 2 + O 2 2 H 2 O Reactants: H = 4 O=2 Products: H = 4 O=2 Now it is balanced!!! Congratulations, you have now learned the hardest thing we will do all year!!
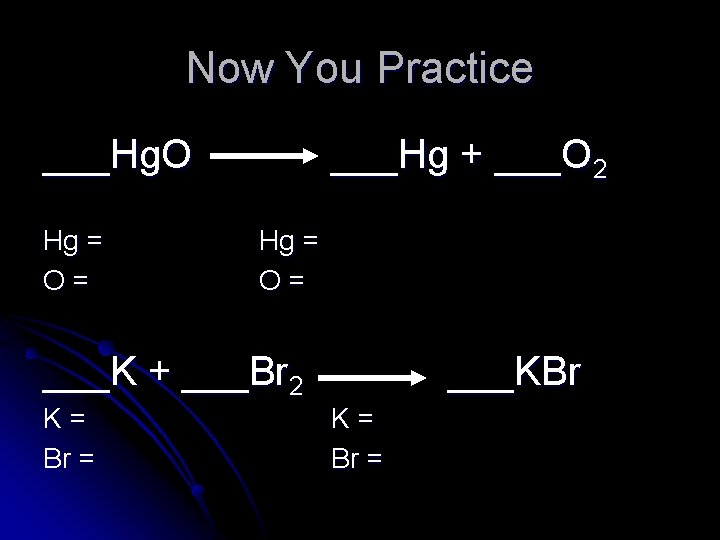
Now You Practice ___Hg. O Hg = O= ___Hg + ___O 2 Hg = O= ___K + ___Br 2 K= Br = ___KBr K= Br =
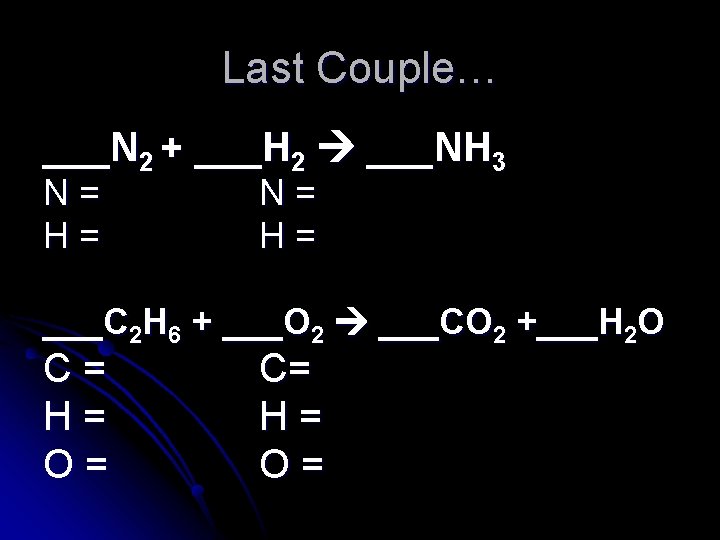
Last Couple… ___N 2 + ___H 2 ___NH 3 N= H= ___C 2 H 6 + ___O 2 ___CO 2 +___H 2 O C= H= O=

Warm Up 3/11 Write Q&A l Why do we have to balance equations? (What law do we have to follow? )

Warm Up 3/10 Write Q&A l Balance the Following: l HNO 3 + Ca(OH)2 Ca(NO 3)2 + H 2 O

Warm Up 3/8 Write the Following: The Law of Conservation of Mass In a chemical reaction, matter cannot be created or destroyed. It can only be changed.

Warm Up 3/5 Write Q&A l How many atoms are in 3 Pb(OH)4? Pb l. O l. H l

Warm Up March 4 Write Q&A l The vertical columns on the periodic table are called _______ and they correspond to the number of _____ l The horizontal rows on the periodic table are called ______ and they correspond to the number of _____

Warm Up 3/9 Is it balanced? Show your work!! l Al + O 2 ---> Al 2 O 3 l Si. Cl 4 + H 2 O ---> H 4 Si. O 4 + HCl

Go !! s r e g n Ra Monday, October 18 Objective: Students will be able to: practice counting atoms 1. Go over Counting Atoms Worksheet What are you learning 2. Chemical Formula Practice Worksheet (in class) today? 3. Grade (if time) Warm Up How many atoms are in Pb(OH)4? Things to do: • Take out a grading pen and Counting Atoms WS • Clear everything else off your desk Pb O H

Go !! s r e g n Ra Monday, October 18 Objective: Students will be able to: practice counting atoms 1. Go over Counting Atoms Worksheet What are you learning 2. Chemical Formula Practice Worksheet (in class) today? 3. Grade (if time) Warm Up How many atoms are in Pb(OH)4? Things to do: • Take out a grading pen and Counting Atoms WS • Clear everything else off your desk Pb O H

Go !! s r e g n Ra Thursday, October 21 Objective: Students will be able to: Review the differences What are between Physical and Chemical Changes you learning 1. Go Over #1 -5 today? 2. Balancing Equations Activity 3. Do #1 -7 for HW 5 H 2 Warm Up Things to do: What number represents the Coefficient? ____ What number represents the Subscript? _____ What element is represented by the letter "H"? ______ How many "H's" do you have? _____ • Take out Worksheet from Yesterday #1 -5 pap

Go !! s r e g n Ra Wednesday, October 20 Objective: Students will be able to: use the Law of What are Conservation of mass to Balance Equations you learning 1. Grade #11 -20 today? 2. Balancing Equations Activity 3. Do #1 -7 for HW Balance the Following: Warm Up __HNO 3 +__Ca(OH)2 __Ca(NO 3)2 + __H 2 O Things to do: • Take out a grading pen • Clear everything else off your desk pap

Go !! s r e g n Ra Wednesday, October 20 Objective: Students will be able to: use the Law of What are Conservation of mass to Balance Equations you learning 1. Go over Homework #1 -10 today? 2. Notes on Balancing Equations 3. Balancing Equations Worksheet #1 -5 4. Do #11 -20 on your Homework from yesterday Warm Up Things to do: What law do we have to follow when we are figuring if equations are balanced or not? Why? • Take out your homework from yesterday

Go !! s r e g n Ra Tuesday, October 19 Objective: Students will be able to: use the skill of counting What are atoms to tell if an equation is balanced you learning 1. Notes on Balancing Equations today? 2. Balancing Equations Worksheet #1 -10 Warm Up How many atoms are in 3 Pb(OH)4? (look at what you did yesterday!!) Pb O H Things to do: • Take out a grading pen • Clear everything else off your desk
- Slides: 38