Solid Liquid Gas a Particles in solid b

Solid, Liquid, Gas (a) Particles in solid (b) Particles in liquid (c) Particles in gas
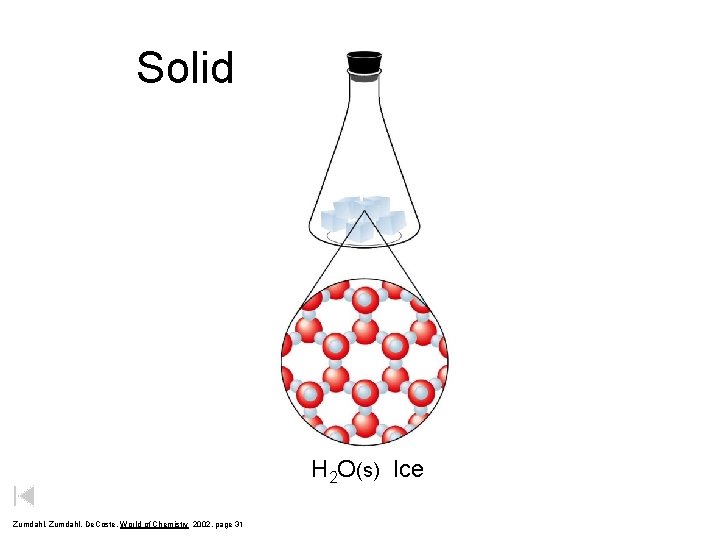
Solid H 2 O(s) Ice Zumdahl, De. Coste, World of Chemistry 2002, page 31
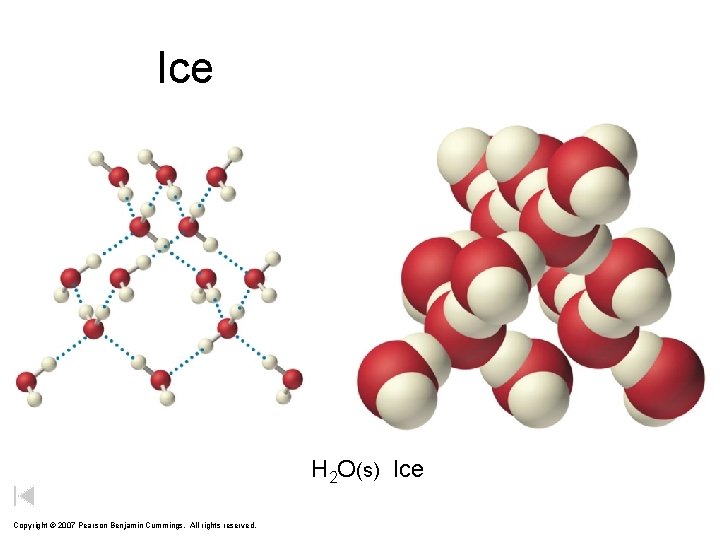
Ice Photograph of snowflakes Photograph of ice model H 2 O(s) Ice Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
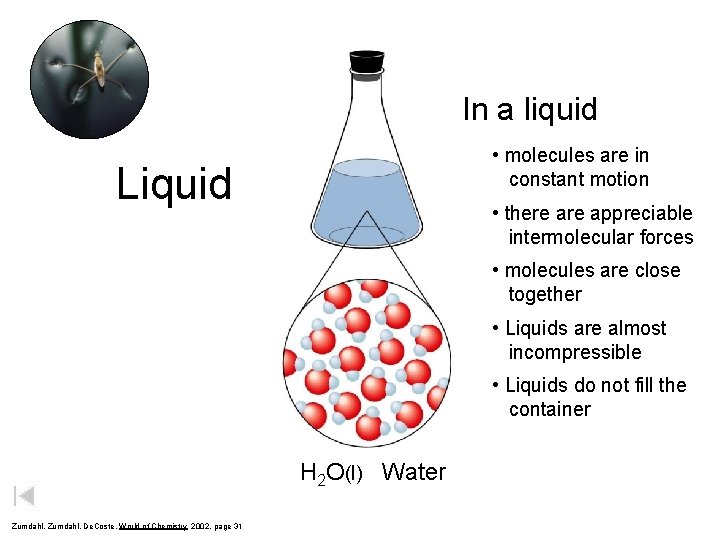
In a liquid • molecules are in constant motion Liquid • there appreciable intermolecular forces • molecules are close together • Liquids are almost incompressible • Liquids do not fill the container H 2 O(l) Water Zumdahl, De. Coste, World of Chemistry 2002, page 31
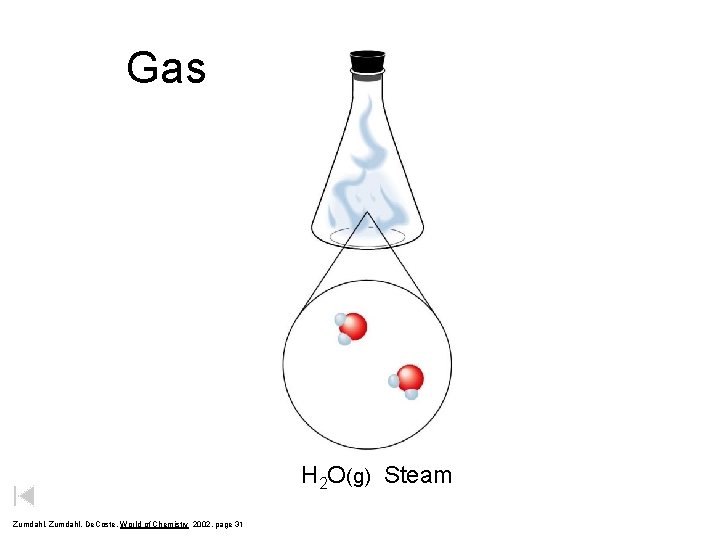
Gas H 2 O(g) Steam Zumdahl, De. Coste, World of Chemistry 2002, page 31
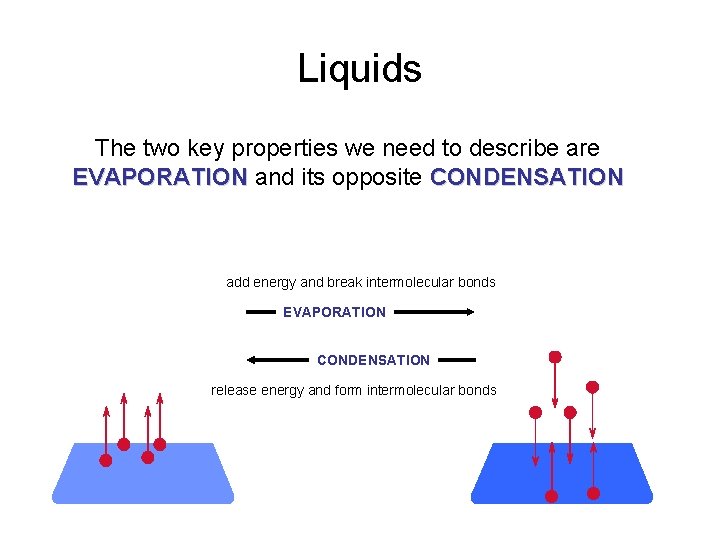
Liquids The two key properties we need to describe are EVAPORATION and its opposite CONDENSATION add energy and break intermolecular bonds EVAPORATION CONDENSATION release energy and form intermolecular bonds

States of Matter

Gas, Liquid, and Solid Gas Zumdahl, De. Coste, World of Chemistry 2002, page 441 Liquid Solid
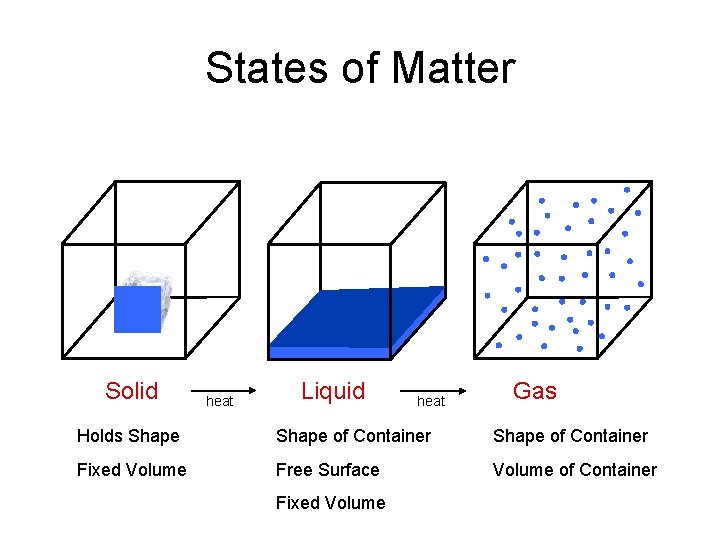
States of Matter Solid heat Liquid heat Gas Holds Shape of Container Fixed Volume Free Surface Volume of Container Fixed Volume
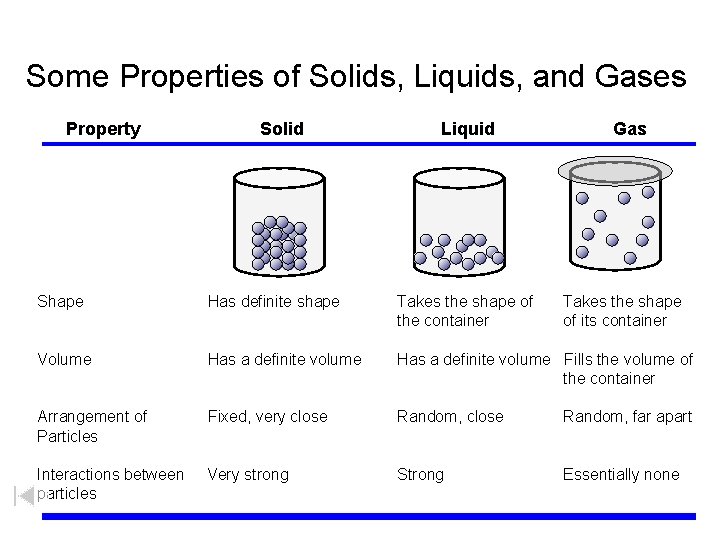
Some Properties of Solids, Liquids, and Gases Property Solid Liquid Gas Shape Has definite shape Takes the shape of the container Takes the shape of its container Volume Has a definite volume Fills the volume of the container Arrangement of Particles Fixed, very close Random, far apart Interactions between particles Very strong Strong Essentially none

Evaporation • To evaporate, molecules must have sufficient energy to break IM forces. • Molecules at the surface break away and become gas. • Only those with enough KE escape. • Breaking IM forces requires energy. The process of evaporation is endothermic • Evaporation is a cooling process. • It requires heat.

Condensation Change from gas to liquid Achieves a dynamic equilibrium with vaporization in a closed system. What is a closed system? A closed system means matter can’t go in or out. (put a cork in it) What the heck is a “dynamic equilibrium? ”

Dynamic Equilibrium When first sealed, the molecules gradually escape the surface of the liquid. As the molecules build up above the liquid - some condense back to a liquid. The rate at which the molecules evaporate and condense are equal.

Dynamic Equilibrium As time goes by the rate of vaporization remains constant but the rate of condensation increases because there are molecules to condense. Equilibrium is reached when: Rate of Vaporization = Rate of Condensation Molecules are constantly changing phase “dynamic” The total amount of liquid and vapor remains constant “equilibrium”

Vaporization • Vaporization is an endothermic process - it requires heat. • Energy is required to overcome intermolecular forces • Responsible for cool earth • Why we sweat
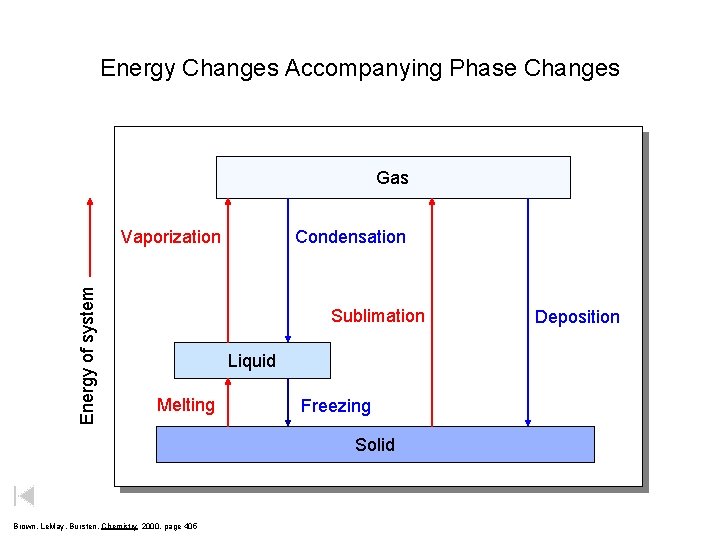
Energy Changes Accompanying Phase Changes Gas Energy of system Vaporization Condensation Sublimation Liquid Melting Freezing Solid Brown, Le. May, Bursten, Chemistry 2000, page 405 Deposition
- Slides: 16