Counting Atoms and Building Molecules How to Count

Counting Atoms and Building Molecules

How to Count Atoms 1. The symbol of an element represents one atom. – Hydrogen = H 2. A subscript indicates the number of atoms. – H 2 O = 2 hydrogen atoms 1 oxygen 3. A subscript outsideatom a bracket multiplies all the elements inside the brackets. – Mg 3(PO 4) 2 = 3 Magnesium Atoms 2 Phosphorous atoms 8 Oxygen Atoms
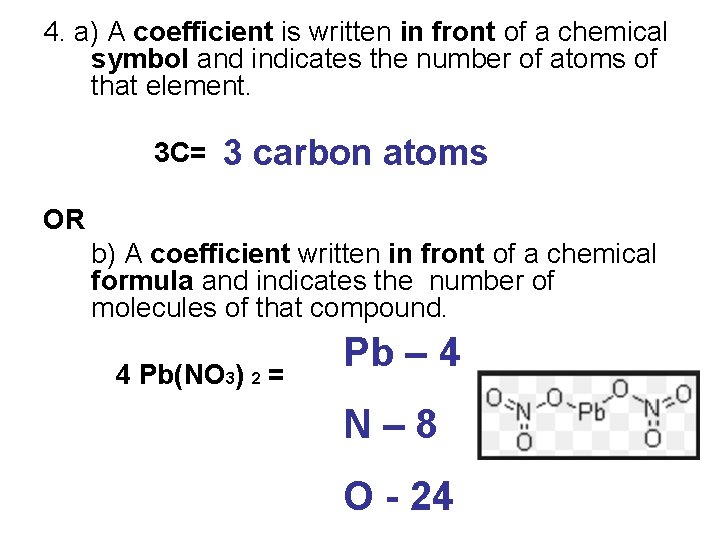
4. a) A coefficient is written in front of a chemical symbol and indicates the number of atoms of that element. 3 C= 3 carbon atoms OR b) A coefficient written in front of a chemical formula and indicates the number of molecules of that compound. 4 Pb(NO 3) 2 = Pb – 4 N– 8 O - 24
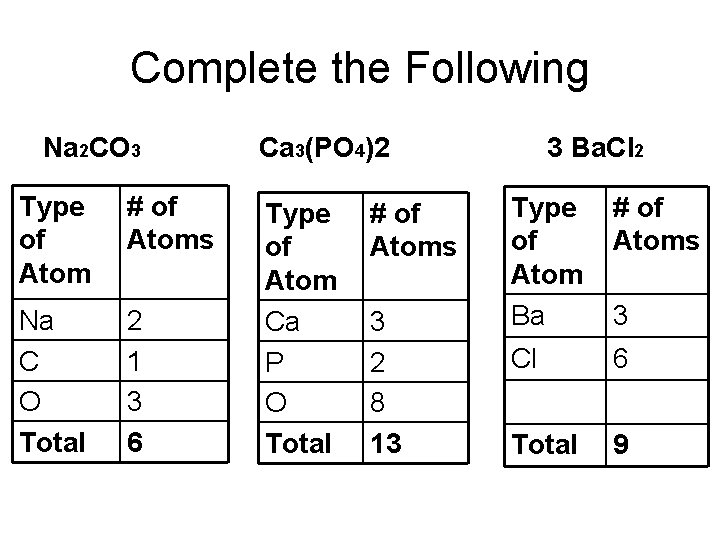
Complete the Following Na 2 CO 3 Type of Atom # of Atoms Na C O Total 2 1 3 6 Ca 3(PO 4)2 Type of Atom Ca P O Total # of Atoms 3 2 8 13 3 Ba. Cl 2 Type of Atom Ba # of Atoms Cl 6 Total 9 3

Names and Formulas for Compounds • Atoms combine to fill combining capacity. It is the number of “connections” an element can make with other elements. • Naming – metal + nonmetal, change nonmetal ending to “ide” – metal + nonmetal + oxygen, change the nonmetal ending to “ate”. Sodium Fluoride Ex. Sodium + fluorine → _______ Potassium + chlorine + oxygen → Potassium Chlorate ________

To Do / Homework • 1) Read pages 190 -194 • 2) Questions pg. 197 #1 -6 • 3) Worksheet

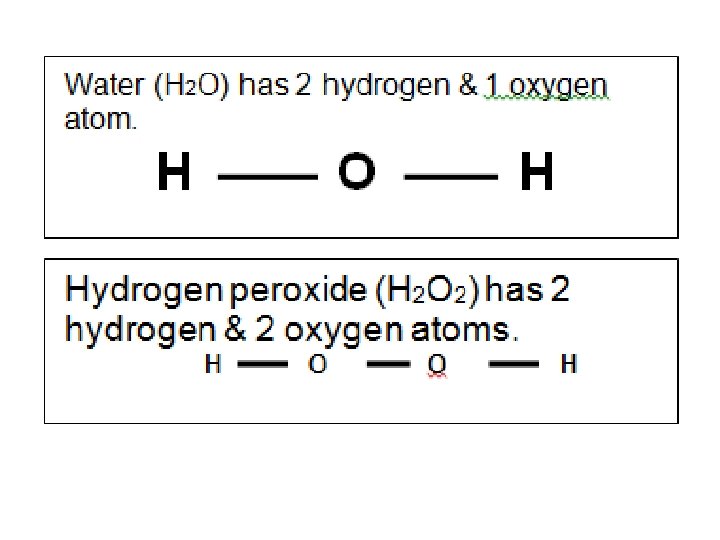
Building Models of Molecules • A molecule is a combination of two or more atoms. • These atoms are held together by bonds, which represent the electrons of the atoms. • We draw the bonds of atoms using structural diagrams.

• Each type of atom has a specific number of bonds it can make • Know this! Atom Hydrogen # of connections per atom One Oxygen Two Nitrogen Three Carbon Four

Build the following Molecules and draw them in your notebook! Symbol Name 1 O 2 oxygen 2 CO 2 carbon dioxide 3 CH 4 methane gas 4 CH 3 OH methanol 5 HCHO formaldehyde 6 C 2 H 6 ethane 7 C 2 H 5 OH ethanol 8 C 6 H 6 benzene (ring) 9 CH 3 COOH acetic acid (vinegar) 10 C 6 H 12 O 6 glucose - a challenge!
- Slides: 10