Marshmallow Atoms Molecules Lab Name Marshmallow Atoms Molecules

Marshmallow Atoms & Molecules Lab Name:
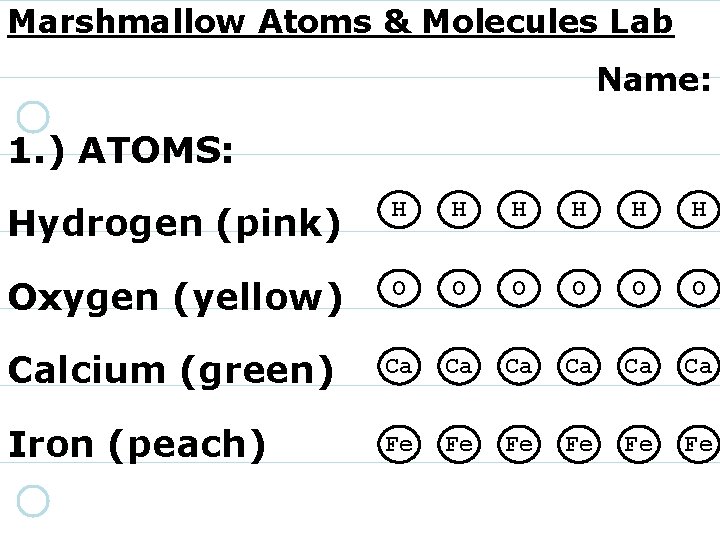
Marshmallow Atoms & Molecules Lab Name: 1. ) ATOMS: Hydrogen (pink) H H H Oxygen (yellow) O O O Calcium (green) Ca Ca Ca Iron (peach) Fe Fe Fe

2) Atoms are the smallest particles of an element that still have the properties of an element.

3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 16. 00 O Oxygen 220 Calcium 40. 08 Ca Calcium 2+ Iron 26 55. 85 Fe Iron 3+/2+
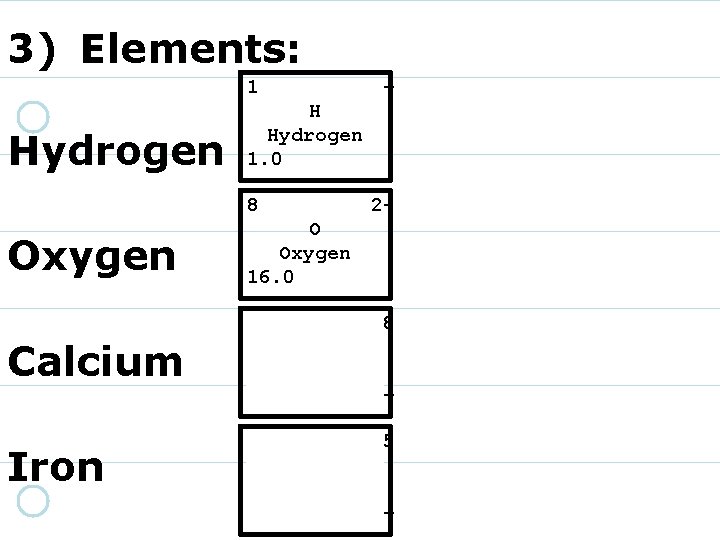
3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium 40. 08 Ca Calcium 2+ Iron 26 55. 85 Fe Iron 3+/2+
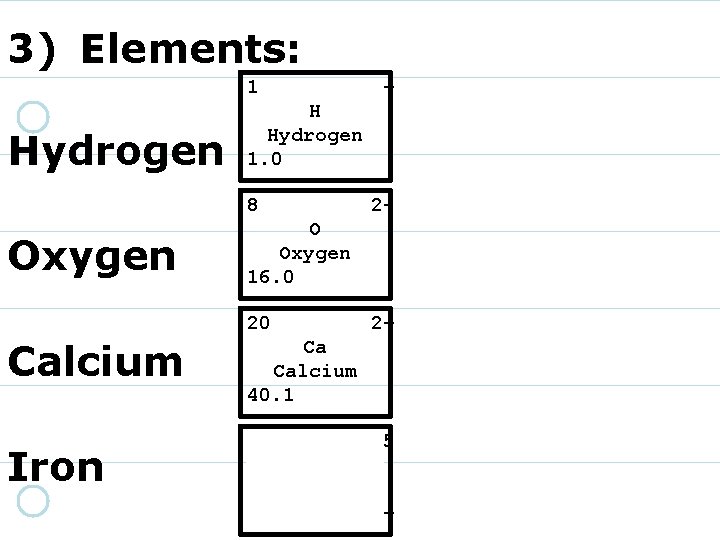
3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium Iron 2+ Ca Calcium 40. 1 26 55. 85 Fe Iron 3+/2+

3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium Iron 2+ Ca Calcium 40. 1 26 3+/2+ Fe Iron 55. 8
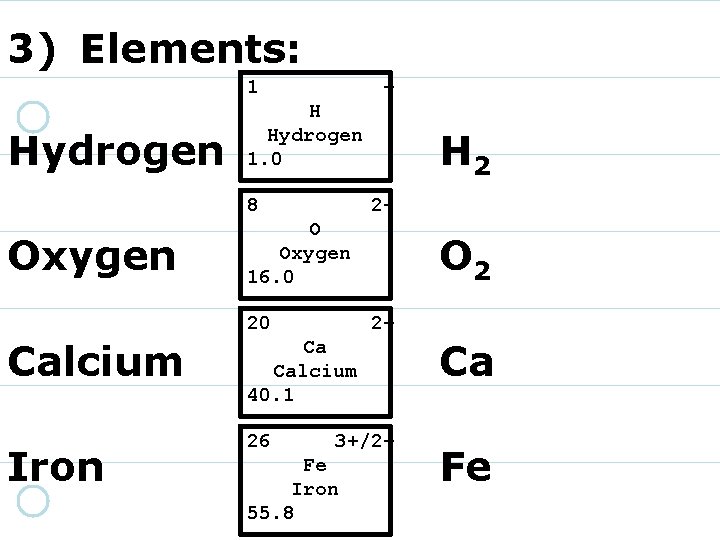
3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium Iron H 2 O 2 2+ Ca Calcium 40. 1 26 3+/2+ Fe Iron 55. 8 Ca Fe
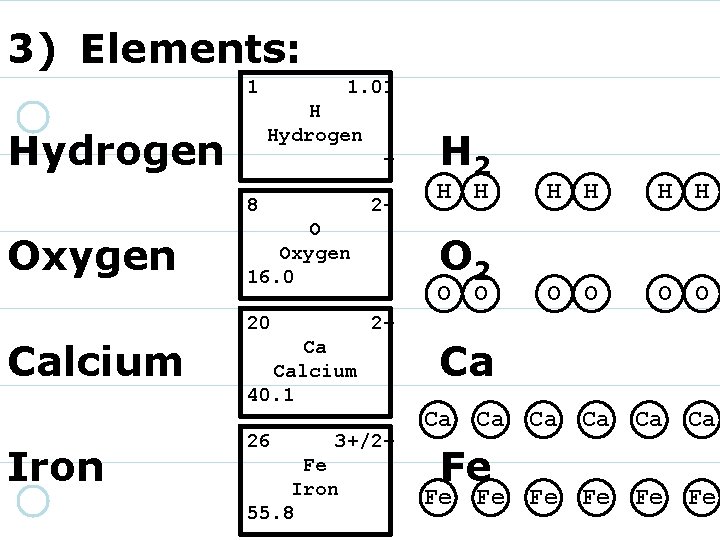
3) Elements: 1 1. 01 H Hydrogen + 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium Iron H H O 2 O O H H O O 2+ Ca Calcium 40. 1 26 H 2 3+/2+ Fe Iron 55. 8 Ca Ca Fe Fe Ca Ca Ca Fe Fe Fe

1) GET 6 each of the 4 colours of miniature marshmallows. 2) PLACE them on the circles in #1. 3) TAKE only what is needed. DON’T BE A GREEDY PIG!
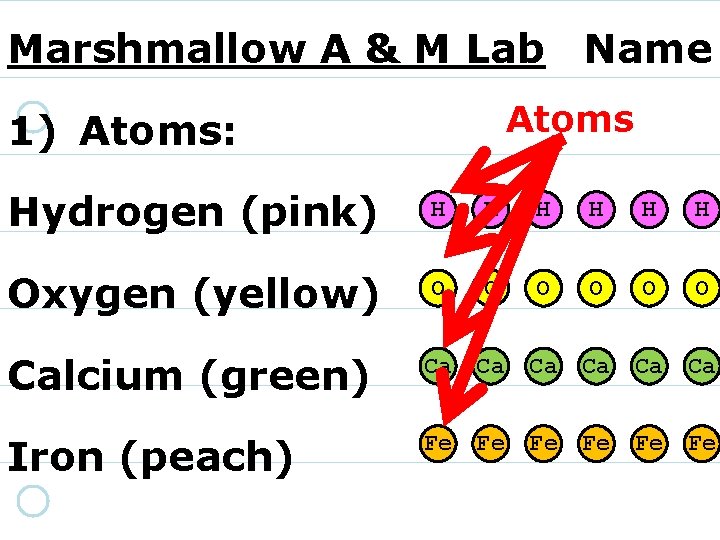
Marshmallow A & M Lab Name Atoms 1) Atoms: Hydrogen (pink) H H H Oxygen (yellow) O O O Calcium (green) Ca Ca Ca Iron (peach) Fe Fe Fe

MOVE all the marshmallows onto the circles in #3.

3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium Iron H 2 O O H H O O 2+ Ca Calcium 40. 1 26 H H 3+/2+ Fe Iron 55. 8 Ca Ca Ca Fe Fe Fe
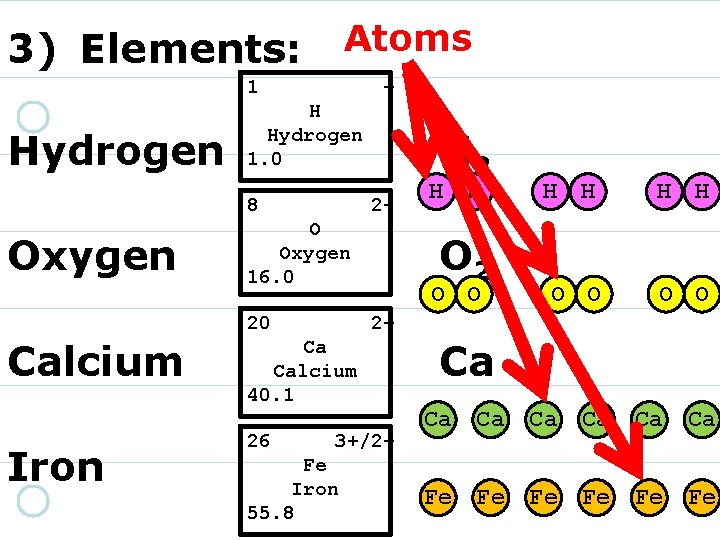
Atoms 3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium Iron H 2 O O H H O O 2+ Ca Calcium 40. 1 26 H H 3+/2+ Fe Iron 55. 8 Ca Ca Fe Fe Fe
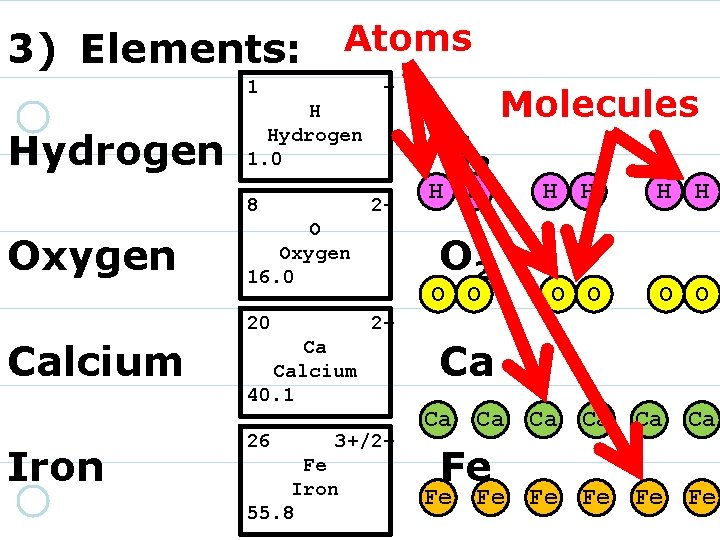
Atoms 3) Elements: 1 Hydrogen + H Hydrogen 1. 0 8 Oxygen 2 - O Oxygen 16. 0 20 Calcium Iron H 2 O O H H O O 2+ Ca Calcium 40. 1 26 H H Molecules 3+/2+ Fe Iron 55. 8 Ca Ca Fe Fe Ca Ca Ca Fe Fe Fe

4) Properties: H = colourless gas, explosive O = colourless gas, burns Ca = metal, reacts with water Fe = metal, rusts

Aren’t protons, neutrons, and electrons the smallest part of an element?

Aren’t protons, neutrons, and electrons the smallest part of an element? Just like bread, peanut butter, and jam make up a sandwich, but are not the sandwich themselves,

Aren’t protons, neutrons, and electrons the smallest part of an element? Just like bread, peanut butter, and jam make up a sandwich, but are not the sandwich themselves, protons, neutrons and electrons make up an atom, but are not the atom themselves.
5) Hindenburg fire 2 H 2 + O 2 2 H 20

5) Hindenburg fire 2 H 2 H H + O 2 2 H 20 H H 1) MOVE your marshmallows into place as needed! 2) You DO NOT need to draw them!

5) Hindenburg fire 2 H 2 H H + H H O 2 O O 1) DON’T GET any more! 2) REARRANGE them instead. 2 H 20
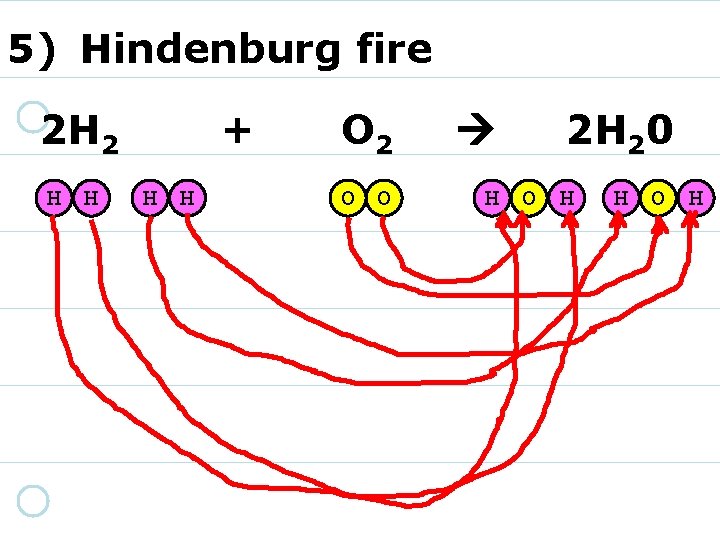
5) Hindenburg fire 2 H 2 H H + H H O 2 O O 2 H 20 H O H

5) Hindenburg fire 2 H 2 H H A? M? + H H O 2 O O A? M? 2 H 20 H O H A? M?
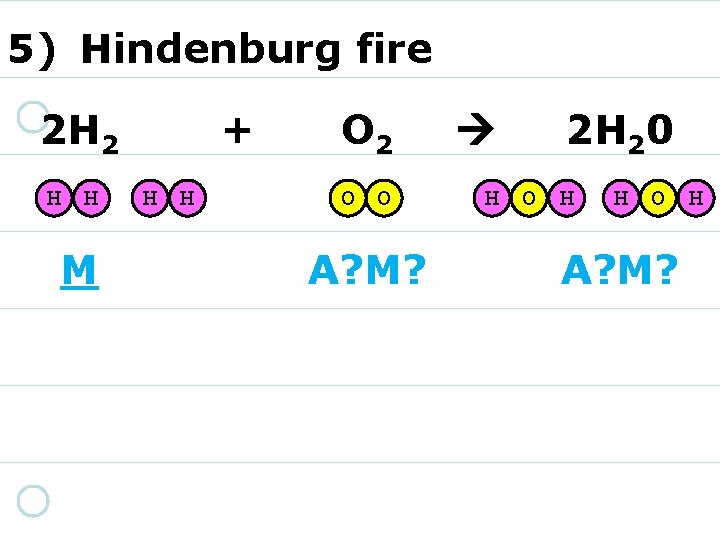
5) Hindenburg fire 2 H 2 H H M + H H O 2 O O A? M? 2 H 20 H O H A? M?
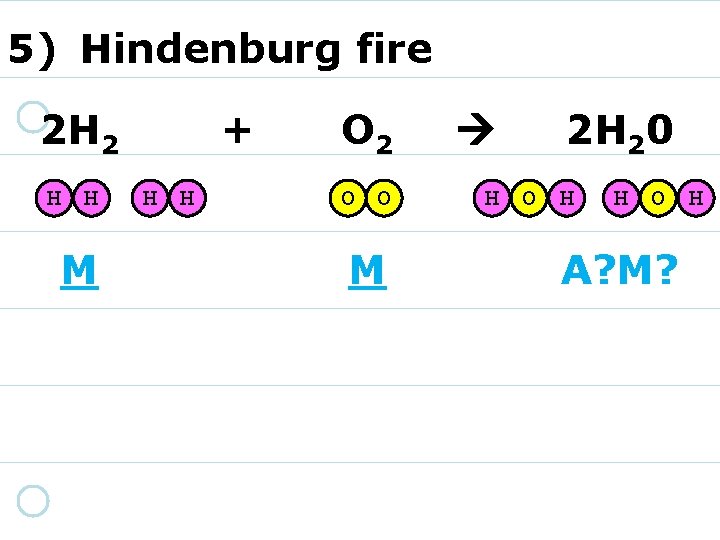
5) Hindenburg fire 2 H 2 H H M + H H O 2 O O M 2 H 20 H O H A? M?
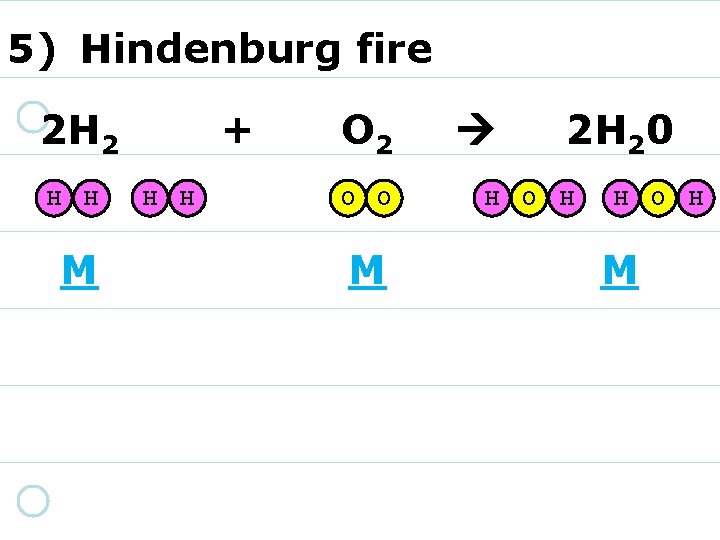
5) Hindenburg fire 2 H 2 H H M + H H O 2 O O M 2 H 20 H O H M

6) Molecules are composed of two or more atoms that are joined together.

7) Rust 4 Fe + 3 O 2 2 Fe 203

7) Rust 4 Fe Fe Fe + Fe 3 O 2 2 Fe 203 Fe 1) MOVE your marshmallows into place as needed! 2) You DO NOT need to draw them!

7) Rust 4 Fe Fe Fe + Fe Fe O O 3 O 2 O O 1) DON’T GET any more! 2) REARRANGE them instead. 2 Fe 203

7) Rust 4 Fe Fe Fe + Fe Fe O O 3 O 2 O O 2 Fe 203 O Fe O

7) Rust 4 Fe Fe Fe + Fe Fe O O 3 O 2 O O 2 Fe 203 O Fe O A? M?

7) Rust 4 Fe Fe Fe + Fe Fe O O 3 O 2 O O 2 Fe 203 O Fe O A A? M?

7) Rust 4 Fe Fe Fe + Fe Fe O O 3 O 2 O O 2 Fe 203 O Fe O A M A? M?

7) Rust 4 Fe Fe Fe + Fe Fe O O 3 O 2 O O 2 Fe 203 O Fe O A M M
8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2

8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca 1) MOVE your marshmallows into place as needed! 2) You DO NOT need to draw them!
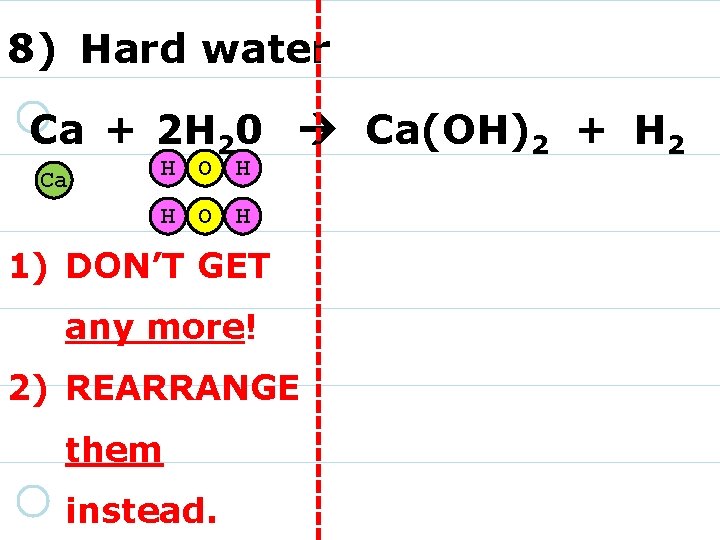
8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H 1) DON’T GET any more! 2) REARRANGE them instead.
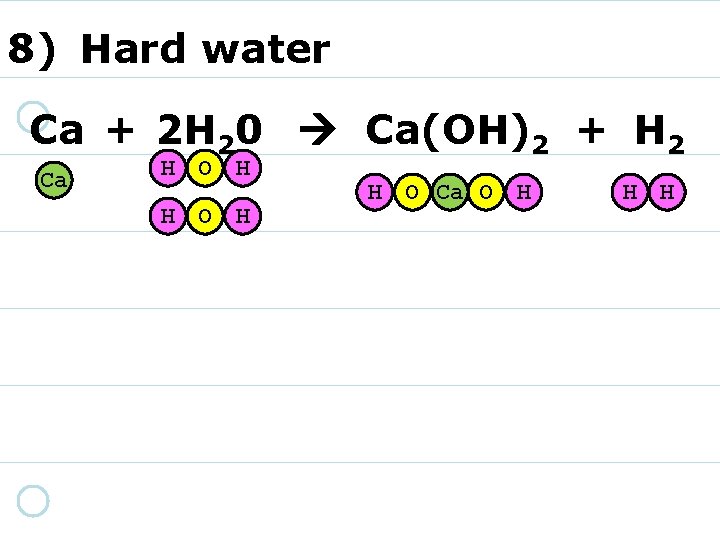
8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H H O Ca O H H H
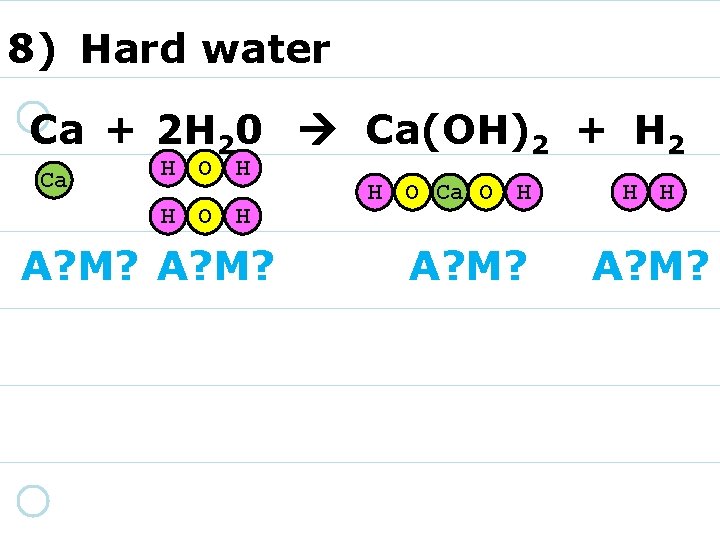
8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H A? M? H O Ca O H A? M? H H A? M?

8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H A A? M? H O Ca O H A? M? H H A? M?
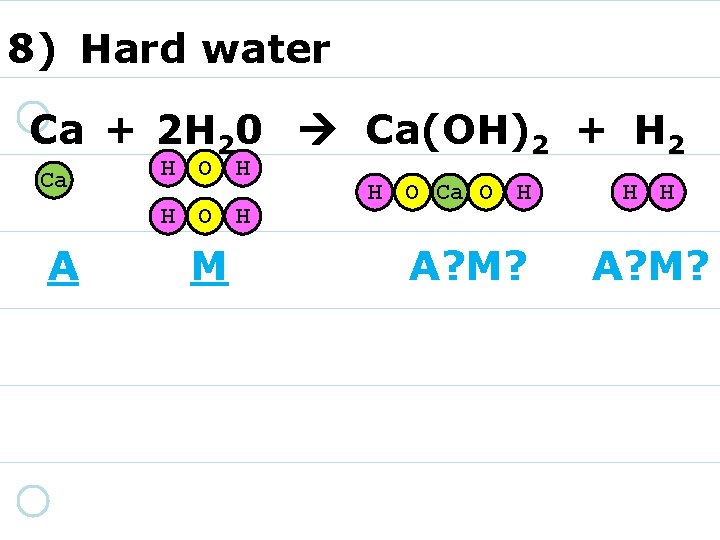
8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H A M H O Ca O H A? M? H H A? M?

8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H A M H O Ca O H M H H A? M?
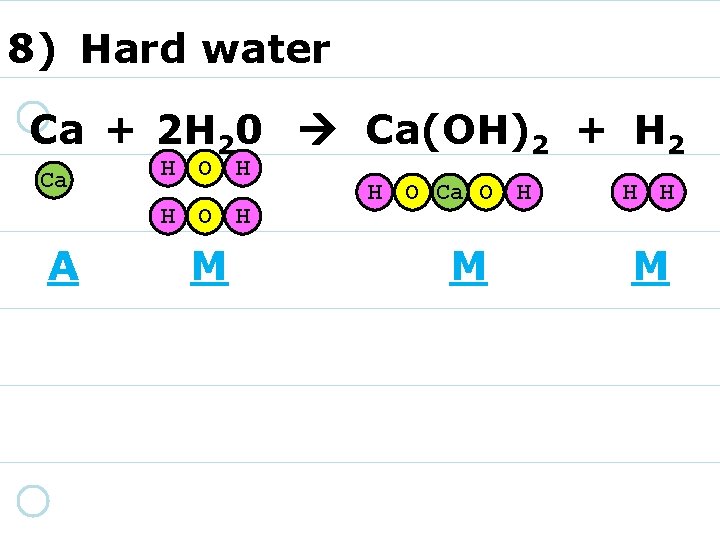
8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H A M H O Ca O H M H H M
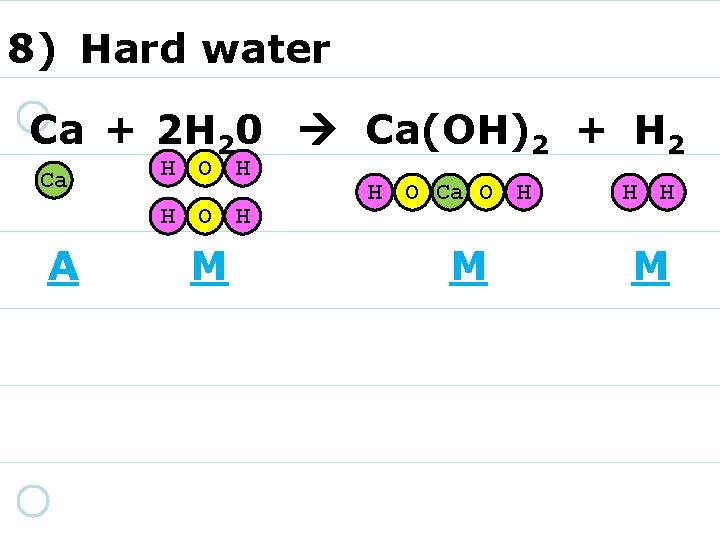
8) Hard water Ca + 2 H 20 Ca(OH)2 + H 2 Ca H O H A M H O Ca O H M H H M

EAT or DISCARD the marshmallows.

9) Write A , M tom olecule or N a) 24 K gold necklace b) ice cube c) Pepsi d) “lead” in a pencil e) diamond f) sugar either for each!

9) Write A , M tom olecule or N either for each! a) 24 K gold necklace = A (pure Au) b) ice cube c) Pepsi d) “lead” in a pencil e) diamond f) sugar

9) Write A , M tom olecule or N either for each! a) 24 K gold necklace = A (pure Au) b) ice cube = M (H 2 O) c) Pepsi d) “lead” in a pencil e) diamond f) sugar

9) Write A , M tom olecule or N either for each! a) 24 K gold necklace = A (pure Au) b) ice cube = M (H 2 O) c) Pepsi = N (water, sugar, etc. ) d) “lead” in a pencil e) diamond f) sugar

9) Write A , M tom olecule or N either for each! a) 24 K gold necklace = A (pure Au) b) ice cube = M (H 2 O) c) Pepsi = N (water, sugar, etc. ) d) “lead” in a pencil = A (pure C) e) diamond f) sugar

9) Write A , M tom olecule or N either for each! a) 24 K gold necklace = A (pure Au) b) ice cube = M (H 2 O) c) Pepsi = N (water, sugar, etc. ) d) “lead” in a pencil = A (pure C) e) diamond = A (pure C) f) sugar

9) Write A , M tom olecule or N either for each! a) 24 K gold necklace = A (pure Au) b) ice cube = M (H 2 O) c) Pepsi = N (water, sugar, etc. ) d) “lead” in a pencil = A (pure C) e) diamond = A (pure C) f) sugar = M (pure C 6 H 12 O 6)

9) Write A , M tom g) salt h) helium i) electron j) platinum k) air l) steam olecule or N either for each!

9) Write A , M tom olecule g) salt = M (Na. Cl) h) helium i) electron j) platinum k) air l) steam or N either for each!

9) Write A , M tom olecule or N either g) salt = M (Na. Cl) h) helium = A (pure He) i) electron j) platinum k) air l) steam for each!

9) Write A , M tom olecule or N either for each! g) salt = M (Na. Cl) h) helium = A (pure He) i) electron = N (too small!) j) platinum k) air l) steam

9) Write A , M tom olecule or N either for each! g) salt = M (Na. Cl) h) helium = A (pure He) i) electron = N (too small!) j) platinum = A (pure Pt) k) air l) steam

9) Write A , M tom olecule or N either for each! g) salt = M (Na. Cl) h) helium = A (pure He) i) electron = N (too small!) j) platinum = A (pure Pt) k) air = N (N 2, O 2, H 2 O, Ar, etc. ) l) steam

9) Write A , M tom olecule or N either for each! g) salt = M (Na. Cl) h) helium = A (pure He) i) electron = N (too small!) j) platinum = A (pure Pt) k) air = N (N 2, O 2, H 2 O, Ar, etc. ) l) steam = M (H 2 O)

9) Write A , M tom olecule m) coal n) baking soda o) copper tubing p) proton q) carbon dioxide r) electricity or N either for each!

9) Write A , M tom olecule or N m) coal = A (pure C) n) baking soda o) copper tubing p) proton q) carbon dioxide r) electricity either for each!

9) Write A , M tom olecule or N either for each! m) coal = A (pure C) n) baking soda = M (Na. HCO 3) o) copper tubing p) proton q) carbon dioxide r) electricity

9) Write A , M tom olecule or N either for each! m) coal = A (pure C) n) baking soda = M (Na. HCO 3) o) copper tubing = A (pure Cu) p) proton q) carbon dioxide r) electricity

9) Write A , M tom olecule or N either for each! m) coal = A (pure C) n) baking soda = M (Na. HCO 3) o) copper tubing = A (pure Cu) p) proton = N (too small!) q) carbon dioxide r) electricity

9) Write A , M tom olecule or N either for each! m) coal = A (pure C) n) baking soda = M (Na. HCO 3) o) copper tubing = A (pure Cu) p) proton = N (too small!) q) carbon dioxide = M (CO 2) r) electricity

9) Write A , M tom olecule or N either for each! m) coal = A (pure C) n) baking soda = M (Na. HCO 3) o) copper tubing = A (pure Cu) p) proton = N (too small!) q) carbon dioxide = M (CO 2) r) electricity = N (pure energy!)

The End
- Slides: 69