Atoms Molecules Ions AP Chemistry Chapter 2 Subatomic

Atoms, Molecules & Ions AP Chemistry Chapter 2

Subatomic Particles • Protons and electrons are the only particles that have a charge. • Protons and neutrons have essentially the same mass. • The mass of an electron is so small we ignore it.

Symbols of Elements are symbolized by one or two letters.

Atomic Number All atoms of the same element have the same number of protons: The atomic number (Z) In a neutral atom, the # of p+ = the # of e-

Atomic Mass The mass of an atom in atomic mass units (amu) is the total number of protons and neutrons in the atom. Remember, electron mass is so tiny we essentially ignore it.

Isotopes • Atoms of the same element with different masses. • Isotopes have different numbers of neutrons. 11 C 6 12 C 6 13 C 6 14 C 6
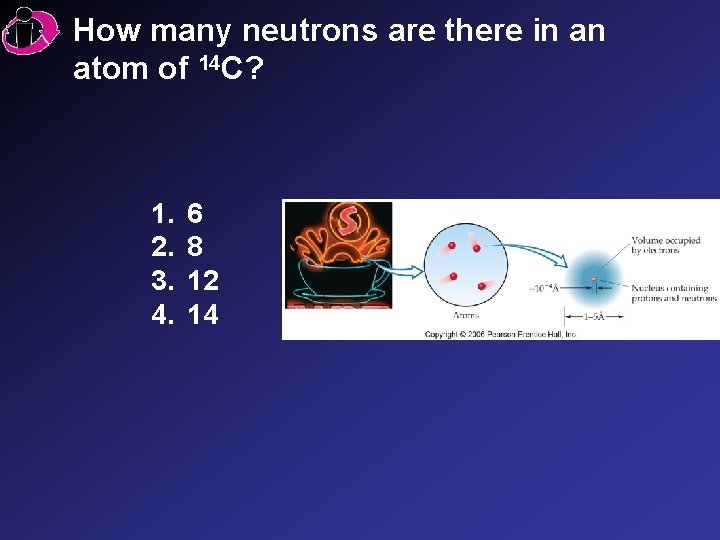
How many neutrons are there in an atom of 14 C? 1. 2. 3. 4. 6 8 12 14

Correct Answer: 1. 6 2. 8 3. 12 4. 14 Mass number 14 Atomic number 6 C The difference between the mass number and the atomic number is the number of neutrons (14 6) = 8.

Determining the Number of Subatomic Particles in Atoms How many protons, neutrons, and electrons are in (a) an atom of 197 Au (b) an atom of strontium -90?

Determining the Number of Subatomic Particles in Atoms How many protons, neutrons, and electrons are in (a) a 138 Ba atom, (b) an atom of phosphorus-31? Answer: (a) 56 protons, 56 electrons, and 82 neutrons; (b) 15 protons, 15 electrons, and 16 neutrons.

Periodic Table • A systematic catalog of elements. • Elements are arranged in order of atomic number.
Periodicity When one looks at the chemical properties of elements, one notices a repeating pattern of reactivities.
Periodic Table • The rows on the periodic chart are periods or series. • Columns are groups or families. • Elements in the same group have similar chemical properties.

Groups These five groups are known by their names.
Solids, Liquids, & Gases
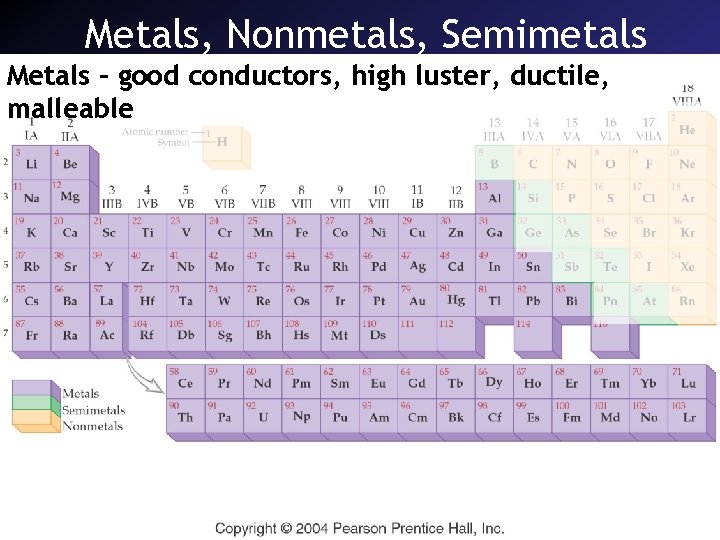
Metals, Nonmetals, Semimetals Metals – good conductors, high luster, ductile, malleable
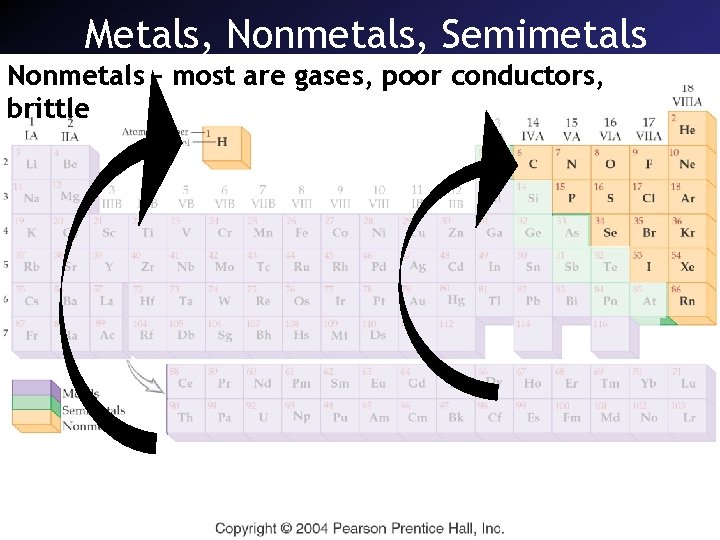
Metals, Nonmetals, Semimetals Nonmetals – most are gases, poor conductors, brittle
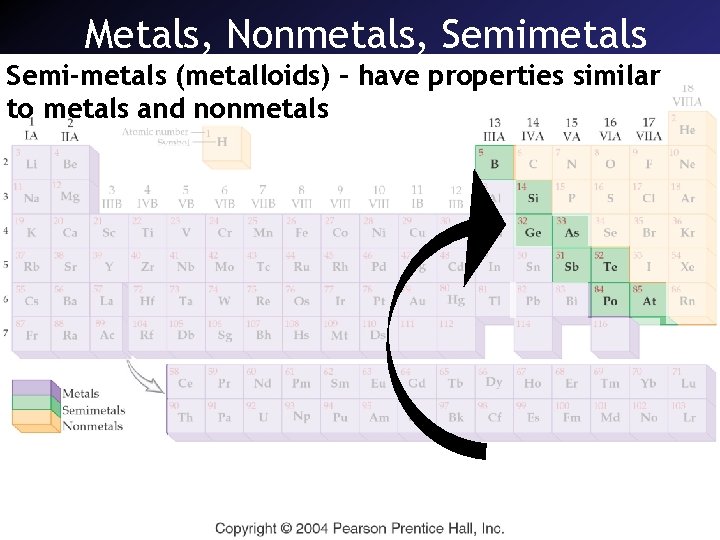
Metals, Nonmetals, Semimetals Semi-metals (metalloids) – have properties similar to metals and nonmetals

Diatomic Molecules These seven elements occur naturally as molecules containing two atoms: • Hydrogen & draw a 7 starting at N (element 7) • Hockey stick & puck • Br. INCl. HOF (Brinklehoff) • I Have No Bright Or Clever Friends

Compounds and Chemical Formulas • Molecule - A single particle, contains two or more nonmetal atoms; covalently bonded • Formula Unit – a particle containing a metal with a nonmetal; ionicly bonded • Chemical formulas – give the number of atoms of each element in a compound

Identifying Ionic and Molecular Compounds Which of the following compounds would you expect to be ionic: N 2 O, Na 2 O, Ca. Cl 2, SF 4? Which of the following compounds are molecular: CBr 4, Fe. S, P 4 O 6, Pb. F 2 ? Answer: CBr 4 and P 4 O 6

Identifying Ionic and Molecular Compounds Which of the following compounds are molecular: CBr 4, Fe. S, P 4 O 6, Pb. F 2 ? Answer: CBr 4 and P 4 O 6

Which combination is likely to produce an ionic compound? • • • C and H S and Cl Ca and F Br and I Xe and F

Which combination is likely to produce an ionic compound? • • • C and H S and Cl Ca and F Br and I Xe and F
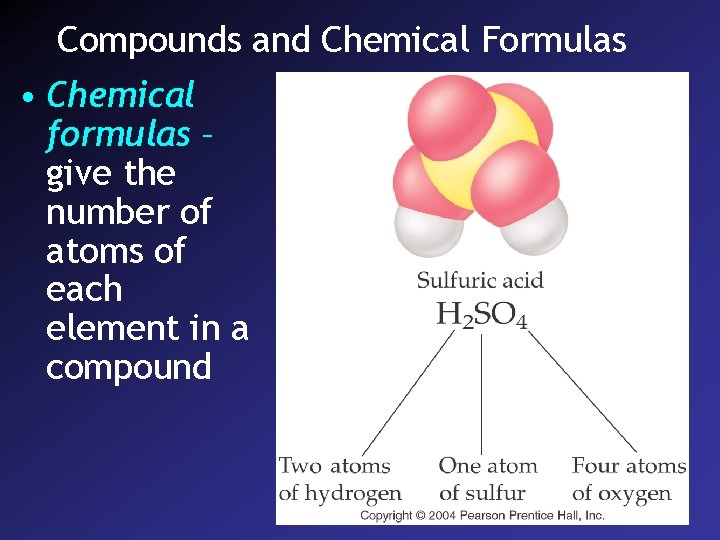
Compounds and Chemical Formulas • Chemical formulas – give the number of atoms of each element in a compound

Compounds and Chemical Formulas State the composition for a molecule of trinitrotoluene, TNT: C 7 H 5(NO 2)3 • 7 carbon atoms, 5 hydrogen atoms, 3 nitrogen atoms, 6 oxygen atoms; 21 atoms total

Types of Formulas • Empirical formulas give the lowest wholenumber ratio of atoms of each element in a compound. • Molecular formulas give the exact number of atoms of each element in a compound. • Sometimes they can be the same

Relating Empirical and Molecular Formulas Write the empirical formulas for the following molecules: (a) glucose, a substance also known as either blood sugar or dextrose, whose molecular formula is C 6 H 12 O 6; (b) nitrous oxide, a substance used as an anesthetic and commonly called laughing gas, whose molecular formula is N 2 O. Answer: BH 3 Give the empirical formula for the substance called diborane, whose molecular formula is B 2 H 6.

Relating Empirical and Molecular Formulas Give the empirical formula for the substance called diborane, whose molecular formula is B 2 H 6. Answer: BH 3

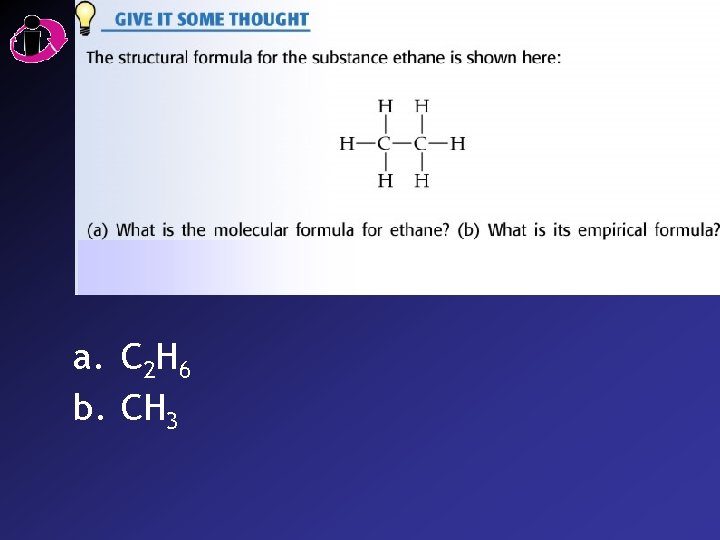
a. C 2 H 6 b. CH 3

Ions • When atoms lose or gain electrons, they become ions.

Ions • Cation – positive ion – Has the “t” like a + sign – Formed by metals and some nonmetals – Lost electrons – #p+ > #e • Anion – negative ion – Gained electrons – Formed by nonmetals – #p+ < #e • Polyatomic ions – Made up of two or more atoms – Can be positive or negative
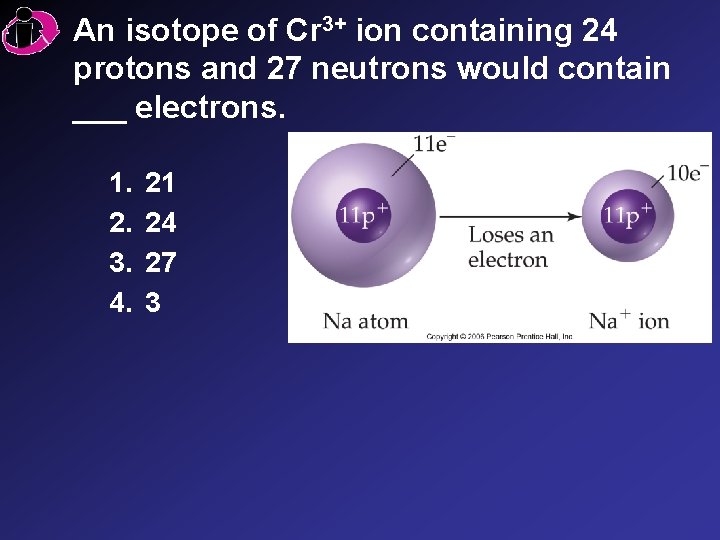
An isotope of Cr 3+ ion containing 24 protons and 27 neutrons would contain ___ electrons. 1. 2. 3. 4. 21 24 27 3

Correct Answer: 1. 2. 3. 4. 21 24 27 3 The +3 charge indicates there are 3 more protons than electrons: 24 3 = 21

Which of the following species has the greatest number of electrons? 1. • • Cl Ca 2+ P 3 K Ar

Correct Answer: 1. • • Cl Ca 2+ P 3 K Ar K has 19 electrons; all the other species listed have only 18 electrons.

Writing Chemical Symbols for Ions Give the chemical symbol, including mass number, for each of the following ions: (a) The ion with 22 protons, 26 neutrons, and 19 electrons; (b) the ion of sulfur that has 16 neutrons and 18 electrons. Solution (a) The symbol for the ion is (b) The symbol for the ion is 32 S 2–. 48 Ti 3+.

Writing Chemical Symbols for Ions How many protons and electrons does the Se 2– ion possess? Answer: 34 protons and 36 electrons

Isoelectronic Atoms and ions are considered isoelectronic when they have the same number of electrons.

Which of the following are isoelectronic? 1. 2. 3. 4. 5. None of the above

Which of the following are isoelectronic? 1. 2. 3. 4. 5. None of the above
Group 1 A = 1+
Group 2 A = 2+
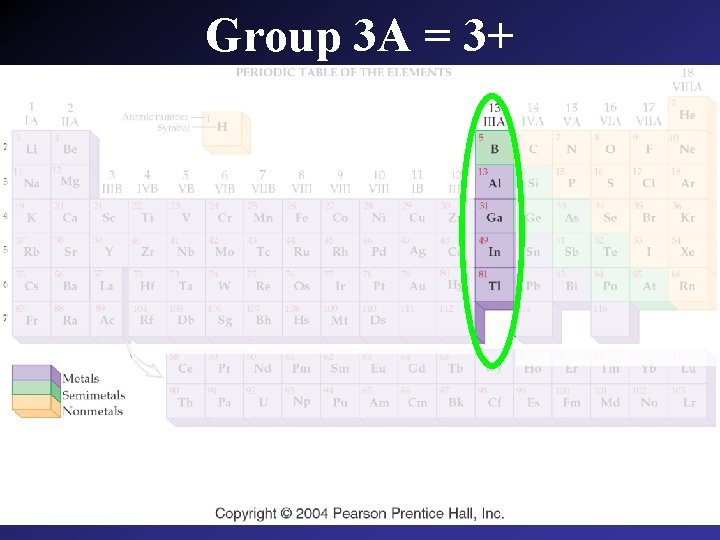
Group 3 A = 3+
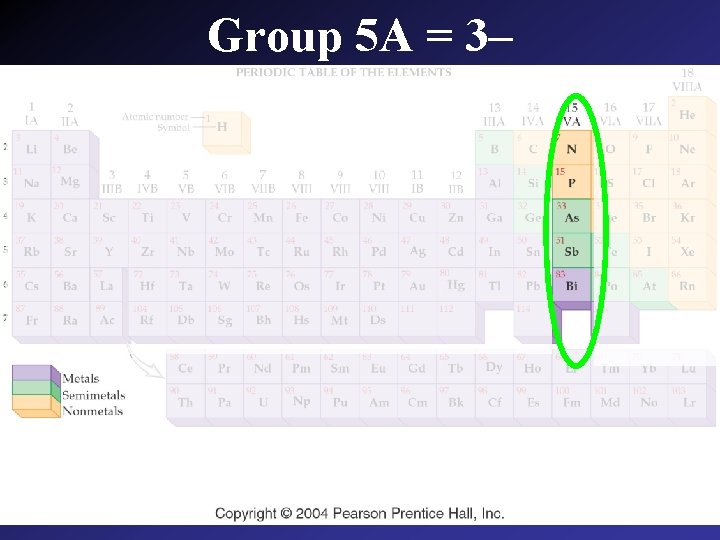
Group 5 A = 3–
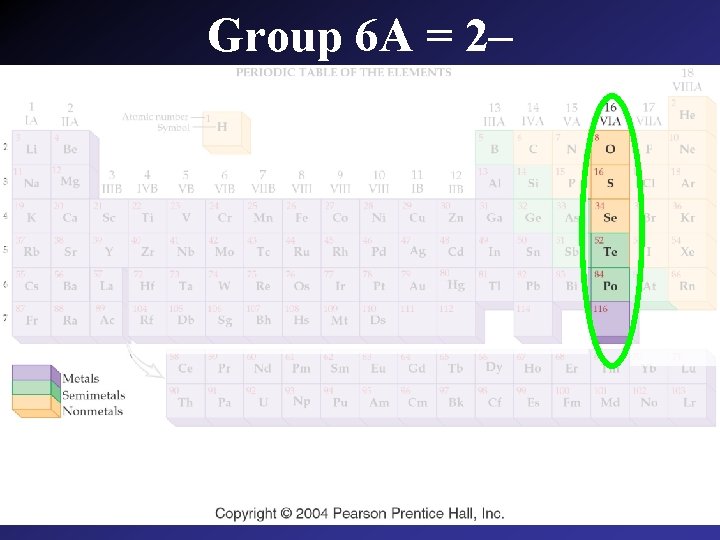
Group 6 A = 2–
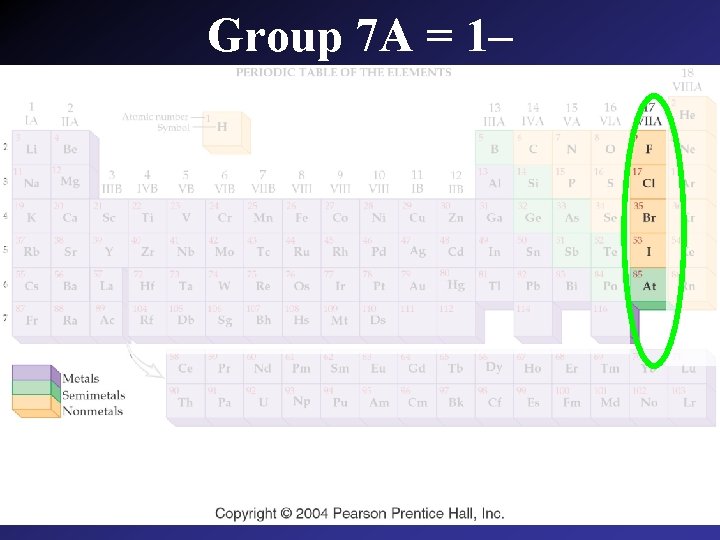
Group 7 A = 1–
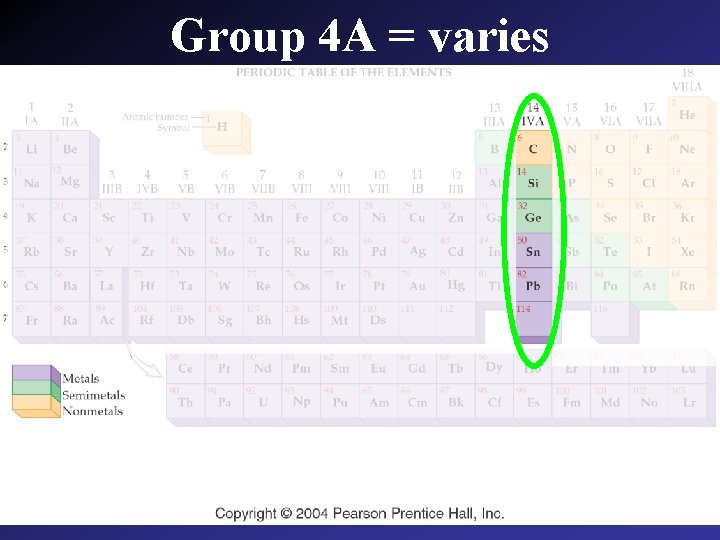
Group 4 A = varies

Monoatomic Ions • Transition metals form more than one cation The Stock System • Iron can be Fe 2+ or Fe 3+ • Iron (II) ion, iron (III) ion • Cu+ and Cu 2+

Polyatomic Ions • Polyatomic anions usually contain one or more elements combined with oxygen • Most end in “-ate” • Those that end in “-ite” have one less oxygen than “-ate” • Exceptions include – CN 1 -, cyanide – OH 1 -, hydroxide – O 22 -, peroxide – NH 41+, ammonium

Patterns in Oxyanion Nomenclature • When there are two oxyanions involving the same element: – The one with fewer oxygens ends in -ite • NO 2− : nitrite; SO 32− : sulfite – The one with more oxygens ends in -ate • NO 3− : nitrate; SO 42− : sulfate

Writing Chemical Formulas • A formula unit is the simplest representative particle in an ionic compound • A formula unit is neutral • Positive and negative charges must cancel out (cation + anion) • Made between a metal & a nonmetal

Naming Formula units • The metal is written first followed by the nonmetal • The metal keeps its name, the nonmetal gets the suffix “-ide” Mg. O magnesium oxide Na. Cl sodium chloride Al 2 O 3 aluminum oxide Ca. S calcium sulfide K 3 N potassium nitride

Formula units from names • • • All formula units have a neutral charge The + and the – must = zero Beryllium fluoride Potassium chloride Strontium nitride

The correct name for the compound, Mg. C 2 is 1. 2. 3. 4. 5. Carbon magneside Magnesium carbonate Manganese dicarbide Magnesium dicarbide

Correct Answer: 1. 2. 3. 4. 5. Carbon magneside Magnesium carbonate Manganese dicarbide Magnesium dicarbide In this unusual ionic compound, the cation is magnesium cation (Mg 2+) and the anion is carbide or acetylide (C 22 ).

The simplest chemical formula for the binary ionic compound Cax. Ny is 1. 2. 3. 4. Ca 2 N 3 Ca 3 N 2 Ca 3 N

Correct Answer: 1. 2. 3. 4. Ca 2 N 3 Ca 3 N 2 Ca 3 N Ca loses two electrons to become Ca 2+, while N gains three electrons to become N 3. To be neutral, positive charges and negative charges have to balance.

Transition metals in formula units • Transition metals have multiple oxidation numbers (aka charges) • Exceptions: Ag+ Cd 2+ Zn 2+ • Must figure out charges from name or formula • iron (II) oxide • Cu. Cl 2

Naming with polyatomics • Polyatomics end mostly in “-ate” and “-ite” – Exceptions: ammonium (NH 4+), hydroxide (OH-), cyanide (CN-), & peroxide (O 2 -2) • A formula with a polyatomic will have 3 or more different elements. • Only one positive polyatomic: NH 4+ ammonium • Ca(NO 3)2 • K 2 SO 4 • aluminum acetate • sodium phosphate

The compound, Cu. SO 4, is called 1. 2. 3. 4. 5. Copper Copper sulfate (II) sulfide (I) sulfite (I) sulfate (II) sulfate

Correct Answer: 1. 2. 3. 4. 5. Copper Copper sulfate (II) sulfide (I) sulfite (I) sulfate (II) sulfate The compound is ionic, derived from the copper(II) cation (Cu 2+) and the sulfate anion 2 (SO 4 ).

Using Ionic Charge to Write Empirical Formulas for Ionic Compounds What are the empirical formulas of the compounds formed by (a) Al 3+ and Cl– ions, (b) Al 3+ and O 2 – ions, (c) Mg 2+ and NO 3– ions? (a) The formula is Al. Cl 3. (b) The formula is Al 2 O 3. (c) The formula is Mg(NO 3)2.

Using Ionic Charge to Write Empirical Formulas for Ionic Compounds Write the empirical formulas for the compounds formed by the following ions: (a) Na+ and PO 43– , (b) Zn 2+ and SO 42– , (c) Fe 3+ and CO 32–. Answers: (a) Na 3 PO 4, (b) Zn. SO 4, (c) Fe 2(CO 3)3

Determining the Names of Ionic Compounds from Their Formulas Name the following compounds: (a) K 2 SO 4 , (b) Ba(OH)2 , (c) Fe. Cl 3. (a) potassium sulfate (b) barium hydroxide (c) Iron (III) chloride

Determining the Names of Ionic Compounds from Their Formulas Name the following compounds: (a) NH 4 Br, (b) Cr 2 O 3, (c) Ca(NO 3)2. Answers: (a) ammonium bromide, (b) chromium(III) oxide, (c) cobalt(II) nitrate

Determining the Formulas of Ionic Compounds from Their Names Write the chemical formulas for the following compounds: (a) potassium sulfide, (b) calcium hydrogen carbonate, (c) nickel(II) perchlorate. (a) K 2 S (b) Ca(HCO 3)2 (c) Ni(Cl. O 4)2.

Determining the Formulas of Ionic Compounds from Their Names Give the chemical formula for (a) magnesium sulfate, (b) silver sulfide, (c) lead(II) nitrate. Answers: (a) Mg. SO 4 , (b) Ag 2 S, (c) Pb(NO 3)2

Nomenclature of Binary Molecular Compounds • The less electronegative atom is usually listed first. • A prefix is used to denote the number of atoms of each element in the compound (monois not used on the first element listed, however. )

Nomenclature of Binary Molecular Compounds • The ending on the more electronegative element is changed to -ide. – CO 2: carbon dioxide – CCl 4: carbon tetrachloride

Nomenclature of Binary Compounds If the prefix ends with a or o and the name of the element begins with a vowel, the two successive vowels are often elided into one: N 2 O 5: dinitrogen pentoxide

Molecular formulas • • NO 2 P 4 S 10 NI 3 Cl 2 O CO Xenon hexafluoride Diphosphorus pentaoxide

Elements by themselves • Solo elements are always just named for themselves no matter how many there are • N 2 • S 8 • Fe

The molecular compound N 2 O 4 is named: 1. 2. 3. 4. 5. Nitrogen oxide Oxygen nitride Dinitrogen quartoxide Dinitrogen tetroxide Nitrous oxide

Correct Answer: 1. 2. 3. 4. 5. Nitrogen oxide Oxygen nitride Dinitrogen quartoxide Dinitrogen tetroxide Nitrous oxide Remember: the vowel “A” on the prefix tetra- is dropped because oxide starts with the vowel “O. ”

Relating the Names and Formulas of Binary Molecular Compounds Name the following compounds: (a) SO 2, (b) PCl 5, (c) N 2 O 3. (a) sulfur dioxide, (b) phosphorus pentachloride, and (c) dinitrogen trioxide.

Relating the Names and Formulas of Binary Molecular Compounds Give the chemical formula for (a) silicon tetrabromide, (b) disulfur dichloride. Answers: (a) Si. Br 4, (b) S 2 Cl 2

Relating the Names and Formulas of Acids Name the following acids: (a) HCN, (b) HNO 3, (c) H 2 SO 4, (d) H 2 SO 3. (a) hydrocyanic acid (b) nitric acid (c) sulfuric acid. (d) sulfurous acid

Relating the Names and Formulas of Acids Give the chemical formulas for (a) hydrobromic acid, (b) carbonic acid. Answers: (a) HBr, (b) H 2 CO 3

Acid Nomenclature • If the anion in the acid ends in -ide, change the ending to -ic acid and add the prefix hydro- : – HCl: hydrochloric acid – HBr: hydrobromic acid – HI: hydroiodic acid

Acid Nomenclature • If the anion in the acid ends in -ite, change the ending to -ous acid: – HCl. O: hypochlorous acid – HCl. O 2: chlorous acid

Acid Nomenclature • If the anion in the acid ends in -ate, change the ending to -ic acid: – HCl. O 3: chloric acid – HCl. O 4: perchloric acid
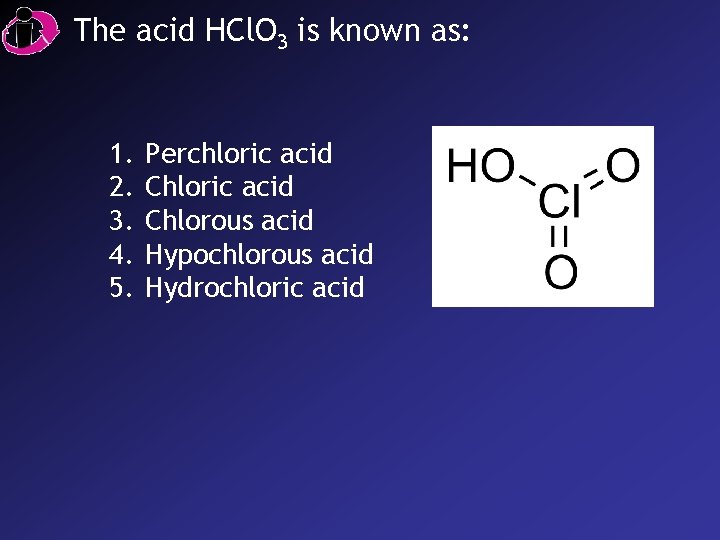
The acid HCl. O 3 is known as: 1. 2. 3. 4. 5. Perchloric acid Chlorous acid Hypochlorous acid Hydrochloric acid
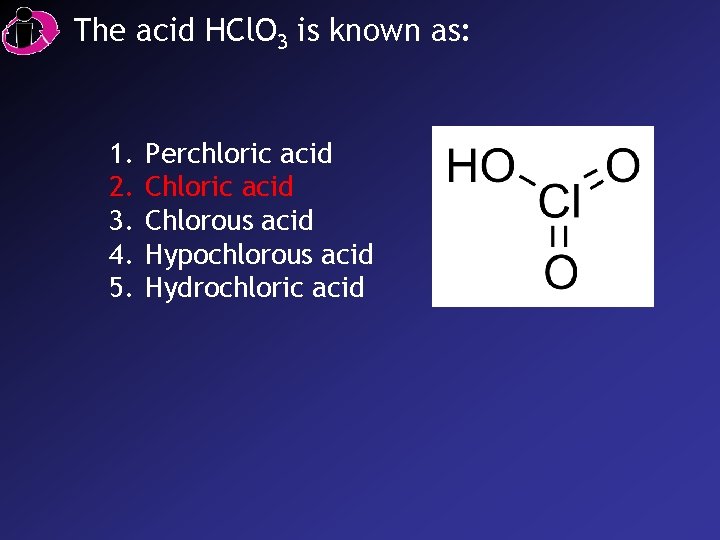
The acid HCl. O 3 is known as: 1. 2. 3. 4. 5. Perchloric acid Chlorous acid Hypochlorous acid Hydrochloric acid

Simple Organic Chemistry and Hydrocarbons • Hydrocarbons contain only two elements: 1) hydrogen, & 2) carbon – simplest hydrocarbons called “alkanes”, which contain only carbon to carbon single covalent bonds (Cn. H 2 n+2) – methane (CH 4) with one carbon is the simplest alkane. It is the major component of natural gas

Simple Organic Chemistry and Hydrocarbons 1. 2. 3. 4. 5. methethpropbutpent- many elephants pee by plants 6. hex 7. hept 8. oct 9. non 10. dec-

Alcohols • Alcohols - a class of organic compounds with an -OH group – The -OH functional group in alcohols is called a “hydroxyl” group; thus R-OH is the formula • How is this different from the hydroxide ion? (covalent bonding with the carbon- not ionic with a metal like bases)

Alcohols • For IUPAC naming: – drop the -e ending of the parent alkane name; add ending of -ol, number the position of –OH – The hydroxyl is given the lowest position number (1 -propanol, 2 propanol)

Properties of Alcohols • Many aliphatic alcohols used in laboratories, clinics, and industry – Isopropyl alcohol (2 -propanol) is rubbing alcohol; used as antiseptic, and a base for perfume, creams, lotions, and other cosmetics

Properties of Alcohols • Ethyl alcohol (ethanol) used in the intoxicating beverages; also an important industrial solvent
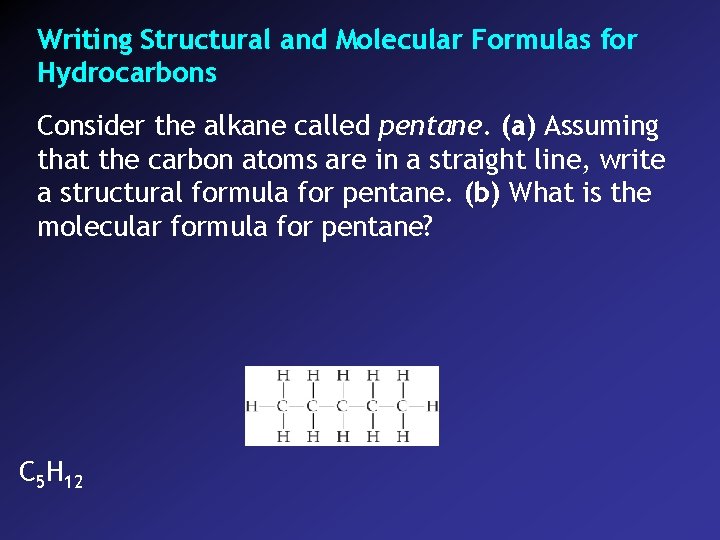
Writing Structural and Molecular Formulas for Hydrocarbons Consider the alkane called pentane. (a) Assuming that the carbon atoms are in a straight line, write a structural formula for pentane. (b) What is the molecular formula for pentane? C 5 H 12

Writing Structural and Molecular Formulas for Hydrocarbons Butane is the alkane with four carbon atoms. (a) What is the molecular formula of butane? (b) What are the name and molecular formula of an alcohol derived from butane? Answers: (a) C 4 H 10 , (b) butanol, C 4 H 10 O or C 4 H 9 OH
- Slides: 93